Scalable Copper-Catalyzed Synthesis of Alkyl Phosphonyl Compounds for Global Pharmaceutical Supply Chains
The global demand for high-purity organophosphorus compounds continues to surge, driven by their critical role in synthesizing life-saving pharmaceuticals and advanced agrochemicals. A significant technological breakthrough in this sector is detailed in Chinese Patent CN110256489B, which discloses a novel preparation method for alkyl phosphonyl compounds. This innovation addresses long-standing challenges in the industry by utilizing readily available alkyl carboxylic acids as starting materials, replacing the traditionally hazardous and expensive phosphine reagents. The versatility of this method is evident in its ability to produce diverse structures, including key intermediates for drugs like Fosinopril and Tenofovir, as well as herbicides like Glyphosate. By shifting the synthetic paradigm towards milder conditions and safer reagents, this technology offers a robust pathway for the reliable alkyl phosphonate supplier to enhance production efficiency while maintaining stringent quality standards required by international regulatory bodies.
The synthesis of organophosphorus compounds has historically been plagued by significant safety and operational hurdles, particularly when constructing the carbon-phosphorus bond. Conventional methodologies often rely on the reaction of alkyl Grignard reagents with diphenylphosphine chloride, a process depicted in older literature that necessitates rigorous anhydrous conditions and low temperatures to prevent side reactions. 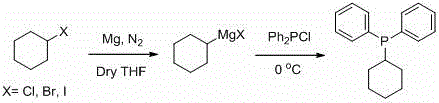 Furthermore, the traditional Arbuzov reaction between halogenated hydrocarbons and trialkyl phosphites typically requires high temperatures, leading to energy-intensive processes and the generation of substantial halogenated waste streams. These legacy methods not only pose severe safety risks due to the pyrophoric nature of the reagents but also suffer from limited substrate scope and difficult post-treatment procedures, making the commercial scale-up of complex organophosphorus intermediates economically challenging for many manufacturers.
Furthermore, the traditional Arbuzov reaction between halogenated hydrocarbons and trialkyl phosphites typically requires high temperatures, leading to energy-intensive processes and the generation of substantial halogenated waste streams. These legacy methods not only pose severe safety risks due to the pyrophoric nature of the reagents but also suffer from limited substrate scope and difficult post-treatment procedures, making the commercial scale-up of complex organophosphorus intermediates economically challenging for many manufacturers.
In stark contrast, the novel approach disclosed in the patent introduces a transformative strategy that leverages the abundance and stability of alkyl carboxylic acids. The core innovation involves a copper-catalyzed oxidative coupling reaction where the carboxylic acid is first activated and then coupled with a phosphorus reagent such as diphenylphosphine oxide. 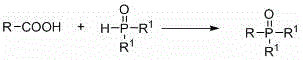 This method operates under remarkably mild conditions, typically ranging from room temperature to 80°C, and utilizes a catalytic system comprising copper salts and bipyridine ligands. The transition from hazardous halides and Grignard reagents to benign carboxylic acids represents a fundamental shift in process safety, enabling cost reduction in pharmaceutical intermediate manufacturing by simplifying reactor requirements and waste treatment protocols. The broad applicability of this route allows for the synthesis of various alkyl phosphonyl derivatives, including those with cyclic, aromatic, and heteroatomic substituents, without compromising yield or purity.
This method operates under remarkably mild conditions, typically ranging from room temperature to 80°C, and utilizes a catalytic system comprising copper salts and bipyridine ligands. The transition from hazardous halides and Grignard reagents to benign carboxylic acids represents a fundamental shift in process safety, enabling cost reduction in pharmaceutical intermediate manufacturing by simplifying reactor requirements and waste treatment protocols. The broad applicability of this route allows for the synthesis of various alkyl phosphonyl derivatives, including those with cyclic, aromatic, and heteroatomic substituents, without compromising yield or purity.
Mechanistic Insights into Copper-Catalyzed Oxidative Coupling
The mechanistic elegance of this synthesis lies in the synergistic interaction between the activating agents and the transition metal catalyst. Initially, the alkyl carboxylic acid undergoes activation through the formation of an O-acylisourea intermediate facilitated by dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP). This activation step is crucial as it converts the relatively inert carboxylic acid into a highly reactive species capable of undergoing subsequent decarboxylative or coupling transformations. The presence of hydrogen peroxide in the initial stirring phase suggests an oxidative environment that may assist in generating radical species or high-valent phosphorus intermediates, setting the stage for the carbon-phosphorus bond formation. This careful orchestration of reagents ensures that the reaction proceeds with high atom economy, minimizing the formation of undesired byproducts that often complicate downstream purification in traditional phosphorus chemistry.
Following the activation phase, the introduction of the copper catalytic system drives the coupling efficiency to new heights. The copper salt, which can range from copper sulfate to copper trifluoromethanesulfonate, coordinates with nitrogen-based ligands such as 2,2'-bipyridine or phenanthroline derivatives to form an active catalytic complex. This complex likely mediates a single-electron transfer process or a concerted insertion mechanism that couples the alkyl fragment derived from the acid with the phosphorus nucleophile. The choice of ligand is critical, as electron-rich or sterically hindered bipyridines can fine-tune the redox potential of the copper center, thereby optimizing the reaction rate and selectivity. This level of mechanistic control allows for the synthesis of high-purity alkyl phosphonyl compounds with minimal impurity profiles, a factor of paramount importance for R&D directors focusing on the杂质谱 (impurity profile) of drug substances.
How to Synthesize Alkyl Phosphonyl Compounds Efficiently
The practical implementation of this patented methodology offers a streamlined workflow that is highly amenable to both laboratory-scale optimization and industrial production. The process begins with the activation of the carboxylic acid substrate, followed by the addition of the phosphorus source and catalyst, and concludes with a straightforward workup procedure. Detailed standardized synthesis steps for specific derivatives, including reaction stoichiometry and purification parameters, are provided in the sections below to guide technical teams in replicating these results.
- Activate the alkyl carboxylic acid by stirring with DMAP, DCC, and hydrogen peroxide in a solvent like dichloromethane at 0-40°C for 2 hours.
- Filter the reaction mixture to remove dicyclohexylurea byproducts and concentrate the filtrate to obtain the activated acid intermediate.
- Add the phosphorus reagent (e.g., diphenylphosphine oxide) along with a copper catalyst and bipyridine ligand, then stir at room temperature to 80°C until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route presents compelling economic and logistical benefits that extend beyond simple yield improvements. The primary advantage stems from the substitution of exotic and hazardous starting materials with commodity chemicals; alkyl carboxylic acids are produced on a massive global scale for the food and polymer industries, ensuring a stable and competitive pricing structure. This shift drastically simplifies the sourcing strategy, reducing dependency on specialized chemical suppliers who often dictate terms for niche phosphorus reagents. Furthermore, the elimination of pyrophoric reagents like Grignard solutions and phosphine chlorides removes the need for specialized storage facilities and hazardous material transport protocols, leading to substantial cost savings in logistics and insurance.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the simplification of the reaction infrastructure. By operating at near-ambient temperatures and avoiding the need for cryogenic cooling or high-pressure reactors, the energy consumption per kilogram of product is significantly lowered. Additionally, the use of earth-abundant copper catalysts instead of precious metals like palladium or platinum further reduces the raw material cost basis. The simplified post-treatment process, which often involves basic filtration and chromatography rather than complex distillation or scrubbing of toxic gases, translates directly into lower operational expenditures and higher overall throughput for the manufacturing facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the robustness of the raw material base. Since the method utilizes widely available carboxylic acids and stable phosphine oxides, the risk of supply disruption due to the discontinuation of a niche reagent is virtually eliminated. This stability allows for longer-term forecasting and inventory planning, ensuring reducing lead time for high-purity phosphine ligands and intermediates. The mild reaction conditions also mean that the process is less sensitive to minor fluctuations in utility supplies, such as cooling water temperature, making the production schedule more predictable and reliable for downstream customers awaiting critical API intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The avoidance of halogenated waste streams, which are typical of the Arbuzov reaction, simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. The high selectivity of the copper-catalyzed system minimizes the generation of organic waste, facilitating easier compliance with increasingly stringent environmental regulations. This 'cleaner' profile not only mitigates regulatory risk but also enhances the marketability of the final product to eco-conscious pharmaceutical clients who prioritize sustainable supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkyl phosphonyl synthesis technology. These insights are derived directly from the experimental data and scope defined within the patent documentation, providing clarity on the method's capabilities and limitations for potential partners.
Q: What are the safety advantages of this new alkyl phosphonate synthesis method?
A: Unlike conventional methods that utilize highly toxic and pyrophoric phosphine chlorides or Grignard reagents requiring strict anhydrous conditions, this patent utilizes stable alkyl carboxylic acids and diphenylphosphine oxide. This significantly reduces operational hazards and eliminates the need for specialized hazardous material handling infrastructure.
Q: Can this method be used to synthesize precursors for Fosinopril?
A: Yes, the patent explicitly demonstrates the synthesis of 4-phenylbutylphosphonic acid derivatives, which are critical intermediates for Fosinopril, an antihypertensive drug. The method allows for the direct conversion of readily available phenylvaleric acid into the necessary phosphonate structure with high purity.
Q: How does the copper catalytic system improve yield compared to thermal methods?
A: The use of a copper salt catalyst coordinated with nitrogen ligands like 2,2'-bipyridine facilitates the oxidative coupling at mild temperatures (room temperature to 80°C). This avoids the high-temperature degradation often seen in traditional Arbuzov-type reactions, resulting in consistently higher yields and simpler purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Phosphonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes described in patent CN110256489B for the production of high-value organophosphorus intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of copper-catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global pharmaceutical industry. We are committed to leveraging this advanced chemistry to deliver superior value to our clients.
We invite forward-thinking organizations to collaborate with us to explore the full commercial potential of this technology. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need to validate the feasibility of a new phosphonate derivative, our technical team is ready to assist. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and supply chain security.
