Scalable Synthesis of Naphthalenone-Based Bisphenol Monomers for Advanced Polyarylene Ethers
Introduction to Advanced Bisphenol Monomer Technology
The development of high-performance polymer materials relies heavily on the availability of specialized monomers with precise structural attributes. Patent CN109851563B discloses a robust and efficient preparation method for a bisphenol monomer containing a naphthalenone structure, specifically designated as DHPPZ. This technology addresses critical challenges in the synthesis of polyarylene ether materials, which are increasingly demanded for their superior thermal stability and mechanical properties in advanced engineering applications. The disclosed route utilizes a strategic two-step synthesis involving a copper-catalyzed coupling reaction followed by a Lewis acid-mediated reduction, achieving a target monomer purity of 98% with yields ranging from 85% to 90%. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a significant opportunity to optimize the supply chain for high-value polymer intermediates while ensuring consistent quality and cost-effectiveness in large-scale manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing complex bisphenol monomers with fused heterocyclic systems often suffer from significant inefficiencies that hinder commercial scalability. Conventional methodologies frequently rely on precious metal catalysts such as Palladium, which not only inflate raw material costs but also introduce stringent requirements for metal residue removal to meet pharmaceutical or electronic grade specifications. Furthermore, many existing processes require harsh reaction conditions, including extremely high temperatures or strong acidic environments, which can lead to side reactions, lower selectivity, and the formation of difficult-to-remove impurities. The solubility issues inherent in rigid aromatic systems often necessitate the use of exotic, high-boiling polar aprotic solvents that are difficult to recover and recycle, thereby increasing the environmental footprint and operational expenditure of the manufacturing process. These factors collectively create bottlenecks in the supply chain, leading to longer lead times and higher volatility in pricing for downstream polymer manufacturers seeking reliable sources of specialized monomers.
The Novel Approach
The innovative strategy outlined in the patent data overcomes these historical barriers by employing a cost-effective Copper(I) Iodide (CuI) catalytic system paired with 1,10-phenanthroline as a bidentate ligand. This Ullmann-type coupling approach facilitates the simultaneous formation of C-O and C-N bonds under relatively mild thermal conditions, typically between 130°C and 160°C, which significantly reduces energy consumption compared to traditional high-temperature fusion methods. The subsequent demethylation step utilizes a Lewis acid system comprising Aluminum Chloride and Potassium Iodide in acetonitrile, a process notable for its tolerance to moisture and lack of requirement for rigorous solvent dehydration. This 'one-pot' capability in the first step and the simplified workup in the second step drastically streamline the production workflow. By eliminating the need for expensive noble metals and complex purification protocols, this novel approach offers a compelling pathway for cost reduction in advanced materials manufacturing, ensuring a more stable and economical supply of high-purity DHPPZ monomers for the global market.
Mechanistic Insights into Cu-Catalyzed Ullmann Coupling and Lewis Acid Reduction
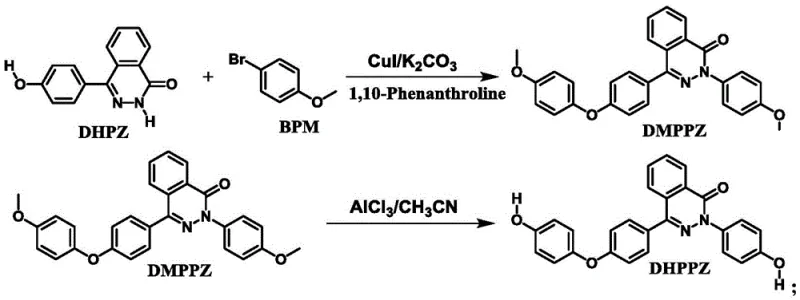
The core of this synthetic breakthrough lies in the mechanistic efficiency of the copper-catalyzed cross-coupling reaction, which serves as the foundation for constructing the complex molecular architecture of the DMPPZ intermediate. In this transformation, the phenolic hydroxyl group of the DHPZ precursor is activated by a base, typically potassium carbonate, to form a nucleophilic phenoxide species. This species then engages in a catalytic cycle with the aryl bromide (BPM) mediated by the CuI/1,10-phenanthroline complex. The ligand plays a crucial role in stabilizing the copper center and facilitating the oxidative addition and reductive elimination steps necessary for C-O bond formation. Simultaneously, the reaction conditions promote C-N bond formation, effectively assembling the diphthalazinone core with high regioselectivity. The use of a gradient temperature profile, ramping from room temperature to 140-160°C, allows for controlled reaction kinetics that minimize the formation of homocoupling byproducts, thereby ensuring a clean reaction profile that simplifies downstream isolation and purification efforts significantly.
Following the successful assembly of the dimethoxy intermediate DMPPZ, the process transitions to a Lewis acid-mediated demethylation to reveal the active phenolic groups required for polymerization. This step employs Aluminum Chloride (AlCl3) in the presence of Potassium Iodide (KI) within an acetonitrile solvent system. The mechanism involves the coordination of the Lewis acid to the ether oxygen, weakening the C-O bond and facilitating nucleophilic attack by iodide ions or chloride species to cleave the methyl group. A distinct advantage of this specific protocol is its operational simplicity; unlike traditional demethylation methods using boron tribromide which require strictly anhydrous conditions and cryogenic temperatures, this system operates effectively at moderate temperatures between 60°C and 90°C. The tolerance for ambient moisture levels eliminates the need for energy-intensive solvent drying procedures, representing a substantial improvement in process safety and throughput. This mechanistic robustness ensures that the final DHPPZ product maintains high structural integrity and purity, essential for producing polyarylene ethers with consistent performance characteristics.
How to Synthesize DHPPZ Efficiently
The synthesis of DHPPZ described in the patent offers a streamlined pathway suitable for both laboratory optimization and industrial scale-up. The process begins with the activation of the starting material DHPZ using potassium carbonate in a high-boiling solvent environment, followed by the introduction of the coupling partners and the copper catalyst system. Careful control of the temperature gradient is essential to maximize the conversion to the DMPPZ intermediate while minimizing thermal degradation. Once the intermediate is isolated and purified via recrystallization, the subsequent reduction step requires precise stoichiometric control of the Lewis acid and iodide source to ensure complete demethylation without over-reaction. Detailed standardized operating procedures regarding reagent addition rates, stirring speeds, and quenching protocols are critical for maintaining batch-to-batch consistency. For comprehensive technical guidance on executing this synthesis with optimal safety and yield parameters, please refer to the step-by-step protocol provided below.
- Perform a one-pot salt formation and Ullmann coupling reaction using DHPZ, K2CO3, BPM, CuI catalyst, and 1,10-phenanthroline ligand at 130-160°C to obtain DMPPZ intermediate.
- Purify the DMPPZ intermediate via recrystallization using DMAc solvent to ensure high purity before the reduction step.
- Execute Lewis acid reduction by reacting DMPPZ with AlCl3 and KI in acetonitrile at 60-90°C, followed by aqueous workup and recrystallization to yield DHPPZ.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound benefits for procurement managers and supply chain directors focused on optimizing total cost of ownership and ensuring supply continuity. The shift from precious metal catalysts to abundant copper-based systems fundamentally alters the cost structure of the monomer production, removing the volatility associated with palladium pricing and reducing the capital tied up in catalyst recovery infrastructure. Furthermore, the mild reaction conditions and simplified workup procedures translate directly into reduced utility consumption and shorter cycle times, allowing manufacturing facilities to increase throughput without significant capital investment in new reactor hardware. The high purity and yield reported in the patent data suggest a robust process with minimal waste generation, aligning with modern sustainability goals and reducing the burden on waste treatment facilities. These factors combine to create a highly competitive supply proposition for buyers of specialty chemical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive palladium catalysts and the use of readily available reagents like CuI and AlCl3 significantly lowers the direct material costs associated with monomer production. Additionally, the avoidance of rigorous solvent dehydration steps in the reduction phase reduces energy consumption and the need for specialized drying equipment, leading to substantial operational savings. The high yield of 85%-90% ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final DHPPZ product and offering a clear economic advantage over less efficient conventional synthetic routes.
- Enhanced Supply Chain Reliability: By utilizing common industrial chemicals and avoiding reliance on scarce or geopolitically sensitive precious metals, this manufacturing route enhances the resilience of the supply chain. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor variations in raw material quality or environmental fluctuations. This stability allows suppliers to offer more reliable lead times and consistent delivery schedules, which is critical for downstream polymer manufacturers who operate on tight production schedules and cannot afford interruptions in their raw material feedstock.
- Scalability and Environmental Compliance: The process is inherently scalable due to its use of standard unit operations such as reflux, filtration, and crystallization, which are well-understood and easily implemented in large-scale reactors. The reduced generation of heavy metal waste and the ability to operate under milder conditions contribute to a lower environmental footprint, facilitating easier compliance with increasingly stringent environmental regulations. This 'green chemistry' aspect not only mitigates regulatory risk but also enhances the brand value of the final polymer products by associating them with sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of DHPPZ monomers. These answers are derived directly from the technical specifications and beneficial effects detailed in the patent literature, providing clarity on the feasibility and advantages of this specific synthetic methodology. Understanding these details is crucial for technical teams evaluating the integration of this monomer into their existing polymer formulations or for procurement teams assessing the long-term viability of the supply source.
Q: What are the key advantages of the Ullmann coupling method described in CN109851563B?
A: The patented method utilizes a cost-effective Copper(I) Iodide (CuI) catalyst system with 1,10-phenanthroline ligand, avoiding expensive Palladium catalysts. It achieves high yields (85%-90%) and purity (98%) under relatively mild thermal conditions (130-160°C), significantly reducing raw material and energy costs compared to traditional noble metal catalysis.
Q: How does the Lewis acid reduction step improve process efficiency?
A: The demethylation of the DMPPZ intermediate using Aluminum Chloride (AlCl3) and Potassium Iodide (KI) in acetonitrile proceeds without the need for rigorous solvent dehydration pretreatment. This simplifies the operational workflow, reduces preparation time, and lowers the overall consumption of drying agents and energy, enhancing the commercial viability of the process.
Q: What represents the primary application for DHPPZ monomers?
A: DHPPZ monomers are critical precursors for synthesizing polyarylene ether materials. The unique twisted, non-coplanar naphthalenone structure imparts exceptional heat resistance and solubility to the resulting polymers, overcoming the poor solubility issues often found in traditional semi-crystalline polyarylene ether ketones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DHPPZ Supplier
As the demand for high-performance polyarylene ethers continues to grow across industries ranging from aerospace to electronics, securing a dependable source of high-quality monomers like DHPPZ is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of copper-catalyzed couplings and Lewis acid reductions, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand the critical nature of supply chain continuity and are committed to delivering consistent quality that enables your downstream polymerization processes to run smoothly and efficiently.
We invite you to engage with our technical team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that leverages our process expertise to optimize your total procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
