Scalable One-Pot Synthesis of 5-Hydroxymethylthiazole for High-Volume Pharmaceutical Manufacturing
Introduction to Advanced Intermediate Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiretroviral intermediates, particularly those serving as building blocks for protease inhibitors like Ritonavir. Patent CN101863853B introduces a transformative one-pot cooking method for the preparation of 5-hydroxymethylthiazole, a pivotal precursor in the synthesis of life-saving HIV medications. This technology addresses long-standing inefficiencies in thiazole chemistry by integrating hydrolysis and reduction steps into a single reactor vessel, thereby streamlining the production workflow. By utilizing 2-chloro-5-chloromethylthiazole as the starting material, the process leverages accessible raw materials to generate high-purity outputs suitable for direct coupling in complex drug assembly. The strategic elimination of intermediate isolation steps not only simplifies operational protocols but also enhances the overall economic viability of large-scale manufacturing.
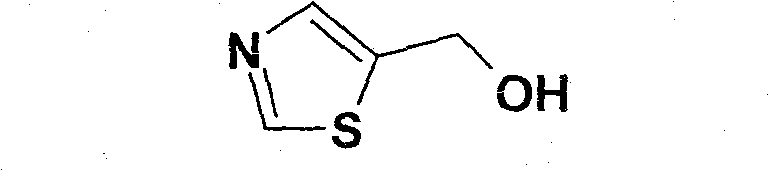
As a key structural motif in modern medicinal chemistry, the thiazole ring system found in 5-hydroxymethylthiazole is essential for the biological activity of numerous therapeutic agents. The ability to produce this scaffold efficiently without resorting to hazardous high-pressure conditions represents a significant leap forward in process safety and environmental compliance. For R&D directors and procurement specialists, understanding the nuances of this patented methodology is crucial for securing a stable supply chain of high-quality pharmaceutical intermediates. The following analysis dissects the technical merits and commercial implications of adopting this novel synthetic route over conventional multi-step procedures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-hydroxymethylthiazole has relied on cumbersome multi-step sequences that introduce significant operational bottlenecks. Prior art, such as the methods disclosed in US patents WO9414436 and US5541206, typically involves the reduction of ethyl thiazole carboxylate, a pathway that necessitates the use of expensive reducing agents and generates substantial stoichiometric waste. Furthermore, alternative routes described in US6710182 utilize a deamination strategy starting from 2-amino-5-hydroxymethylthiazole, which requires the preliminary synthesis of glycidaldehyde from acrolein using peroxides, introducing severe safety hazards associated with explosive peroxy-species. Another common approach involves catalytic hydrogenation using Pd/C under high pressure, as seen in US5773625; however, sulfur-containing heterocycles are notorious for poisoning noble metal catalysts, leading to inconsistent reaction rates and increased catalyst consumption. These traditional methods often mandate the isolation and purification of unstable intermediates like 2-chloro-5-hydroxymethylthiazole, which increases solvent usage, extends cycle times, and lowers overall throughput.
The Novel Approach
In stark contrast, the one-pot method detailed in CN101863853B circumvents these pitfalls by executing the transformation in a unified reaction environment. This innovative protocol initiates with the hydrolysis of 2-chloro-5-chloromethylthiazole in an aqueous medium at elevated temperatures, specifically around 80°C, to generate the hydroxy-intermediate in situ. Without isolating this species, the reaction mixture is immediately subjected to reductive dechlorination using a metal-acid system, such as zinc powder and concentrated hydrochloric acid. This seamless transition from hydrolysis to reduction eliminates the need for high-pressure hydrogenation equipment and avoids the catalyst poisoning issues inherent to sulfur chemistry. By consolidating two distinct chemical transformations into a single vessel, the process drastically reduces the physical footprint required for production and minimizes the handling of potentially hazardous intermediates. The result is a streamlined workflow that delivers the target molecule with high fidelity while adhering to stricter safety and cost-efficiency standards demanded by modern GMP facilities.
Mechanistic Insights into Metal-Acid Reductive Hydrolysis
The core chemical ingenuity of this process lies in the sequential activation of two distinct chloro-substituents on the thiazole ring under controlled conditions. Initially, the chloromethyl group at the 5-position undergoes nucleophilic substitution by water molecules, facilitated by thermal energy at 80°C. This hydrolysis step converts the reactive alkyl chloride into the corresponding primary alcohol, yielding 2-chloro-5-hydroxymethylthiazole. Crucially, this intermediate remains in the aqueous phase, stabilized by the reaction medium, preventing decomposition or polymerization that might occur during isolation. Subsequently, the introduction of zinc powder and hydrochloric acid initiates a single-electron transfer mechanism typical of dissolving metal reductions. The acidic environment protonates the nitrogen of the thiazole ring, increasing its electrophilicity, while the zinc serves as the electron donor to cleave the carbon-chlorine bond at the 2-position. This reductive dechlorination effectively replaces the chlorine atom with a hydrogen atom, finalizing the formation of the 5-hydroxymethylthiazole scaffold.
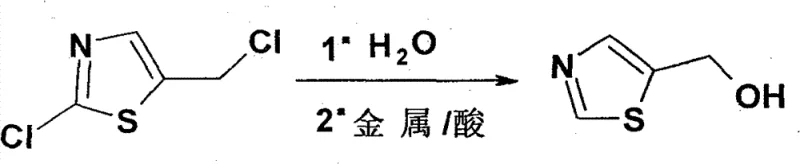
From an impurity control perspective, the one-pot nature of the reaction offers distinct advantages in managing byproduct profiles. In traditional stepwise syntheses, the isolation of the 2-chloro-5-hydroxymethylthiazole intermediate often leads to the accumulation of oligomeric impurities or hydrolysis byproducts that are difficult to remove in later stages. By maintaining the reaction continuity, the concentration of the reactive chloro-intermediate is kept low, minimizing side reactions such as ether formation or further degradation. The use of zinc and hydrochloric acid also ensures that any residual oxidizing species are quenched, contributing to the high chemical purity observed in the final crude product. Experimental data indicates that this mechanistic pathway consistently yields products with purity exceeding 98%, demonstrating the robustness of the metal-acid reduction system in tolerating the heterocyclic nitrogen and sulfur atoms without ring opening or desulfurization. This level of selectivity is paramount for ensuring that the intermediate meets the stringent specifications required for subsequent coupling reactions in API synthesis.
How to Synthesize 5-Hydroxymethylthiazole Efficiently
Implementing this one-pot synthesis requires precise control over reaction parameters to maximize yield and safety. The process begins by charging the reactor with 2-chloro-5-chloromethylthiazole and water, followed by heating to 80°C for approximately 2 hours to ensure complete hydrolysis. Once the first stage is confirmed, zinc powder is added, and concentrated hydrochloric acid is introduced slowly to manage the exotherm and gas evolution. The mixture is stirred for an additional 30 minutes to complete the reduction before cooling and filtration. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are critical for reproducibility.
- Hydrolyze 2-chloro-5-chloromethylthiazole in water at 80°C for 2 hours to form the intermediate 2-chloro-5-hydroxymethylthiazole.
- Add zinc powder and slowly drip concentrated hydrochloric acid into the same reactor to effect reductive dechlorination without isolating the intermediate.
- Filter off excess metal, adjust pH to approximately 9, extract with ethyl acetate, dry over anhydrous sodium sulfate, and distill to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot technology translates into tangible improvements in cost structure and supply reliability. The elimination of high-pressure hydrogenation steps removes the dependency on specialized autoclaves and expensive noble metal catalysts, which are subject to volatile market pricing and supply constraints. Furthermore, the ability to run the reaction in water as the primary solvent significantly reduces the volume of organic solvents required for the reaction phase, lowering both raw material costs and waste disposal expenses. The simplified workflow also means fewer unit operations, which directly correlates to reduced labor hours and lower utility consumption per kilogram of product produced. These factors combine to create a more resilient supply chain capable of responding rapidly to fluctuations in demand for antiretroviral therapies.
- Cost Reduction in Manufacturing: The removal of the intermediate isolation step is a primary driver for cost savings, as it eliminates the need for separate filtration, drying, and charging operations that consume time and resources. By avoiding the use of palladium on carbon catalysts, the process sidesteps the significant capital expenditure associated with catalyst recovery systems and the risk of catalyst loss due to sulfur poisoning. Additionally, the use of commodity chemicals like zinc powder and hydrochloric acid instead of specialized reducing agents ensures that raw material costs remain stable and predictable. The overall reduction in processing steps leads to a substantial decrease in the cost of goods sold, allowing for more competitive pricing in the global marketplace.
- Enhanced Supply Chain Reliability: Relying on a process that does not require high-pressure infrastructure mitigates the risk of production delays caused by equipment maintenance or regulatory inspections of pressure vessels. The robustness of the metal-acid reduction system ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that could disrupt downstream API production schedules. Since the starting material, 2-chloro-5-chloromethylthiazole, is a widely available commodity, the supply chain is less vulnerable to shortages of exotic reagents. This stability is crucial for maintaining continuous manufacturing lines for critical medicines like Ritonavir, where interruptions can have serious public health consequences.
- Scalability and Environmental Compliance: The one-pot design is inherently scalable, as demonstrated by the successful translation from laboratory flasks to 1500L industrial reactors without loss of efficiency. The aqueous nature of the reaction medium aligns with green chemistry principles by minimizing the use of volatile organic compounds during the synthesis phase. Waste streams are primarily composed of zinc salts and aqueous acids, which are easier to treat and neutralize compared to the complex organic waste generated by multi-step organic syntheses. This simplified waste profile facilitates compliance with increasingly stringent environmental regulations, reducing the administrative burden and potential fines associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: Why is the one-pot method superior to traditional catalytic hydrogenation for this intermediate?
A: Traditional methods often require high-pressure hydrogenation reactors and expensive Pd/C catalysts, which are prone to poisoning by sulfur compounds. The one-pot metal-acid reduction operates at atmospheric pressure, significantly reducing equipment costs and safety risks while avoiding catalyst deactivation issues.
Q: What is the expected purity and yield of 5-hydroxymethylthiazole using this process?
A: According to the experimental data in patent CN101863853B, the process consistently achieves a chemical purity of greater than 98% with yields ranging between 66% and 69% on scales up to 1500L, making it suitable for direct use in downstream API synthesis.
Q: Does this process eliminate the need for intermediate isolation?
A: Yes, the core innovation is performing both the hydrolysis and the reduction steps in the same reactor. This eliminates the need to isolate and purify the unstable 2-chloro-5-hydroxymethylthiazole intermediate, thereby reducing solvent consumption and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Hydroxymethylthiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of multinational drug manufacturers. We are committed to delivering 5-hydroxymethylthiazole with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our facility is equipped to handle the specific requirements of thiazole chemistry, including corrosion-resistant reactors and specialized waste treatment systems, guaranteeing a safe and compliant production environment.
We invite you to collaborate with us to optimize your supply chain for antiretroviral intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of the one-pot synthesis technology can enhance your operational efficiency and reduce overall manufacturing costs.
