Advanced One-Pot Synthesis of Stable Platelet Aggregation Inhibitor Intermediates for Commercial Scale-Up
Advanced One-Pot Synthesis of Stable Platelet Aggregation Inhibitor Intermediates for Commercial Scale-Up
The pharmaceutical landscape for cardiovascular therapeutics is constantly evolving, driven by the need for more stable and bioavailable active ingredients. A pivotal development in this sector is detailed in patent CN101781311A, which discloses a novel preparation method for a class of platelet aggregation inhibition compounds designated as Formula I. This technology represents a significant leap forward from traditional synthetic routes, addressing the inherent instability and processing difficulties associated with earlier generations of ADP receptor inhibitors like Clopidogrel. By shifting from an oily, moisture-sensitive free base to a stable solid intermediate, this innovation not only enhances the physicochemical profile of the drug candidate but also streamlines the manufacturing process through a sophisticated one-pot telescoped synthesis. For R&D directors and procurement specialists alike, understanding the nuances of this pathway is crucial for securing a reliable supply of high-purity pharmaceutical intermediates that meet rigorous global standards.
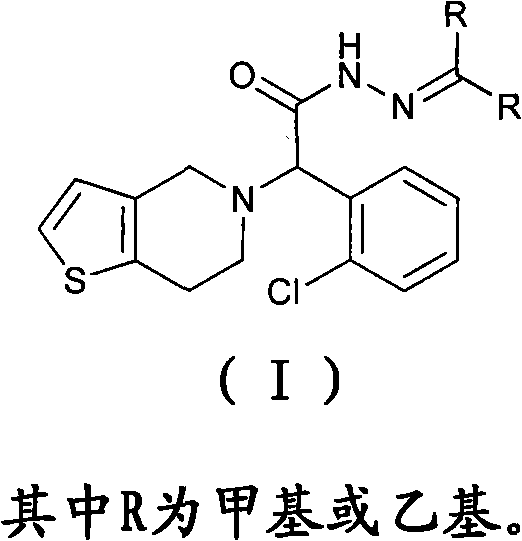
The core of this technological breakthrough lies in the structural transformation of the thienopyridine scaffold. As illustrated in the general formula, the compound features a chiral center adjacent to a chlorophenyl group and a tetrahydrothienopyridine moiety, capped with a hydrazone functionality. This specific architecture confers superior stability compared to its predecessors, eliminating the need for complex salt formation with strong acids which often complicates downstream formulation. The ability to produce this molecule as a free base solid simplifies the entire value chain, from initial synthesis to final tablet compression, offering a compelling value proposition for manufacturers seeking to optimize their cardiovascular drug portfolios with robust and scalable chemical entities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar antiplatelet agents has been plagued by inefficiencies that drive up costs and extend lead times. Traditional literature methods, such as those referenced in prior art patents, typically involve a multi-step sequence where the intermediate hydrazide must be isolated, purified, and dried before proceeding to the final condensation step. This discontinuous approach results in significant material loss; for instance, legacy processes often report yields for the initial hydrazide formation step as low as 61.6%, creating a bottleneck that severely impacts overall throughput. Furthermore, the isolation of intermediates necessitates large volumes of solvents for extraction and washing, generating substantial chemical waste and increasing the environmental footprint of the manufacturing process. These operational complexities not only inflate the cost of goods sold but also introduce multiple points of failure where impurities can be introduced or yield can be compromised, posing a risk to supply chain continuity.
The Novel Approach
In stark contrast, the methodology outlined in CN101781311A employs a clever one-pot strategy that telescopes multiple reaction stages into a single vessel without intermediate purification. This approach allows the reaction mixture to proceed directly from the activated acid species to the final hydrazone product, bypassing the tedious isolation of the hydrazide intermediate entirely. By integrating the activation, nucleophilic substitution, and condensation reactions, the process achieves a total recovery rate exceeding 77%, representing a dramatic improvement in atom economy and resource utilization. The elimination of isolation steps drastically reduces solvent consumption and operational time, aligning perfectly with the principles of green chemistry. This streamlined workflow not only enhances the economic viability of the production but also ensures a more consistent quality profile by minimizing human intervention and exposure of sensitive intermediates to potentially degrading conditions.
Mechanistic Insights into Mixed Anhydride Activation and Condensation
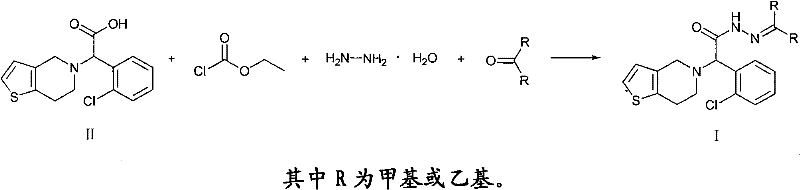
The success of this one-pot synthesis hinges on the precise generation and utilization of a mixed anhydride intermediate. The process initiates by dissolving the carboxylic acid precursor (Compound II) in a suitable solvent such as dioxane and cooling the mixture to a controlled low temperature range of 7-12°C. At this stage, an acid binding agent, which can be selected from a group including sodium hydroxide, potassium carbonate, or sodium bicarbonate, is introduced to neutralize the system. Subsequently, vinyl chloroformate is added dropwise, reacting with the carboxylic acid to form a highly reactive mixed anhydride species. This activation step is critical; the low temperature prevents thermal decomposition and suppresses side reactions that could lead to racemization or the formation of symmetric anhydrides. The mixed anhydride serves as a superior electrophile, primed for nucleophilic attack by hydrazine hydrate in the subsequent phase, ensuring high conversion efficiency without the need for harsh coupling reagents.
Following the formation of the mixed anhydride, the reaction temperature is carefully allowed to rise to between 20-30°C, at which point hydrazine hydrate and the corresponding ketone (acetone for the methyl derivative or 3-pentanone for the ethyl derivative) are introduced. The hydrazine attacks the activated carbonyl carbon, displacing the leaving group to form the acethydrazide in situ. Without isolation, this newly formed hydrazide immediately undergoes a condensation reaction with the ketone present in the same pot, facilitated by the slightly elevated temperature and the basic environment. This tandem sequence ensures that the equilibrium is driven towards the formation of the stable hydrazone product (Formula I). The result is a crude product with exceptional purity, often exceeding 99% as determined by HPLC analysis, which can be further purified by simple recrystallization or washing, demonstrating the robustness of this mechanistic design in controlling impurity profiles.
How to Synthesize (S)-α-[2-chlorophenyl-2-(4,5,6,7-tetrahydrothieno[3,2-c]pyridin-5-yl)]-N'-[(dimethyl)methylene]acethydrazide Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management to replicate the high yields reported in the patent data. The molar ratio of the starting acid, vinyl chloroformate, acid binding agent, hydrazine hydrate, and ketone is optimized to approximately 1:(1-1.3):(1-3):(0.9-1.1):(0.9-1.1), ensuring that the activation is complete while minimizing excess reagents that could complicate workup. The detailed standardized synthetic steps below outline the precise addition rates and temperature ramps necessary to achieve the target specifications consistently. Operators must ensure that the initial cooling phase is strictly maintained to prevent exothermic runaway during the activation step, which is vital for preserving the stereochemical integrity of the chiral center.
- Dissolve the carboxylic acid precursor (Compound II) in dioxane and cool to 7-12°C, then add an acid binding agent and vinyl chloroformate to form the mixed anhydride.
- Without isolation, introduce hydrazine hydrate and the corresponding ketone (acetone or 3-pentanone) to the reaction mixture while warming to 20-30°C.
- Maintain the reaction until completion, then cool to precipitate the solid product, followed by filtration and washing to achieve high purity (>99%).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis methodology translates into tangible strategic advantages that go beyond simple yield metrics. The consolidation of multiple reaction steps into a single operational unit significantly reduces the demand for reactor volume and turnaround time, allowing manufacturing facilities to increase their batch frequency without capital expenditure on new equipment. This intensification of the process directly addresses the industry-wide challenge of scaling up complex pharmaceutical intermediates, offering a pathway to meet growing global demand for cardiovascular therapies with greater agility. Furthermore, the reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, mitigating the risk of compliance-related shutdowns and reducing the overhead costs associated with waste disposal and treatment.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation and purification steps fundamentally alters the cost structure of production. By avoiding the need to dry and store the hydrazide intermediate, manufacturers save significantly on energy costs associated with drying ovens and vacuum pumps, as well as the labor hours required for handling solids. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final product, effectively lowering the cost per kilogram. The simplified workflow also reduces the consumption of auxiliary materials such as filtration media and washing solvents, contributing to a leaner and more cost-effective manufacturing operation that enhances margin potential.
- Enhanced Supply Chain Reliability: A shorter and simpler production cycle inherently reduces the lead time required to fulfill orders, providing a buffer against market volatility and raw material shortages. The robustness of the one-pot method minimizes the risk of batch failures due to handling errors during intermediate transfers, ensuring a more predictable and consistent output. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages. Moreover, the use of readily available and inexpensive reagents like vinyl chloroformate and acetone ensures that the supply chain is not dependent on exotic or scarce catalysts, further stabilizing the sourcing strategy and protecting against price fluctuations in the raw material market.
- Scalability and Environmental Compliance: The green synthesis nature of this process, characterized by high atom economy and reduced solvent intensity, makes it highly scalable from pilot plant to commercial production. The ability to run the reaction in a single vessel simplifies the engineering controls required for scale-up, reducing the complexity of validation and technology transfer. From an environmental perspective, the drastic reduction in chemical waste supports corporate sustainability goals and facilitates easier permitting in regions with strict environmental oversight. This compliance advantage future-proofs the supply chain against tightening regulations, ensuring long-term operational viability and enhancing the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this platelet aggregation inhibitor intermediate. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing drug development pipelines or supply networks.
Q: What is the primary advantage of the one-pot method described in CN101781311A?
A: The primary advantage is the elimination of intermediate isolation and purification steps. By telescoping the activation, hydrazide formation, and condensation into a single vessel, the process significantly reduces solvent consumption, operational time, and overall production costs while increasing total yield to over 77%.
Q: How does this intermediate improve upon traditional Clopidogrel derivatives?
A: Unlike Clopidogrel free base, which is an unstable oily substance requiring salt formation with strong acids, this compound (Formula I) exists as a stable solid. This physical state offers superior physicochemical properties, making it easier to purify, handle, and formulate into final dosage forms without the stability issues associated with moisture sensitivity.
Q: What are the critical reaction conditions for maintaining high purity?
A: Critical conditions include maintaining a low temperature (7-12°C) during the initial activation with vinyl chloroformate to prevent side reactions, followed by a controlled warm-up to 20-30°C for the condensation step. The use of appropriate acid binding agents like sodium hydroxide or potassium carbonate is also essential to neutralize byproducts effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Platelet Aggregation Inhibitor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation cardiovascular therapeutics. Our team of expert chemists has extensively analyzed the one-pot methodology described in CN101781311A and possesses the technical capability to implement this process at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthesis technology for your projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency in your supply chain.
