Scalable Manufacturing of Chiral Bicyclic Amino Esters for Advanced Antiviral Drug Production
Scalable Manufacturing of Chiral Bicyclic Amino Esters for Advanced Antiviral Drug Production
The rapid advancement of antiviral therapeutics, particularly influenza virus RNA polymerase inhibitors like Pimodivir, has placed immense pressure on the supply chain for complex chiral intermediates. A pivotal breakthrough in this domain is detailed in patent CN111454166A, which discloses a robust and economically viable process for the preparation of (2S,3S)-3-amino-bicyclo[2.2.2]octane-2-carboxylate. This specific bicyclic amino acid derivative serves as a critical structural fragment in next-generation antiviral agents, addressing the urgent need for drug resistance solutions. The patented methodology transcends traditional synthetic limitations by employing a novel chiral reductive amination strategy coupled with a stereoselective configuration inversion. For global pharmaceutical developers, this represents a significant leap forward, offering a pathway to secure reliable supplies of high-purity intermediates while mitigating the safety risks associated with older, more hazardous chemistries. As a leading entity in fine chemical synthesis, understanding the nuances of this technology is essential for optimizing the manufacturing of these vital life-saving medicines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the (2S,3S)-3-amino-bicyclo[2.2.2]octane scaffold has been plagued by severe operational hazards and economic inefficiencies. As illustrated in the prior art, specifically Route One, the traditional approach relies heavily on the Curtius rearrangement. This classical method necessitates the use of diphenyl phosphoryl azide, an explosive and highly toxic reagent that poses unacceptable safety risks during kilogram-to-ton scale production. Furthermore, Route One depends on expensive chiral organic bases, such as quinidine derivatives, to induce asymmetry during the alcoholysis step, driving up the raw material costs significantly. The overall yield of these legacy processes is often dismally low, frequently reported below 20%, which creates a bottleneck for commercial viability. Additionally, alternative routes involving nitro-compounds (Route Three) introduce further instability and safety concerns due to the potential explosiveness of nitro intermediates, making them unsuitable for modern GMP manufacturing environments.
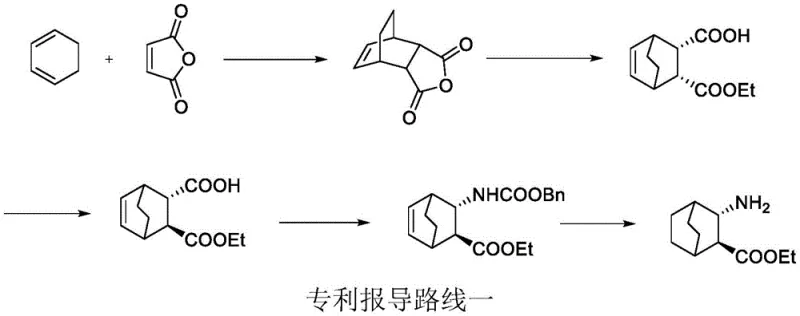
The Novel Approach
In stark contrast to these perilous legacy methods, the innovative process described in CN111454166A introduces a streamlined and safe synthetic trajectory. The new route initiates with a condensation reaction between 3-carbonyl-bicyclo[2.2.2]octane-2-formate and a chiral amine, bypassing the need for dangerous azide chemistry entirely. By leveraging a reductive amination followed by a strategic base-catalyzed epimerization, the process achieves a total yield exceeding 65%, which is a threefold improvement over the conventional Curtius route. This approach not only enhances material throughput but also drastically simplifies the purification workflow. The elimination of cryogenic conditions and explosive reagents means that the process can be executed in standard stainless steel reactors, significantly lowering the barrier to entry for contract manufacturing organizations. This shift from hazard-intensive chemistry to a controlled, catalytic sequence marks a paradigm shift in the production of bicyclic amino acid derivatives for the pharmaceutical industry.
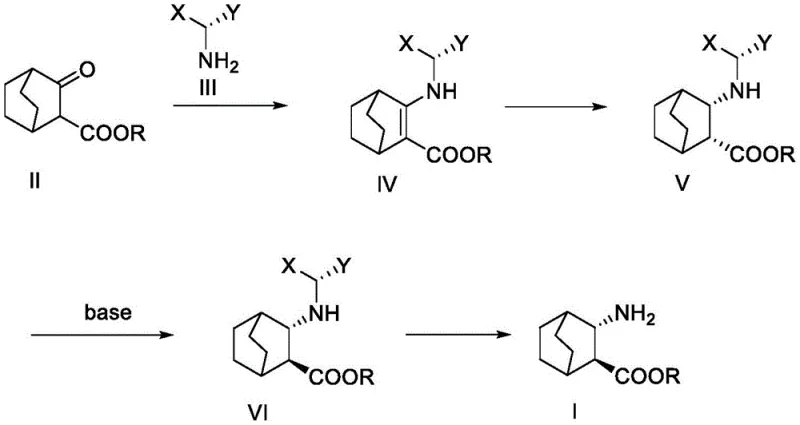
Mechanistic Insights into Chiral Reductive Amination and Epimerization
The core brilliance of this patented technology lies in its sophisticated control over stereochemistry through a combination of kinetic and thermodynamic factors. The process begins with the formation of an enamine intermediate via the condensation of the ketone starting material with a chiral amine auxiliary, such as S-1-phenylethylamine or S-1-(1-naphthyl)ethylamine. The steric bulk of the chiral amine directs the subsequent reduction step, whether performed via metal-catalyzed hydrogenation using platinum or palladium, or through hydride reduction with sodium triacetoxyborohydride. This step selectively generates the (2R,3S)-diastereomer with high fidelity. The true mechanistic masterpiece, however, is the subsequent configuration inversion. Under strong basic conditions, typically employing sodium tert-butoxide in a THF/tert-butanol system, the ester moiety at the C2 position undergoes epimerization. This transformation is driven by the thermodynamic stability of the (2S,3S)-isomer, where the bulky amino and ester groups adopt a more favorable spatial arrangement. This base-mediated equilibration ensures that even if the initial reduction yields a mixture, the system converges towards the desired stereochemical outcome, thereby maximizing the efficiency of the chiral pool usage.
Impurity control is inherently built into this mechanistic design, addressing a primary concern for R&D directors focused on regulatory compliance. The use of distinct chiral auxiliaries allows for easy monitoring of diastereomeric ratios via standard HPLC or NMR techniques throughout the synthesis. The final hydrogenolysis step, which removes the chiral auxiliary to reveal the free amine, is highly selective and clean, minimizing the formation of racemization byproducts. Because the epimerization step effectively 'corrects' any minor stereochemical deviations from the reduction phase, the final crude product often possesses a chiral purity approaching 97-98%. A simple crystallization or salt formation step post-deprotection can then effortlessly elevate the optical purity to greater than 99.5% ee. This robust impurity profile reduces the need for complex chromatographic separations, which are often the most costly and time-consuming aspects of intermediate manufacturing, ensuring a consistent and high-quality supply of the active pharmaceutical ingredient precursor.
How to Synthesize (2S,3S)-3-Amino-Bicyclo[2.2.2]Octane-2-Carboxylate Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize the benefits of the novel pathway. The process is divided into four distinct operational stages: enamine formation, stereoselective reduction, base-catalyzed inversion, and final deprotection. Each stage utilizes commercially available reagents and standard unit operations, making the technology transfer to pilot and production plants straightforward. The following guide outlines the critical operational phases derived from the patent examples, emphasizing the specific conditions required to achieve the reported high yields and purity levels. Operators should pay close attention to the stoichiometry of the chiral amine and the temperature control during the epimerization phase to ensure optimal results.
- Condense 3-carbonyl-bicyclo[2.2.2]octane-2-formate with a chiral amine (e.g., S-1-phenylethylamine) under acidic conditions to form an enamine intermediate.
- Perform reductive amination or catalytic hydrogenation on the enamine to obtain the (2R,3S)-amino ester derivative with high diastereoselectivity.
- Treat the (2R,3S)-isomer with a strong base like sodium tert-butoxide to invert the ester configuration, yielding the thermodynamically stable (2S,3S)-isomer.
- Execute catalytic hydrogenolysis to remove the chiral auxiliary protecting group, isolating the final target amino ester with >99.5% chiral purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative advantages in terms of cost structure and logistical reliability. The shift away from proprietary, expensive chiral catalysts and hazardous reagents fundamentally alters the cost of goods sold (COGS) for this intermediate. By utilizing commodity chemicals like ethyl 3-oxobicyclo[2.2.2]octane-2-carboxylate and simple chiral amines, the raw material basket becomes significantly more stable and less prone to price volatility. Furthermore, the elimination of special waste treatment protocols required for azide and heavy metal residues translates into substantial downstream cost savings in environmental compliance and waste disposal. This process optimization directly contributes to cost reduction in API manufacturing, allowing pharmaceutical companies to maintain healthier margins while securing a sustainable supply of critical antiviral components.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the replacement of high-cost reagents with affordable alternatives. Traditional methods often require stoichiometric amounts of expensive chiral bases or precious metal catalysts that are difficult to recover. In contrast, this novel process uses catalytic hydrogenation or inexpensive borohydride reductants, and the chiral amine auxiliary can potentially be recovered and recycled after the final deprotection step. Additionally, the higher overall yield (>65% vs <20%) means that less starting material is wasted, effectively tripling the output per batch without increasing reactor volume. This dramatic improvement in atom economy and resource utilization leads to a significantly lower unit cost, making the final drug product more accessible to the market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on niche reagents with limited global suppliers. The reagents specified in this patent, such as sodium tert-butoxide, palladium on carbon, and common organic solvents like toluene and ethanol, are ubiquitous in the fine chemical industry. This ubiquity ensures that production is not held hostage by single-source supplier bottlenecks. Moreover, the mild reaction conditions—operating at ambient or moderately elevated temperatures rather than cryogenic lows—reduce the risk of batch failures due to equipment malfunction or cooling capacity limitations. This operational robustness ensures consistent lead times and reduces the likelihood of supply disruptions, providing a reliable pharmaceutical intermediate supplier advantage for downstream partners.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, but this route is inherently designed for large-scale industrial production. The absence of explosive azides removes a major regulatory hurdle, simplifying the permitting process for new manufacturing lines. The waste stream is predominantly composed of benign organic salts and solvents, which are easier to treat and dispose of compared to the toxic byproducts of the Curtius rearrangement. This alignment with green chemistry principles not only lowers environmental fees but also enhances the corporate social responsibility profile of the manufacturing partner. The process is fully compatible with existing multipurpose reactors, facilitating rapid commercial scale-up of complex pharmaceutical intermediates from pilot batches to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical application of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of this new synthesis route compared to the Curtius rearrangement method?
A: The novel route eliminates the use of explosive azide reagents and diphenyl phosphoryl azide required in the traditional Curtius rearrangement, significantly reducing operational hazards and facilitating safer large-scale production.
Q: How does the process ensure high chiral purity without expensive chiral catalysts?
A: The method utilizes a chiral pool strategy with readily available chiral amines (like S-1-phenylethylamine) for stereocontrol, followed by a base-catalyzed epimerization step that thermodynamically drives the formation of the desired (2S,3S) configuration, achieving over 99.5% purity after crystallization.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the process operates under mild reaction conditions without the need for cryogenic temperatures or specialized high-pressure equipment beyond standard hydrogenation reactors, making it highly adaptable for commercial manufacturing from 100 kgs to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3S)-3-Amino-Bicyclo[2.2.2]Octane-2-Carboxylate Supplier
The technological advancements detailed in CN111454166A underscore the evolving landscape of antiviral intermediate synthesis, demanding partners who can navigate complex chemistry with precision and safety. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this reductive amination and epimerization sequence, ensuring stringent purity specifications are met for every batch. With rigorous QC labs and a commitment to process excellence, we guarantee that our (2S,3S)-3-amino-bicyclo[2.2.2]octane-2-carboxylate meets the highest global standards for pharmaceutical applications, providing a secure foundation for your drug development pipeline.
We invite you to collaborate with us to leverage this superior synthetic route for your upcoming projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel method can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your time-to-market with a supply chain partner dedicated to innovation, quality, and reliability.
