Scalable Liquid Phase Synthesis of Taltirelin Intermediates for Commercial Production
Introduction to Advanced Taltirelin Synthesis Technology
The pharmaceutical landscape for neurological agents continues to evolve, driven by the demand for more efficient and scalable synthetic routes for complex peptides like Taltirelin. Patent CN102002004A introduces a groundbreaking liquid phase synchronization method that addresses the longstanding bottlenecks in producing 3-methyl hydroorotic acid and L-histidine-L-proline amide. Unlike traditional approaches that often rely on cumbersome solid-phase techniques or hazardous azide chemistry, this innovation leverages a streamlined liquid phase process that significantly enhances operational simplicity. The core breakthrough lies in the strategic selection of protecting groups and coupling reagents, which allows for mild reaction conditions that are inherently safer and more conducive to industrial scaling. By optimizing the synthesis of the key 3-methyl hydroorotic acid moiety, the patent provides a robust foundation for manufacturing high-purity Taltirelin intermediates. This technological leap is particularly relevant for supply chain leaders seeking reliable sources of neuroactive peptide precursors that can be produced consistently without the volatility associated with older, more complex methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Taltirelin and its critical intermediates has been plagued by inefficient purification steps and hazardous reaction conditions that hinder commercial viability. Prior art methods frequently employed Z-His-OH and H-Pro-OH condensation followed by ammonification, necessitating the use of the dangerous azide method to prevent side-chain reactions due to the lack of protection on the histidine imidazole ring. This approach not only introduced significant safety risks regarding explosive intermediates but also resulted in a reaction process that was notoriously difficult to control and monitor. Furthermore, the purification of the crude product traditionally required the use of expensive Mitsubishi CHP-20P gel chromatography, a technique that is cost-prohibitive for large-scale manufacturing and creates substantial waste streams. The reliance on vacuum concentration to remove solvents like DMF in earlier iterations was also time-consuming and often led to incomplete solvent removal, compromising the quality of the final active pharmaceutical ingredient. These cumulative inefficiencies created a high barrier to entry for manufacturers, limiting the availability of cost-effective Taltirelin for the global market.
The Novel Approach
The novel approach detailed in the patent fundamentally restructures the synthetic pathway to eliminate these historical pain points through intelligent molecular design and process engineering. By utilizing Boc-His(Trt)-OH and H-Pro-NH2 as starting materials, the new method enables direct condensation using standard coupling agents like DCC or EDC, completely bypassing the need for hazardous azide chemistry. This shift not only enhances safety but also simplifies the reaction system, making it far more amenable to scale-up operations. A critical innovation is the workup procedure for the hydroorotic acid derivatives, where the reaction solution is poured directly into frozen water to precipitate the product, replacing the energy-intensive vacuum concentration of DMF. This simple physical change dramatically improves production efficiency and pushes yields from approximately 61% in prior art to over 80%. Additionally, the final purification of Taltirelin is achieved through simple pH adjustment and filtration rather than expensive column chromatography, representing a massive reduction in downstream processing costs. 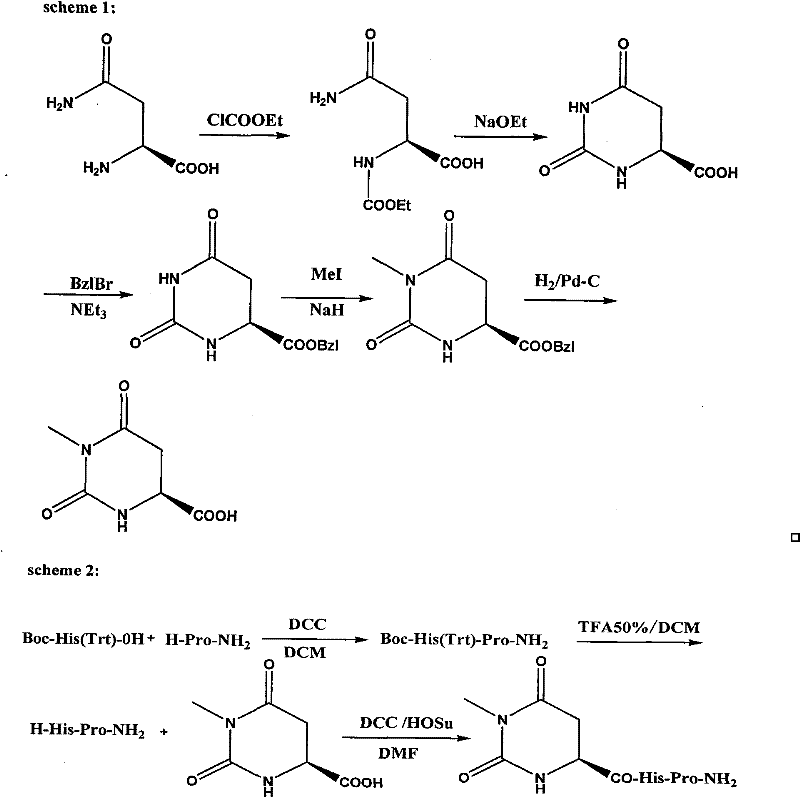
Mechanistic Insights into Liquid Phase Cyclization and Coupling
The mechanistic elegance of this synthesis lies in the precise control of reactivity during the formation of the dihydropyrimidine ring and the subsequent peptide coupling. The process begins with the reaction of L-Asparagine with ethyl chloroformate to form an N-alpha-ethoxycarbonyl intermediate, which serves as a activated species for the subsequent intramolecular cyclization. Under the influence of sodium ethoxide, this intermediate undergoes a nucleophilic attack that closes the ring to form the hydroorotic acid scaffold, a crucial structural motif for the biological activity of the final drug. The introduction of the 3-methyl group is achieved through a controlled alkylation using methyl iodide and sodium hydride, where the base deprotonates the nitrogen to facilitate nucleophilic substitution. This step is critical for mimicking the metabolic stability of the natural TRH hormone while enhancing CNS excitation. The final assembly involves the coupling of the 3-methyl hydroorotic acid with the dipeptide amide L-His-L-Pro-NH2. The use of HOSu (N-hydroxysuccinimide) alongside DCC forms an active ester in situ, which reacts efficiently with the amine component to form the final peptide bond with high fidelity. 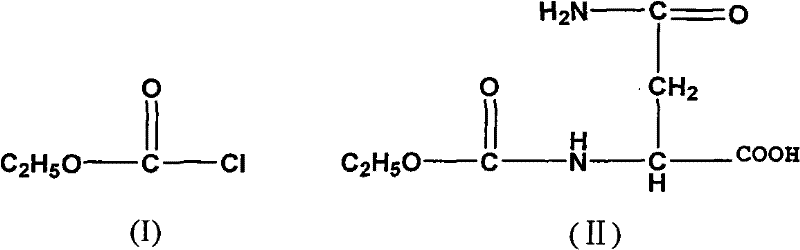
Impurity control is meticulously managed through the selection of orthogonal protecting groups that prevent unwanted side reactions during the synthesis. The Trityl (Trt) group on the histidine imidazole ring effectively blocks alkylation at the nitrogen position, ensuring that the methylation occurs exclusively at the desired position on the hydroorotic acid ring. Furthermore, the Boc group on the histidine amino terminus prevents self-polymerization during the initial dipeptide formation, ensuring that the coupling with the acid component proceeds cleanly. The workup strategy of pouring the reaction mixture into frozen water serves a dual purpose: it quenches any remaining reactive species like sodium hydride safely and induces rapid crystallization of the product, leaving soluble impurities in the aqueous phase. This physical separation mechanism is far superior to chemical scavenging methods, as it avoids introducing additional reagents that could become new impurities. The final pH adjustment to 8.0 using saturated sodium bicarbonate ensures that the zwitterionic nature of the peptide is optimized for precipitation, further enhancing the purity profile without the need for chromatographic intervention.
How to Synthesize 3-Methyl Hydroorotic Acid Efficiently
The synthesis of 3-methyl hydroorotic acid serves as the cornerstone of this entire production route, requiring precise attention to stoichiometry and temperature control to maximize yield. The process initiates with the activation of L-Asparagine, followed by a base-mediated cyclization that constructs the heterocyclic core essential for the drug's function. Subsequent protection as a benzyl ester allows for the selective methylation at the 3-position, a step that defines the pharmacological potency of the final Taltirelin molecule. The detailed standardized synthetic steps below outline the exact conditions, reagent ratios, and workup procedures necessary to replicate this high-efficiency pathway in a GMP environment. Operators should note the critical importance of the frozen water quenching step, as this specific technique is the key differentiator that drives the yield improvement from legacy methods.
- React L-Asparagine with ethyl chloroformate to form N-alpha-ethoxycarbonyl-L-asparagine, followed by cyclization with sodium ethoxide to obtain the hydroorotic acid derivative.
- Protect the hydroorotic acid as a benzyl ester, then perform methylation using methyl iodide and sodium hydride to introduce the 3-methyl group.
- Remove the benzyl protecting group via hydrogenation to yield 3-methyl hydroorotic acid, then couple with L-Histidine-L-Proline amide to finalize Taltirelin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative advantages in terms of cost structure and logistical reliability. The elimination of expensive chromatographic purification media, such as the Mitsubishi gel used in prior art, translates directly into a significant reduction in variable manufacturing costs per kilogram. By replacing complex vacuum distillation steps with simple precipitation and filtration, the process drastically reduces energy consumption and equipment occupancy time, allowing for faster batch turnover. The use of readily available starting materials like L-Asparagine and common reagents like benzyl bromide ensures that the supply chain is not vulnerable to the shortages often associated with exotic or highly specialized catalysts. Furthermore, the mild reaction conditions reduce the requirement for specialized high-pressure or cryogenic equipment, lowering the capital expenditure barrier for contract manufacturing organizations looking to onboard this product. These factors combine to create a supply profile that is both economically attractive and resilient against market fluctuations.
- Cost Reduction in Manufacturing: The strategic redesign of the workup procedure eliminates the need for costly vacuum concentration of high-boiling solvents like DMF, which is both energy-intensive and time-consuming. By simply pouring the reaction mixture into frozen water, the process achieves product isolation through precipitation, saving substantial manpower and equipment costs while simultaneously improving yield. The removal of the expensive gel chromatography step for final purification represents a massive saving in consumable costs, as silica-based or polymer-based columns are significant line items in peptide manufacturing budgets. Additionally, the use of standard coupling reagents like DCC instead of specialized azide precursors reduces the raw material cost burden, making the overall cost of goods sold much more competitive.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as L-Asparagine, ethyl chloroformate, and methyl iodide ensures that the raw material supply chain is robust and less prone to disruption compared to routes requiring custom-synthesized intermediates. The simplified operational workflow, characterized by ambient temperature reactions and straightforward filtration steps, minimizes the risk of batch failures due to equipment malfunction or operator error. This operational simplicity facilitates easier technology transfer between different manufacturing sites, ensuring continuity of supply even if one facility faces downtime. The ability to produce high-purity intermediates without complex purification trains means that lead times can be significantly shortened, allowing for more responsive inventory management and just-in-time delivery models.
- Scalability and Environmental Compliance: The liquid phase nature of the synthesis is inherently scalable, allowing for seamless transition from pilot plant batches to multi-ton commercial production without the need for re-optimizing reaction parameters. The avoidance of hazardous azide chemistry significantly improves the safety profile of the plant, reducing insurance premiums and regulatory compliance burdens associated with handling explosive intermediates. The simplified waste stream, primarily consisting of aqueous salts and organic solvents that can be recovered, is easier to treat and dispose of in accordance with environmental regulations. This green chemistry approach not only aligns with corporate sustainability goals but also future-proofs the manufacturing process against tightening environmental legislation regarding solvent emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for Taltirelin intermediates. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of adopting this route for their own production pipelines.
Q: How does this liquid phase method improve upon traditional Taltirelin synthesis?
A: This method replaces complex azide coupling and expensive chromatographic purification with simple DCC-mediated coupling and pH-adjusted crystallization, drastically reducing operational complexity and equipment costs.
Q: What are the key purity advantages of the new workup procedure?
A: By pouring reaction mixtures into frozen water instead of vacuum concentrating DMF, the process achieves higher yields (over 80%) and eliminates difficult-to-remove solvent residues, ensuring high-purity intermediates.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route utilizes readily available raw materials like L-Asparagine and employs mild reaction conditions with simple filtration steps, making it highly adaptable for metric-ton scale production without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taltirelin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who can translate complex patent methodologies into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this liquid phase synthesis are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle the analytical challenges of peptide intermediates, guaranteeing that every batch meets the highest international standards. Our commitment to quality assurance means that we can consistently deliver 3-methyl hydroorotic acid and related Taltirelin precursors with the impurity profiles required for regulatory submission.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of these high-quality intermediates into your pharmaceutical development pipeline.
