Advanced Regioselective Synthesis of CCI-779 Using Boronic Acid Intermediates for Commercial Scale
Advanced Regioselective Synthesis of CCI-779 Using Boronic Acid Intermediates for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for complex oncology agents like CCI-779 (Temsirolimus), an ester of rapamycin with potent mTOR inhibitory activity. Patent CN1829722A introduces a transformative methodology leveraging boronic acid chemistry to achieve high regioselectivity at the 42-position of the rapamycin macrocycle. This innovation addresses critical bottlenecks in traditional manufacturing, specifically the reliance on tedious chromatographic purifications that hinder cost-effective commercialization. By utilizing a novel boronate intermediate, this process enables the production of high-purity CCI-779 through crystallization rather than column chromatography. For global procurement teams and R&D directors, this represents a significant opportunity to optimize the supply chain for this vital anticancer intermediate. The following analysis dissects the technical merits and commercial implications of this boronate-mediated pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of rapamycin 42-esters has been plagued by poor regioselectivity due to the presence of multiple hydroxyl groups on the macrocyclic scaffold. Conventional non-regioselective acylation methods typically yield complex mixtures containing the desired 42-ester alongside unwanted 31-esters, 31,42-di-esters, and unreacted starting material. Separating these structurally similar analogs traditionally necessitates extensive column chromatography, a technique that is notoriously difficult to scale for industrial production. The heavy solvent consumption associated with chromatography not only drives up raw material costs but also creates significant environmental burdens regarding waste disposal. Furthermore, the low throughput of chromatographic processes limits overall production capacity, creating supply chain vulnerabilities for high-demand oncology drugs. These inefficiencies make conventional routes economically unsustainable for large-scale API intermediate manufacturing.
The Novel Approach
The methodology disclosed in CN1829722A circumvents these challenges by employing a specialized boronic acid derivative, specifically 5-methyl-2-phenyl-1,3,2-dioxaborinane-5-carboxylic acid (Compound [A]). This reagent acts as a protecting and activating group that directs acylation exclusively to the 42-hydroxyl position of 31-O-silylated rapamycin. The resulting boronate ester intermediate possesses distinct physicochemical properties that allow it to be purified via simple crystallization from solvents like acetone. This shift from chromatography to crystallization is a paradigm change in process chemistry, drastically reducing the number of unit operations required. By eliminating the need for silica gel columns and vast volumes of elution solvents, the new approach streamlines the workflow, enhances safety profiles, and significantly improves the overall mass balance of the synthesis. This makes it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming for lean manufacturing.
Mechanistic Insights into Boronate-Mediated Regioselective Acylation
The core of this synthetic strategy lies in the unique reactivity of the boronic acid mixed anhydride. The process begins with the selective silylation of rapamycin at the 31-position using trimethylsilyl chloride, leaving the 42-hydroxyl group free for reaction. Subsequently, Compound [A] is activated in situ using 2,4,6-trichlorobenzoyl chloride to form a highly reactive mixed anhydride species. This activation step is critical and is typically monitored via in-situ IR spectroscopy to ensure complete conversion before the addition of the rapamycin derivative. The steric bulk and electronic nature of the boronate ring system facilitate nucleophilic attack specifically at the 42-position, minimizing side reactions at other sites. The reaction is conducted at low temperatures, typically between -11°C and -5°C, to maintain the stability of the mixed anhydride and control the exotherm. This precise temperature control ensures that the kinetic product is favored, leading to high regioselectivity without the formation of di-ester impurities that plague other methods.
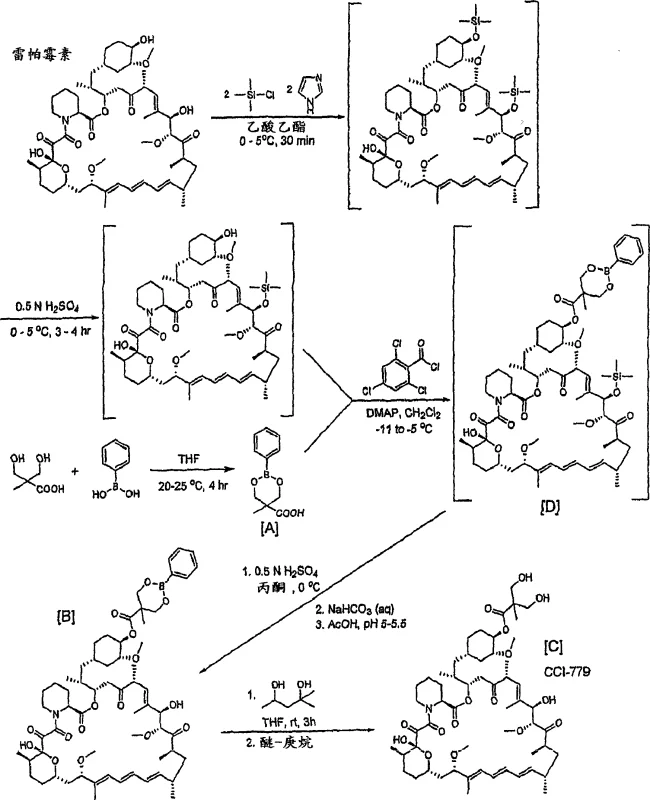
Following the acylation, the crude reaction mixture undergoes a hydrolysis step under mild acidic conditions to remove the 31-silyl protecting group. A crucial aspect of this mechanism is the behavior of the resulting boronate intermediate [B], which exists as B and C isomers. The patent highlights that the B-isomer crystallizes preferentially, especially when the pH is adjusted to 5-6 using acetate buffers. This isomer enrichment during crystallization serves as an additional purification layer, effectively excluding impurities and the less soluble C-isomer from the final solid. The final step involves a trans-borylation reaction where the phenylboronate moiety is exchanged for a diol, such as 2-methyl-2,4-pentanediol, to release the free CCI-779. This cleavage is efficient and proceeds with high yields, typically ranging from 80% to 90% in the final step, demonstrating the robustness of the boronate linkage as a temporary masking group for the carboxylic acid side chain.
How to Synthesize CCI-779 Efficiently
Implementing this synthesis requires strict adherence to the patented sequence of silylation, activation, coupling, and deprotection to ensure optimal yield and purity. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates while maximizing the efficiency of the crystallization steps. Detailed standard operating procedures for each stage, including specific solvent swaps and temperature ramps, are essential for reproducibility at scale. For technical teams looking to adopt this route, understanding the nuances of the mixed anhydride formation and the pH-controlled crystallization of intermediate [B] is paramount.
- Protect Rapamycin at the 31-position using trimethylsilyl chloride to form 31-O-TMS rapamycin.
- Activate 5-methyl-2-phenyl-1,3,2-dioxaborinane-5-carboxylic acid [A] into a mixed anhydride using 2,4,6-trichlorobenzoyl chloride.
- Couple the mixed anhydride with 31-O-TMS rapamycin at -11 to -5°C to form the 42-ester boronate intermediate [D].
- Hydrolyze the silyl group under mild acidic conditions to precipitate intermediate [B], then perform trans-borylation with a diol to yield CCI-779.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this boronate-based route offers profound advantages for cost reduction in pharmaceutical intermediates manufacturing. The most significant impact comes from the elimination of chromatographic purification, which is often the most expensive and time-consuming step in complex molecule synthesis. By replacing columns with crystallization vessels, manufacturers can drastically reduce solvent procurement costs and waste treatment fees. The patent explicitly notes that production reaction time and resources can be reduced by 50%, a figure that translates directly into lower operational expenditures and higher asset utilization rates. For procurement managers, this means a more stable pricing structure for CCI-779, as the process is less susceptible to fluctuations in silica gel or specialized solvent availability. The ability to produce high-purity material without chromatography also simplifies the regulatory filing process, as the impurity profile is cleaner and more consistent.
- Cost Reduction in Manufacturing: The removal of chromatography eliminates the need for large quantities of high-grade organic solvents and silica gel, which are major cost drivers in fine chemical production. Additionally, the reduced processing time allows for more batches to be run per year in the same facility, effectively lowering the fixed cost per kilogram of product. The streamlined workflow also reduces labor hours associated with column packing, running, and solvent recovery, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Crystallization-based processes are inherently more scalable and robust than chromatographic ones, reducing the risk of batch failures due to column channeling or overloading. The stability of the key boronate intermediate [B] allows for strategic stockpiling, decoupling the upstream synthesis from the final deprotection step. This flexibility ensures a continuous supply of CCI-779 even if there are disruptions in the supply of specific reagents, thereby enhancing the resilience of the global supply chain for this critical oncology ingredient.
- Scalability and Environmental Compliance: Scaling a chromatographic process from grams to tons is technically challenging and often requires disproportionate increases in equipment footprint. In contrast, crystallization scales linearly and fits easily into standard stainless steel reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates. Furthermore, the significant reduction in solvent waste aligns with modern green chemistry principles and environmental regulations, reducing the carbon footprint of the manufacturing process and simplifying compliance with increasingly strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boronate-mediated synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios.
Q: How does the boronic acid method improve purity compared to traditional acylation?
A: Traditional acylation produces a mixture of 31-ester, 42-ester, and di-esters requiring chromatography. The boronic acid method utilizes steric and electronic properties to achieve high regioselectivity at the 42-position, allowing purification via crystallization instead of column chromatography.
Q: What are the scalability benefits of eliminating chromatography in CCI-779 production?
A: Removing chromatographic steps significantly reduces solvent consumption, processing time, and reactor volume requirements. This transition to crystallization-based purification enables easier commercial scale-up from kilograms to metric tons while lowering operational costs.
Q: Is the boronate intermediate stable for long-term storage?
A: Yes, the key intermediate [B] (Rapamycin 42-ester boronate) is described as a stable solid white powder at room temperature, which facilitates inventory management and flexible production scheduling for supply chain optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CCI-779 Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic routes like the boronate-mediated synthesis of CCI-779 in securing a competitive edge in the oncology market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of CCI-779 meets the highest international standards. Our state-of-the-art facilities are equipped to handle the specific temperature and handling requirements of boronic acid chemistry, ensuring safe and efficient manufacturing.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing strategy available in the market.
