Revolutionizing Cannabinoid Production: A High-Yield, Metal-Free Synthetic Route for Δ9-THC
Revolutionizing Cannabinoid Production: A High-Yield, Metal-Free Synthetic Route for Δ9-THC
The pharmaceutical and fine chemical industries have long grappled with the complexities of synthesizing bioactive cannabinoids, particularly (-)-Δ9-tetrahydrocannabinol (Δ9-THC). As detailed in Chinese Patent CN1266145C, a groundbreaking methodology has emerged that fundamentally alters the landscape of cannabinoid intermediate production. This patent discloses a novel process for preparing compound (A), specifically Δ9-THC and its analogs, by reacting a uniquely protected terpenoid derivative (Compound B) with a resorcinol derivative such as olivetol (Compound C). Unlike legacy methods that struggle with selectivity and purification, this approach leverages specific ether or ester protecting groups to modulate reactivity, enabling a streamlined one-step or two-step synthesis. For global procurement leaders and R&D directors, this represents a pivotal shift towards more robust, scalable, and compliant manufacturing protocols for high-value pharmaceutical intermediates.
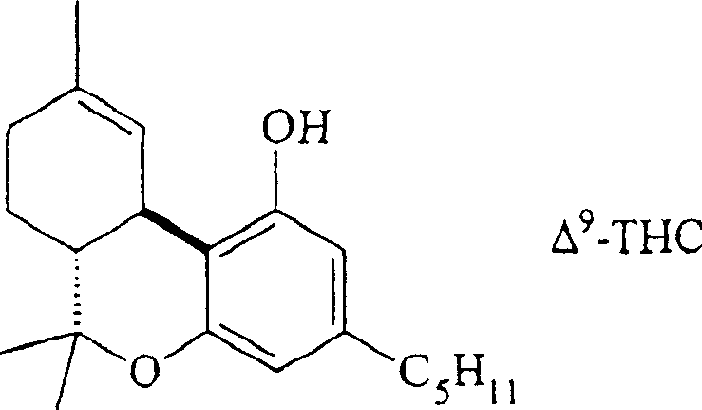
The significance of this technology lies in its ability to bypass the inherent instability of the target molecule. Δ9-THC is notoriously difficult to handle; it is a viscous liquid with a high boiling point that is prone to degradation. The patent highlights that traditional isolation or synthesis often leads to significant impurities due to the molecule's sensitivity to light, heat, and acidic conditions. By introducing a controlled coupling mechanism using protected precursors, the inventors have created a pathway that not only improves yield but also ensures the structural integrity of the final active pharmaceutical ingredient (API) precursor, addressing a critical pain point for reliable cannabinoid intermediate suppliers worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of Δ9-THC has been plagued by inefficiencies that render it suboptimal for large-scale commercial production. The most cited prior art, such as the Stoss method (US 5,227,537), relies on the acid-catalyzed coupling of (+)-p-mentha-2-ene-1,8-diol with olivetol. While conceptually straightforward, this route suffers from severe selectivity issues. The reaction rarely stops at the desired open-chain intermediate, instead producing a difficult-to-separate mixture of the intermediate and the cyclized product. Furthermore, the subsequent cyclization step typically employs heavy metal catalysts like zinc bromide (ZnBr2) under reflux conditions. This introduces a catastrophic risk for pharmaceutical applications: the potential for toxic heavy metal residues in the final product, necessitating expensive and time-consuming purification protocols to meet stringent regulatory standards.
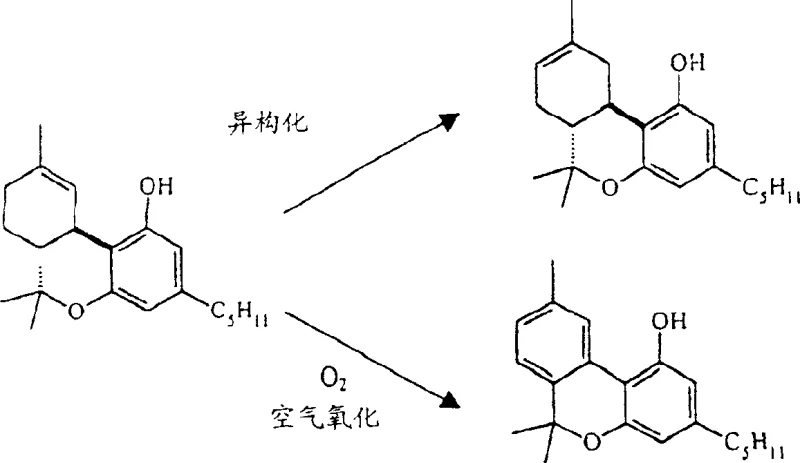
Compounding these issues is the thermodynamic instability of the product during synthesis. As illustrated in the degradation pathways, Δ9-THC easily undergoes acid-catalyzed isomerization to form the Δ8-isomer, which is thermodynamically more stable but pharmacologically distinct. Additionally, exposure to oxygen can oxidize the molecule into inactive cannabinol (CBN). Alternative methods, such as the Razdan route, attempt to mitigate these issues but fail on economic grounds; they require nearly 14 equivalents of zinc chloride and 6 equivalents of olivetol, yet still achieve a meager yield of only 28%. These factors combined create a bottleneck for cost reduction in pharmaceutical intermediates manufacturing, making traditional routes financially and technically unsustainable for modern supply chains.
The Novel Approach
The patented innovation introduces a paradigm shift by modifying the terpenoid starting material before the coupling event. Instead of using the free diol, the process utilizes Compound B, where the hydroxyl groups are masked as ethers or esters (e.g., acetates, benzoates, or diphenylacetates). This structural modification serves a dual purpose: it prevents premature cyclization during the initial Friedel-Crafts alkylation and modulates the electronic properties of the terpenoid to favor the formation of the desired stereochemistry. The reaction between this protected Compound B and olivetol (Compound C) can be conducted in a single pot or via a two-step sequence involving the isolation of an open-chain intermediate (Compound D). This flexibility allows manufacturers to optimize for either speed or purity, depending on their specific quality control requirements.
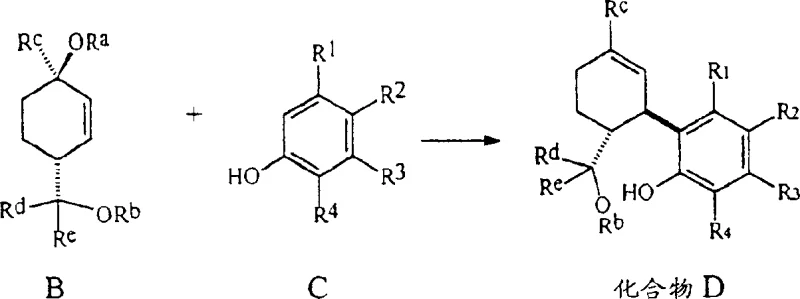
Crucially, this novel approach eliminates the reliance on stoichiometric heavy metal catalysts. The cyclization is promoted by non-metallic Lewis acids such as boron trifluoride etherate (BF3·(OEt)2) or p-toluenesulfonic acid (TsOH), used in catalytic amounts (0.1 to 1.5 equivalents). This change is transformative for the purity profile of the output. By removing zinc from the equation, the risk of metal contamination is eradicated, simplifying the downstream processing significantly. Moreover, the reaction conditions are milder, typically conducted at temperatures below 0°C (preferably -20°C to 0°C), which kinetically traps the desired Δ9-isomer and suppresses the formation of the unwanted Δ8-byproduct. This results in a cleaner crude reaction mixture, reducing the burden on purification systems and enhancing the overall viability of commercial scale-up of complex cannabinoids.
Mechanistic Insights into BF3-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the precise control of the Friedel-Crafts alkylation and subsequent intramolecular cyclization. When the protected terpenoid (Compound B) interacts with the electron-rich resorcinol ring of olivetol (Compound C) in the presence of a Lewis acid like BF3, the protecting group (ORa) acts as a leaving group. This generates a reactive carbocation species at the terpenoid backbone. The regioselectivity of this attack is critical; the specific steric bulk of the protecting groups (such as the diphenylacetate moiety) helps direct the olivetol to couple at the correct position on the terpenoid ring. This minimizes the formation of regioisomers that would otherwise complicate the purification process. The use of polar aprotic solvents like dichloromethane further stabilizes the ionic intermediates, ensuring a smooth progression towards the open-chain precursor (Compound D).
In the second stage, the cyclization mechanism is equally nuanced. The remaining protecting group (ORb) on the terpenoid side chain facilitates the ring closure to form the pyran ring characteristic of THC. The choice of acid catalyst is paramount here. BF3·(OEt)2 is preferred over protic acids like p-TsOH because it is less prone to catalyzing the double-bond migration that leads to Δ8-THC. The patent data indicates that by carefully controlling the stoichiometry of the acid and the temperature, the equilibrium can be shifted decisively towards the kinetic Δ9-product. This mechanistic understanding allows R&D teams to fine-tune reaction parameters, ensuring that the impurity profile remains within tight specifications, which is essential for producing high-purity OLED material or pharmaceutical grades where trace isomers are unacceptable.
How to Synthesize Δ9-THC Efficiently
Implementing this synthesis requires strict adherence to the patented conditions to maximize yield and minimize isomerization. The process generally begins with the preparation of the protected terpenoid, followed by the coupling reaction under inert atmosphere to prevent oxidation. The reaction temperature must be maintained strictly below freezing during the addition of the catalyst to control exothermicity and selectivity. For those seeking to implement this technology, the following guide outlines the critical operational phases derived from the patent examples, ensuring reproducibility and safety in a pilot or production environment.
- Preparation of Protected Terpenoid (Compound B): React (+)-p-mentha-2-ene-1,8-diol with acyl chlorides or anhydrides to form diesters or monoesters, ensuring the hydroxyl groups are masked to prevent premature cyclization.
- Coupling Reaction: Mix the protected terpenoid with olivetol (Compound C) in a polar aprotic solvent like dichloromethane at low temperatures (-20°C to 0°C) using a non-metallic Lewis acid catalyst such as BF3·(OEt)2.
- Cyclization and Purification: Depending on the protecting group, proceed via a one-pot direct cyclization or isolate the open-chain intermediate (Compound D) for purification before final ring closure to yield high-purity Δ9-THC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage is the drastic simplification of the purification workflow. Traditional methods relying on heavy metals require dedicated scavenging steps and rigorous testing to ensure compliance with ICH Q3D guidelines for elemental impurities. By adopting a metal-free catalytic system, manufacturers can eliminate these costly unit operations entirely. This reduction in processing steps translates directly into lower operational expenditures and a reduced carbon footprint, aligning with modern sustainability goals while simultaneously accelerating the time-to-market for finished cannabinoid products.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by improved atom economy and reagent efficiency. Unlike the Razdan method which consumes vast excesses of olivetol and zinc chloride, this novel route operates with near-stoichiometric ratios of reactants. The ability to isolate solid intermediates, such as the diphenylacetate derivatives described in the patent, allows for purification via crystallization rather than expensive preparative chromatography. Crystallization is inherently more scalable and cost-effective, significantly lowering the cost of goods sold (COGS) for high-volume production runs. Furthermore, the higher yields (exceeding 75% in preferred embodiments) mean less raw material waste, providing substantial cost savings in the procurement of specialized terpenoid starting materials.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes. This method enhances reliability by utilizing robust, commercially available catalysts like BF3·(OEt)2 and standard solvents like dichloromethane, which are less subject to supply shocks than specialized organometallic reagents. The tolerance of the process to various protecting groups (acetates, benzoates, etc.) provides flexibility; if one specific acyl chloride faces supply constraints, the chemistry can be adapted to use an alternative ester without redesigning the entire process. This adaptability ensures that production schedules remain uninterrupted, securing the supply of critical pharmaceutical intermediates even in volatile market conditions.
- Scalability and Environmental Compliance: Scaling viscous, unstable liquids like Δ9-THC is a notorious engineering challenge. This process mitigates those risks by generating solid or semi-solid intermediates that are easier to handle in large reactors. The elimination of zinc waste streams simplifies environmental compliance and wastewater treatment, reducing the regulatory burden on manufacturing sites. The patent explicitly notes the suitability of this method for industrial scale production, indicating that the reaction kinetics and heat transfer profiles are manageable in larger vessels. This scalability ensures that the technology can grow with demand, supporting the transition from clinical trial materials to full commercial manufacturing without the need for disruptive process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption. The following questions address common concerns regarding purity, scalability, and regulatory compliance, drawing directly from the experimental data and claims within patent CN1266145C. These insights are intended to clarify the operational advantages and assist in the decision-making process for integrating this technology into existing production pipelines.
Q: How does this new synthesis method improve upon the traditional Stoss and Razdan routes?
A: Unlike the Stoss method which uses heavy metal catalysts like ZnBr2 leading to potential toxic residues, and the Razdan method which requires excessive reagents and yields only 28%, this patented process utilizes protected terpenoid esters and non-metallic Lewis acids (e.g., BF3). This results in significantly higher selectivity, yields exceeding 75%, and eliminates the need for complex heavy metal removal steps.
Q: What are the critical stability challenges in Δ9-THC production addressed by this patent?
A: Δ9-THC is highly susceptible to acid-catalyzed isomerization into the thermodynamically stable but less active Δ8-isomer, and oxidation into inactive cannabinol (CBN). This process mitigates these risks by controlling reaction temperatures below 0°C and using specific protecting groups that modulate reactivity, thereby minimizing side reactions and simplifying the purification of the viscous final product.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly designs the method for industrial scalability. By avoiding chromatography-dependent purifications where possible (through crystallization of intermediates) and using readily available non-metallic catalysts, the process reduces operational complexity. The ability to isolate solid intermediates (like diphenylacetate derivatives) further enhances the feasibility of commercial scale-up compared to handling viscous oils in traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Δ9-THC Supplier
The technological advancements detailed in patent CN1266145C represent a significant leap forward in cannabinoid chemistry, yet realizing their full potential requires a manufacturing partner with deep technical expertise and robust infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of detecting trace isomers and impurities, ensuring that every batch meets stringent purity specifications required by global regulatory bodies. We understand that the difference between a viable commercial process and a laboratory curiosity lies in the details of execution, and our team is dedicated to mastering these complexities.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate the development of next-generation cannabinoid therapeutics, ensuring a reliable supply of high-quality intermediates that drive innovation in the healthcare sector.
