Scalable Production of High-Purity Taltirelin via Novel DKP-Free Route
Scalable Production of High-Purity Taltirelin via Novel DKP-Free Route
The pharmaceutical landscape for treating spinocerebellar degeneration has long relied on Thyrotropin-Releasing Hormone (TRH) analogs, with Taltirelin standing out due to its superior stability and endocrine profile. However, the commercial viability of this critical active pharmaceutical ingredient (API) has historically been hampered by complex synthesis routes plagued by impurity formation. A groundbreaking patent, CN109369779B, filed in late 2021, introduces a transformative synthetic methodology that fundamentally alters the production economics of this molecule. By shifting the synthetic strategy to utilize 2-cyanopyrrolidine as a key starting material, this innovation effectively bypasses the notorious Diketopiperazine (DKP) formation pathway that has burdened manufacturers for decades. This technical breakthrough not only enhances the chemical purity of the final product but also streamlines the downstream processing requirements, offering a compelling value proposition for global supply chains seeking reliable sources of high-quality neuroactive intermediates.
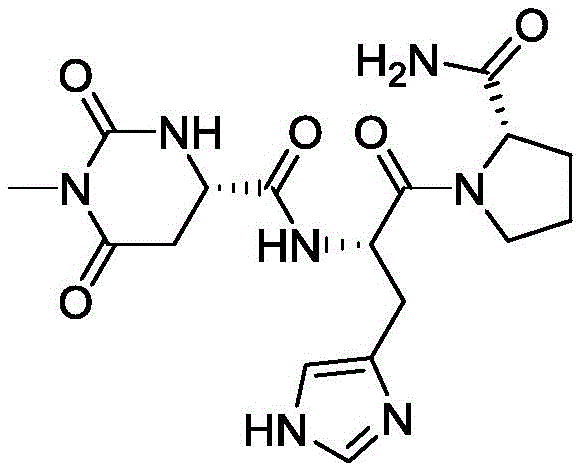
The structural integrity of Taltirelin is paramount for its biological efficacy, and the new synthesis route described in the patent ensures that the delicate peptide bonds remain intact while minimizing side reactions. Traditional methods often struggle with the cyclization of the histidine-proline motif, leading to difficult-to-remove impurities that require extensive chromatographic purification. In contrast, the approach detailed in CN109369779B leverages a linear assembly strategy that maintains control over stereochemistry and functional group compatibility throughout the sequence. For R&D directors and process chemists, this represents a significant opportunity to reduce the impurity burden early in the synthesis, thereby lowering the overall cost of goods sold (COGS) and improving the robustness of the manufacturing process. The ability to produce Taltirelin with consistently high purity levels directly impacts the regulatory filing success and market readiness of generic or novel formulations targeting neurodegenerative disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Taltirelin has been dominated by routes that involve the condensation of methyl orotic acid with a protected L-histidine-L-proline dipeptide fragment. While conceptually straightforward, this classical approach suffers from a critical thermodynamic flaw: the propensity of the histidine-proline junction to undergo intramolecular cyclization, forming a Diketopiperazine (DKP) byproduct. This DKP impurity is structurally similar to the target molecule, making its removal via standard crystallization or extraction techniques exceptionally difficult and often necessitating expensive preparative HPLC purification. Furthermore, legacy patents such as CN85105655A and CN103588862A rely on harsh deprotection conditions, utilizing strong acids like hydrobromic acid (HBr) or toxic reagents like methyl chloroformate and methyl iodide. These aggressive chemical environments not only pose significant safety risks to operators and environmental compliance challenges but also degrade the sensitive peptide backbone, leading to reduced overall yields and inconsistent batch-to-batch quality. The reliance on such hazardous materials creates a bottleneck for scale-up, as specialized corrosion-resistant equipment and rigorous waste treatment protocols become mandatory, driving up capital expenditure and operational costs.
The Novel Approach
The innovative methodology disclosed in patent CN109369779B circumvents these historical pitfalls by reversing the logical assembly of the peptide chain and introducing a nitrile-functionalized pyrrolidine precursor. Instead of starting with a pre-formed proline derivative prone to cyclization, the process initiates with the condensation of 2-cyanopyrrolidine with protected histidine. This strategic modification effectively blocks the nucleophilic attack of the amine on the carbonyl carbon that leads to DKP formation, as the nitrile group acts as a stable placeholder that is only converted to the final amide functionality in a controlled oxidation step later in the sequence. The subsequent coupling with 1-methyl-4,5-dihydroorotic acid is performed under mild conditions using standard peptide coupling reagents like DCC and HOBt, ensuring high fidelity in bond formation. This route allows for the use of orthogonal protecting groups such as Fmoc, Trt, and Boc, which can be removed selectively without compromising the rest of the molecule. By eliminating the DKP pathway entirely, the process yields a crude product of significantly higher purity, simplifying the final isolation steps and enabling the recycling of solvents, which contributes to a more sustainable and economically efficient manufacturing cycle.
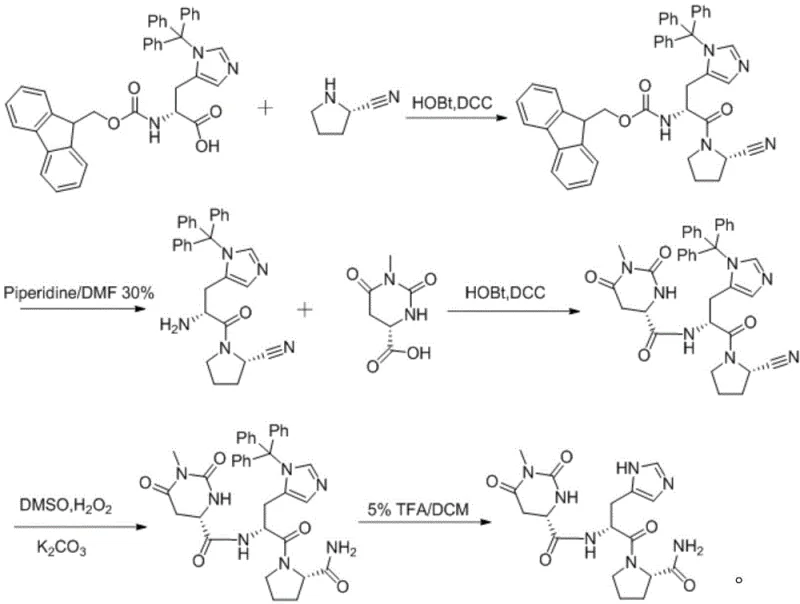
Mechanistic Insights into Peptide Coupling and Oxidative Hydrolysis
The core of this synthetic advancement lies in the precise control of amide bond formation and the subsequent transformation of the nitrile group. The initial condensation between 2-cyanopyrrolidine and the protected histidine derivative is facilitated by the activation of the carboxylic acid using dicyclohexylcarbodiimide (DCC) and 1-hydroxybenzotriazole (HOBt). This activation generates an active ester intermediate that reacts rapidly with the amine of the pyrrolidine ring at low temperatures (0-5°C), minimizing racemization of the chiral centers. The use of HOBt is particularly crucial here, as it suppresses the formation of N-acylurea byproducts and ensures that the coupling proceeds with high stereochemical retention, a critical factor for the biological activity of the final TRH analog. Following this, the deprotection of the Fmoc group using piperidine in DMF reveals the free amine necessary for the next coupling step with 1-methyl-4,5-dihydroorotic acid. This second condensation mirrors the efficiency of the first, utilizing the same DCC/HOBt system to link the orotic acid moiety to the growing peptide chain, creating the central scaffold of the Taltirelin molecule with minimal epimerization.
The final transformation involves the oxidative hydrolysis of the nitrile group to a primary amide, a step that replaces the harsh acidic hydrolysis used in older methods. The patent specifies the use of dimethyl sulfoxide (DMSO) and hydrogen peroxide in the presence of potassium carbonate. This mild oxidative system converts the electron-deficient nitrile carbon into the corresponding amide without affecting the sensitive imidazole ring of the histidine residue or the urea-like structure of the dihydroorotic acid component. The mechanism likely involves the nucleophilic attack of a perhydroxyl anion on the nitrile carbon, followed by hydrolysis to the amide. This gentle condition preserves the integrity of the protecting groups until the final global deprotection stage, where trifluoroacetic acid (TFA) is used to cleave the trityl and other acid-labile groups. The result is a clean conversion to the final API with an HPLC purity exceeding 99%, demonstrating the robustness of this oxidative strategy in handling complex, multifunctional intermediates.
How to Synthesize Taltirelin Efficiently
The synthesis of Taltirelin via this novel route is designed for operational simplicity and high reproducibility, making it an ideal candidate for technology transfer to commercial manufacturing sites. The process begins with the preparation of the key intermediate through the coupling of Fmoc-His(Trt)-OH with 2-cyanopyrrolidine, followed by Fmoc deprotection and subsequent coupling with the dihydroorotic acid derivative. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and temperature profiles for each reaction stage, are outlined below to ensure consistent quality and yield. Operators should pay close attention to the temperature control during the DCC activation steps to prevent side reactions and ensure the complete removal of the dicyclohexylurea (DCU) byproduct by filtration. The final oxidative and deprotection steps require careful monitoring by TLC or HPLC to determine the exact endpoint, ensuring that the nitrile is fully converted to the amide without over-oxidation or degradation of the peptide backbone.
- Condense 2-cyanopyrrolidine with amino and side-chain protected histidine to form an amide bond using DCC/HOBt.
- React the intermediate with 1-methyl-4,5-dihydroorotic acid to extend the peptide chain.
- Perform oxidative hydrolysis using DMSO and hydrogen peroxide, followed by deprotection with TFA to obtain pure Taltirelin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this DKP-free synthesis route offers substantial strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification process. By avoiding the formation of the structurally similar Diketopiperazine impurity, the need for resource-intensive preparative chromatography is significantly reduced or potentially eliminated in favor of simpler crystallization or extraction techniques. This reduction in downstream processing complexity translates directly into lower manufacturing costs, as it decreases the consumption of expensive stationary phases, solvents, and labor hours associated with purification. Furthermore, the high yields reported in the patent examples, such as the 97.5% yield in the initial coupling and 90.2% in the final isolation, indicate a highly atom-economical process that maximizes the output from raw materials, thereby reducing the cost per kilogram of the final API.
- Cost Reduction in Manufacturing: The elimination of toxic reagents like methyl chloroformate and strong acids like HBr removes the need for specialized corrosion-resistant reactors and complex waste neutralization systems. This shift to milder reagents such as DCC, HOBt, and hydrogen peroxide allows for the use of standard stainless steel equipment, significantly lowering capital investment barriers. Additionally, the ability to recover and reuse solvents like DMF and ethyl acetate, as noted in the patent's workup procedures, further drives down operational expenses. The overall process efficiency means that less raw material is wasted on byproducts, leading to a leaner and more cost-effective production model that can withstand market price fluctuations for starting materials.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as 2-cyanopyrrolidine and protected amino acids, ensures a robust supply chain that is less susceptible to disruptions compared to routes requiring custom-synthesized or hazardous precursors. The mild reaction conditions (0-30°C) reduce the risk of thermal runaways or equipment failures, ensuring consistent batch cycles and reliable delivery schedules. Moreover, the high purity of the crude product reduces the likelihood of batch failures during quality control testing, enhancing the predictability of supply for downstream formulation partners. This reliability is crucial for maintaining continuous production lines for finished dosage forms treating chronic conditions like spinocerebellar degeneration.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. The avoidance of heavy metal catalysts and halogenated solvents in the main reaction steps aligns with modern green chemistry principles and stringent regulatory standards. The oxidative hydrolysis step using hydrogen peroxide generates water as a benign byproduct, minimizing the environmental footprint of the manufacturing process. The scalability is further supported by the straightforward workup procedures involving simple filtrations and washes, which are easily adapted from laboratory to pilot and commercial scales without the need for complex engineering solutions. This makes the process highly attractive for contract development and manufacturing organizations (CDMOs) looking to offer sustainable and compliant manufacturing services.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Taltirelin synthesis method. These insights are derived directly from the experimental data and technical disclosures within patent CN109369779B, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for their specific supply chain needs.
Q: How does this method avoid Diketopiperazine (DKP) formation?
A: Unlike conventional methods that react methyl orotate with L-histidine-L-proline leading to DKP cyclization, this novel route starts with 2-cyanopyrrolidine. The nitrile group prevents the intramolecular cyclization that typically forms the DKP impurity, resulting in a cleaner reaction profile.
Q: What are the yield and purity improvements compared to traditional routes?
A: The patent data indicates significant improvements, with specific examples showing HPLC purity reaching 99.67% and yields around 90.2% in the final purification step. Intermediate steps also demonstrate high efficiency, such as 97.5% yield in the initial condensation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions (0-30°C) and avoids toxic reagents like methyl chloroformate or strong acids like HBr used in older methods. The ease of purification and solvent recyclability makes it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taltirelin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN109369779B to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this DKP-free method are fully realized in a GMP-compliant manufacturing environment. We are committed to delivering high-purity Taltirelin intermediates and APIs that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle the specific solvent recovery and mild reaction conditions required by this process, guaranteeing a consistent and reliable supply for your drug development and commercialization programs.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your project's cost structure and timeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how the elimination of DKP impurities and the use of milder reagents can impact your bottom line. Please contact us today to request specific COA data, route feasibility assessments, and samples to evaluate the quality of our Taltirelin offerings. Let us be your partner in bringing this vital therapeutic agent to patients efficiently and sustainably.
