Scalable Synthesis of Olmesartan Medoxomil Intermediates via Novel Tetrazole Protection Strategies
The pharmaceutical industry continuously seeks more efficient pathways for producing high-volume cardiovascular medications, and the synthesis of Olmesartan Medoxomil, a potent Angiotensin II receptor antagonist, remains a critical focus area. Patent CN103319461A introduces a groundbreaking preparation method for key olmesartan medoxomil intermediates, specifically addressing the longstanding challenges associated with tetrazole protection groups. This innovation offers a robust alternative to traditional trityl-based or SEM-based protection strategies, which have historically plagued manufacturers with complex purification requirements and high reagent costs. By employing a 1-methyl-1-phenylethyl protecting group, the disclosed technology streamlines the synthetic route, enhancing both the economic viability and the chemical purity of the final active pharmaceutical ingredient (API). For global procurement teams and R&D directors, understanding this technological shift is essential for securing a reliable olmesartan medoxomil intermediate supplier capable of delivering cost-effective solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of olmesartan medoxomil has relied heavily on the use of trityl (triphenylmethyl) groups to protect the tetrazole ring during the construction of the biphenyl scaffold. While effective for protection, the removal of the trityl group necessitates harsh acidic conditions that often lead to the formation of stubborn impurities such as triphenylcarbinol and various MTT-Eliminate byproducts. These impurities are notoriously difficult to separate from the desired product, requiring extensive chromatographic purification or multiple recrystallization steps that drastically reduce overall yield and increase manufacturing costs. Alternatively, some processes utilize 2-(trimethylsilyl)ethoxymethyl (SEM) groups; however, the high cost of SEM reagents and the complexity of their handling make this approach economically unattractive for large-scale commercial production. Consequently, there is a pressing industry need for a simpler, more economical synthetic method that eliminates these bottlenecks without compromising product quality.
The Novel Approach
The methodology described in the patent overcomes these deficiencies by introducing a 1-methyl-1-phenylethyl group as the tetrazole protecting moiety, which offers superior stability during synthesis and easier removal in the final stages. This novel approach begins with the synthesis of a specialized biphenyl tetrazole intermediate (Compound 2) through a sequence involving azide reaction, addition with alpha-methyl styrene, and bromination. 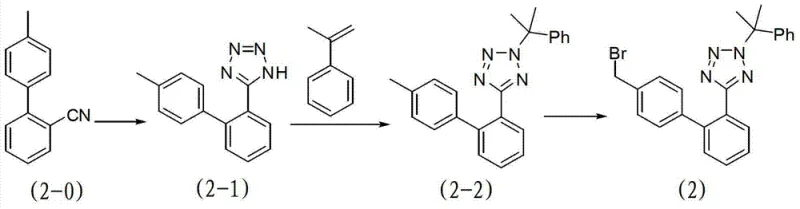 This building block is then coupled with an imidazole precursor under mild basic conditions, avoiding the degradation issues seen with acid-sensitive groups. The resulting intermediate can be deprotected using either mild acid hydrolysis or catalytic hydrogenation, pathways that generate volatile or easily separable byproducts, thereby significantly simplifying the downstream purification process and ensuring a high-purity final product suitable for pharmaceutical applications.
This building block is then coupled with an imidazole precursor under mild basic conditions, avoiding the degradation issues seen with acid-sensitive groups. The resulting intermediate can be deprotected using either mild acid hydrolysis or catalytic hydrogenation, pathways that generate volatile or easily separable byproducts, thereby significantly simplifying the downstream purification process and ensuring a high-purity final product suitable for pharmaceutical applications.
Mechanistic Insights into Phase Transfer Catalyzed Condensation
The core of this synthetic advancement lies in the efficient coupling of the protected tetrazole biphenyl bromide (Compound 2) with the imidazole lactone derivative (Compound 3). This condensation reaction is facilitated by the presence of an alkaline carbonate base, such as potassium carbonate or sodium carbonate, in a polar organic solvent environment like acetone or acetonitrile. Crucially, the reaction employs a quaternary ammonium salt, such as tetrabutylammonium bromide, acting as a phase transfer catalyst to enhance the nucleophilicity of the imidazole nitrogen. 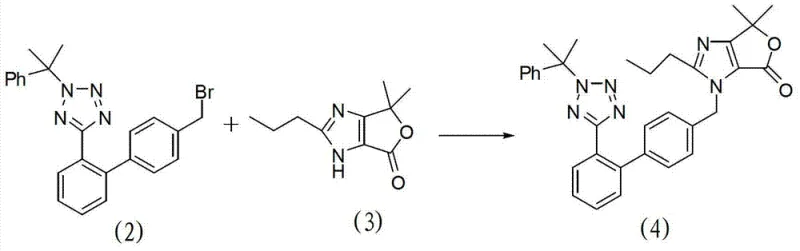 The mechanism involves the deprotonation of the imidazole nitrogen followed by a nucleophilic substitution on the benzylic bromide of the tetrazole component. This step is optimized to proceed at moderate temperatures between 50°C and 80°C, ensuring high conversion rates while minimizing thermal degradation of the sensitive tetrazole ring. The subsequent hydrolysis of the lactone ring to the carboxylic acid (Compound 1) is achieved using caustic alkali in alcoholic solvents, a straightforward transformation that preserves the integrity of the newly formed biphenyl-imidazole linkage.
The mechanism involves the deprotonation of the imidazole nitrogen followed by a nucleophilic substitution on the benzylic bromide of the tetrazole component. This step is optimized to proceed at moderate temperatures between 50°C and 80°C, ensuring high conversion rates while minimizing thermal degradation of the sensitive tetrazole ring. The subsequent hydrolysis of the lactone ring to the carboxylic acid (Compound 1) is achieved using caustic alkali in alcoholic solvents, a straightforward transformation that preserves the integrity of the newly formed biphenyl-imidazole linkage.
Impurity control is inherently built into this mechanistic design by avoiding the generation of non-volatile organic residues common in trityl chemistry. The use of the 1-methyl-1-phenylethyl group ensures that upon deprotection, the cleaved fragments are either soluble in aqueous washes or can be removed via filtration, preventing the carryover of genotoxic or mutagenic impurities into the final API. Furthermore, the esterification step to form the prodrug intermediate (Compound A) utilizes benzotriazole activation, which provides a highly reactive acylating species that reacts selectively with the hydroxymethyl dioxolone moiety. This specificity reduces the formation of regioisomers and over-acylated byproducts, resulting in a cleaner crude product that requires minimal refinement. Such mechanistic precision is vital for meeting the stringent regulatory standards required for cardiovascular drug substances.
How to Synthesize Olmesartan Medoxomil Intermediate Efficiently
The practical execution of this synthesis involves a series of well-defined unit operations that balance reaction kinetics with ease of workup. The process initiates with the preparation of the tetrazole building block, followed by the critical coupling step and final functionalization. Each stage is designed to maximize yield while maintaining safety and environmental compliance. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles derived from the patent embodiments, are outlined below to guide process engineers in replicating this high-efficiency route.
- Synthesize the protected tetrazole biphenyl bromide (Compound 2) via azide reaction, addition with alpha-methyl styrene, and bromination.
- Perform condensation between Compound 2 and the imidazole lactone (Compound 3) using potassium carbonate and a phase transfer catalyst in acetone.
- Hydrolyze the resulting lactone to the carboxylic acid (Compound 1) and subsequently esterify with 4-hydroxymethyl-5-methyl-1,3-dioxol-2-one to form the final prodrug intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into tangible operational benefits and risk mitigation. By eliminating the reliance on expensive and supply-constrained reagents like SEM-Cl, manufacturers can achieve significant cost reduction in API manufacturing. The simplified purification protocols reduce the consumption of chromatography media and solvents, lowering both material costs and waste disposal expenses. Furthermore, the robustness of the reaction conditions allows for flexible scheduling and reduced batch cycle times, enhancing the overall responsiveness of the supply chain to market demands for antihypertensive medications.
- Cost Reduction in Manufacturing: The replacement of the trityl group with the 1-methyl-1-phenylethyl group eliminates the need for complex and costly purification steps required to remove triphenylcarbinol impurities. This structural change allows for the use of cheaper starting materials and reduces the number of processing units needed, leading to substantial cost savings in the overall production budget. Additionally, the avoidance of precious metal catalysts in certain deprotection variants further decreases the direct material costs associated with the synthesis.
- Enhanced Supply Chain Reliability: The starting materials for this route, such as 2-cyano-4'-methylbiphenyl and alpha-methyl styrene, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The synthetic pathway does not depend on specialized reagents that are subject to volatile pricing or limited availability, ensuring a consistent and reliable supply of high-purity pharmaceutical intermediates. This stability is crucial for maintaining continuous production schedules and meeting long-term contractual obligations with downstream API manufacturers.
- Scalability and Environmental Compliance: The process utilizes common organic solvents like acetone, acetonitrile, and methylene chloride, which are easily recovered and recycled in standard industrial distillation units, supporting sustainable manufacturing practices. The reaction conditions are mild and do not require extreme pressures or temperatures, facilitating safe scale-up from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation, particularly the absence of heavy metal residues from trityl removal, simplifies environmental compliance and lowers the ecological footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of purity, yield, and operational simplicity. Understanding these nuances is key for technical teams evaluating potential technology transfers or vendor qualifications.
Q: How does the 1-methyl-1-phenylethyl protection group improve purity compared to trityl?
A: Unlike trityl groups which require harsh acidic deprotection generating difficult-to-remove triphenylcarbinol impurities, the 1-methyl-1-phenylethyl group can be removed under milder acidic conditions or via catalytic hydrogenation, significantly simplifying the purification process and improving the impurity profile.
Q: What are the key reaction conditions for the condensation step?
A: The condensation reaction is optimally performed in polar aprotic solvents like acetone or acetonitrile at temperatures between 50-80°C, utilizing alkaline carbonates such as potassium carbonate and quaternary ammonium salts like tetrabutylammonium bromide as phase transfer catalysts.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route utilizes readily available starting materials and avoids expensive reagents like SEM-Cl. The reaction conditions are robust, using common solvents and standard workup procedures, making it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olmesartan Medoxomil Intermediate Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to bring complex synthetic pathways like the one described in CN103319461A to commercial reality. Our R&D team specializes in optimizing reaction parameters to ensure maximum efficiency and purity, translating laboratory success into industrial performance. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical techniques to verify the identity and quality of every batch, guaranteeing that our olmesartan medoxomil intermediates are ready for immediate API synthesis.
We invite global pharmaceutical partners to collaborate with us to explore the full potential of this advanced manufacturing technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain. Let us be your strategic partner in delivering high-quality cardiovascular intermediates to the global market.
