Advanced One-Pot Synthesis of Olmesartan Medoxomil Intermediates for Commercial Scale-Up
Introduction to Next-Generation ARB Manufacturing
The global demand for Angiotensin II Receptor Blockers (ARBs) continues to surge as hypertension remains a critical public health challenge worldwide. At the forefront of this therapeutic class is olmesartan medoxomil, a potent prodrug known for its superior efficacy and duration of action. However, the economic viability of producing this complex molecule often hinges on the efficiency of its synthetic route. Patent CN103435602A introduces a transformative preparation method that addresses longstanding bottlenecks in the manufacturing of olmesartan medoxomil intermediates. By leveraging a telescoped one-pot strategy, this technology merges multiple discrete chemical transformations into a single continuous process flow. This innovation not only streamlines the production workflow but also significantly enhances the overall yield and purity profile of the final active pharmaceutical ingredient (API). For industry stakeholders, understanding the nuances of this patented methodology is essential for optimizing supply chains and reducing the cost of goods sold (COGS) in competitive generic markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for olmesartan medoxomil, such as those described in earlier literature like Yanagisawa et al., typically rely on a stepwise approach that necessitates the isolation of key intermediates. Specifically, the conventional pathway involves the initial alkylation of the imidazole ring, followed by a separate hydrolysis step to generate the free carboxylic acid, and finally, an esterification reaction to attach the medoxomil moiety. Each of these discrete steps requires independent work-up procedures, including solvent exchanges, drying, and purification, which cumulatively increase the operational burden. Furthermore, legacy methods often employ aggressive reagents like thionyl chloride for activation, mandating strictly anhydrous conditions that are difficult and costly to maintain on a large industrial scale. These harsh conditions can also lead to the formation of unwanted by-products, complicating downstream purification and potentially compromising the safety profile of the final drug substance.
The Novel Approach
In stark contrast, the methodology disclosed in CN103435602A utilizes a sophisticated one-pot technique that seamlessly integrates alkylation, hydrolysis, and esterification without isolating the intermediate species. This approach begins with the reaction of 4-(1-hydroxy-1-methylethyl)-2-propylimidazole-5-ethylcarboxylate and N-(triphenylmethyl)-5-(4'-bromomethyl diphenyl-2-yl)tetrazole. Instead of stopping to isolate the ethyl ester intermediate, the process proceeds directly to hydrolysis within the same reaction vessel by adjusting the alkalinity and temperature. Subsequently, the resulting carboxylate salt is immediately reacted with 4-chloromethyl-5-methyl-1,3-dioxol-2-one to form the final cyclic carbonate ester. This telescoping of reactions drastically reduces the number of unit operations, minimizes solvent usage, and eliminates the yield losses typically associated with physical transfer and isolation of unstable intermediates.
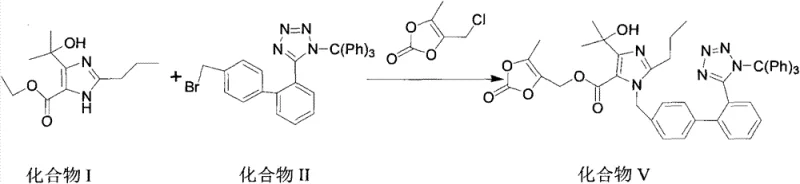
Mechanistic Insights into One-Pot Telescoped Synthesis
The core chemical ingenuity of this process lies in the precise control of reaction conditions that allow for sequential transformations in a single medium. The initial phase involves a nucleophilic substitution where the imidazole nitrogen attacks the benzylic bromide of the tetrazole component. This step is facilitated by sodium hydroxide in a polar aprotic solvent like DMF, which solubilizes the ionic species effectively. Crucially, the reaction temperature is maintained between 40°C and 80°C, a range that is sufficiently energetic to drive the alkylation to completion without degrading the sensitive trityl-protected tetrazole group. Following this, the addition of further base and a moderate increase in temperature to 60-70°C triggers the saponification of the ethyl ester. This in-situ hydrolysis generates the sodium carboxylate salt, which remains soluble and reactive in the mixture, ready for the subsequent coupling.
The final esterification step is particularly noteworthy for its selectivity and mildness. By introducing cesium carbonate and the chloromethyl dioxolone reagent at lower temperatures (0-5°C initially, then warming), the process ensures that the carboxylate anion selectively attacks the chloromethyl group. The use of cesium carbonate is strategic; its large cation radius enhances the nucleophilicity of the carboxylate anion through weak ion pairing, thereby accelerating the reaction rate without requiring harsh activating agents. This mechanistic pathway avoids the generation of acidic by-products that could otherwise catalyze the decomposition of the acid-sensitive trityl group or the cyclic carbonate ring. Consequently, the impurity profile is markedly cleaner compared to acid-catalyzed esterification methods, leading to a final product that meets stringent pharmaceutical purity specifications with minimal downstream processing.
How to Synthesize Olmesartan Medoxomil Intermediate Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management to maximize the benefits of the telescoped design. The process is designed to be robust, tolerating minor variations in moisture content that would halt traditional anhydrous protocols. Operators should focus on the precise timing of reagent additions, particularly the transition from the alkylation phase to the hydrolysis phase, to ensure complete conversion before introducing the final electrophile. The detailed standardized operating procedures for this high-efficiency route are outlined below.
- Perform N-alkylation of the imidazole derivative with the biphenyl tetrazole bromide in DMF using sodium hydroxide at 40-80°C.
- Directly hydrolyze the ethyl ester intermediate in the same reaction vessel by adding additional sodium hydroxide and heating to 60-70°C.
- Cool the mixture and react the resulting carboxylate salt with 4-chloromethyl-5-methyl-1,3-dioxol-2-one using cesium carbonate to form the final cyclic carbonate ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis represents a significant opportunity to optimize the cost structure of olmesartan medoxomil production. By collapsing three distinct chemical steps into a single reactor train, manufacturers can drastically reduce the consumption of solvents, reagents, and energy. The elimination of intermediate isolation steps means that large volumes of solvent do not need to be evaporated and recovered between stages, leading to substantial utility savings and a smaller environmental footprint. Furthermore, the reduced processing time translates directly into increased reactor turnover rates, allowing facilities to produce higher volumes of API within the same timeframe without requiring capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the workflow. By removing the need for intermediate drying and purification, the labor hours required per kilogram of product are significantly decreased. Additionally, the avoidance of expensive and hazardous reagents like thionyl chloride reduces both raw material costs and the expenses associated with handling corrosive waste streams. The use of commodity chemicals such as sodium hydroxide and cesium carbonate ensures that the input costs remain stable and predictable, shielding the supply chain from volatility associated with specialty reagents.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this method enhances supply security. The reaction conditions are mild and do not require specialized anhydrous infrastructure, making the technology transferable to a wider range of manufacturing sites, including those in emerging markets. This flexibility allows for a more diversified supplier base, reducing the risk of disruption from single-source dependencies. The high yield and purity demonstrated in the patent data suggest that the process is reliable and reproducible, ensuring consistent delivery of quality material to downstream formulation partners.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the absence of exothermic hazards associated with acid chloride formations. The gentle thermal profile allows for safe operation in large-scale reactors, facilitating the commercial scale-up of complex antihypertensives. Moreover, the reduction in solvent usage and waste generation aligns with modern green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This compliance advantage minimizes the risk of regulatory delays and supports sustainable manufacturing initiatives that are increasingly valued by global pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's potential impact on your production capabilities.
Q: What are the primary advantages of the one-pot method over traditional multi-step synthesis?
A: The one-pot method eliminates the need for isolating and purifying unstable intermediates, significantly reducing solvent consumption, processing time, and potential product loss during transfer steps.
Q: Does this process require strict anhydrous conditions?
A: No, unlike prior art methods utilizing thionyl chloride which demand rigorous anhydrous environments, this novel approach operates under mild alkaline conditions that are more tolerant to moisture, simplifying equipment requirements.
Q: What purity levels can be achieved with this synthetic route?
A: Patent data indicates that the final olmesartan medoxomil produced via this route achieves HPLC purity levels exceeding 99.6%, demonstrating excellent impurity control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olmesartan Medoxomil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of the one-pot synthesis described in CN103435602A, guaranteeing stringent purity specifications and rigorous QC labs oversight for every batch produced.
We invite you to leverage our technical proficiency to optimize your olmesartan medoxomil supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing processes can deliver high-purity olmesartan medoxomil at a competitive market price, securing your position in the global cardiovascular therapy sector.
