Scalable Synthesis of Naphthalenone-Based Bisphenol Monomers for High-Performance Polymers
Introduction to Advanced Bisphenol Monomer Technology
The development of high-performance polymeric materials often hinges on the availability of specialized monomers that can impart unique thermal and mechanical properties to the final resin. Patent CN109851563B discloses a highly efficient preparation method for a bisphenol monomer containing a naphthalenone structure, specifically designated as DHPPZ. This innovation addresses critical limitations in the field of polyarylene ether synthesis by introducing a monomer with a novel fully aromatic ring system and a twisted, non-coplanar configuration. Such structural characteristics are essential for breaking the crystallinity of traditional polymers, thereby enhancing solubility without sacrificing thermal stability. For R&D directors and procurement specialists in the advanced materials sector, understanding this synthetic pathway offers a strategic advantage in sourcing reliable polymer intermediate suppliers who can deliver complex structures with consistent quality.
The significance of this technology extends beyond mere academic interest; it represents a tangible solution for manufacturing next-generation engineering plastics and specialty coatings. The patent outlines a robust two-step synthetic route that balances high yield with operational simplicity, making it particularly attractive for commercial scale-up. By leveraging copper-catalyzed coupling followed by a specific Lewis acid reduction, the process avoids the extreme conditions often associated with similar transformations. This report analyzes the technical depth of this invention, providing actionable insights for supply chain heads looking to secure a steady stream of high-purity OLED material precursors or advanced polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional polyarylene ether materials, such as standard polyarylene ether ketones, have long been valued for their chemical stability and mechanical strength. However, these conventional polymers suffer from a significant drawback: they are semi-crystalline and exhibit poor solubility, often dissolving only in aggressive solvents like concentrated sulfuric acid at room temperature. This limitation severely restricts their processability and application scope, particularly in industries requiring solution-based coating or spinning techniques. Furthermore, historical synthetic routes for modifying these backbones often involve harsh reaction conditions, multi-step protections, or expensive catalysts that drive up the cost of production. The inability to easily tune the solubility profile without compromising thermal performance has been a persistent bottleneck for material scientists seeking to expand the utility of high-performance thermoplastics in diverse industrial applications.
The Novel Approach
In contrast, the methodology described in patent CN109851563B introduces a paradigm shift by utilizing a naphthalenone-based scaffold that inherently disrupts polymer chain packing. The novel approach employs a 'one-pot two-step' strategy for the intermediate DMPPZ, followed by a controlled reduction to the final DHPPZ. This route is characterized by mild reaction conditions and a short synthesis cycle, which directly translates to improved energy efficiency and reduced equipment wear. By incorporating ether linkages through a copper-catalyzed Ullmann coupling, the process effectively increases the ductility and flexibility of the resulting polymer matrix. This strategic modification allows manufacturers to achieve outstanding heat resistance and solubility simultaneously, opening new avenues for application in demanding environments where traditional materials fail to perform adequately.
Mechanistic Insights into Cu-Catalyzed Ullmann Coupling and Lewis Acid Reduction
The core of this synthetic innovation lies in the precise orchestration of two distinct chemical transformations. The first step involves a C-O and C-N Ullmann coupling reaction, utilizing CuI as the catalyst and 1,10-phenanthroline as a bidentate ligand. This catalytic system is crucial for facilitating the nucleophilic substitution between the phenolic hydroxyl groups of the DHPZ precursor and the aryl bromide (BPM). The use of potassium carbonate as a base ensures effective deprotonation, while the specific ligand environment stabilizes the copper center, promoting high turnover and selectivity. The reaction proceeds through a gradient temperature profile, starting from room temperature and ramping up to 140-160°C, which helps manage exothermicity and minimize side reactions. This careful control is vital for maintaining the integrity of the sensitive naphthalenone core during the bond-forming event.
Following the formation of the dimethoxy intermediate (DMPPZ), the second step employs a Lewis acid-mediated reduction to unveil the phenolic hydroxyl groups. Aluminum chloride (AlCl3) acts as the primary demethylating agent in the presence of potassium iodide (KI) and acetonitrile. This reagent combination is highly effective for cleaving aryl methyl ethers under relatively mild thermal conditions (60-90°C). The mechanism likely involves the coordination of aluminum to the ether oxygen, followed by nucleophilic attack by iodide, ultimately regenerating the phenol functionality essential for subsequent polymerization. This step is particularly noteworthy because it avoids the use of hazardous boron tribromide or strong mineral acids at high temperatures, thereby enhancing the safety profile of the overall process.
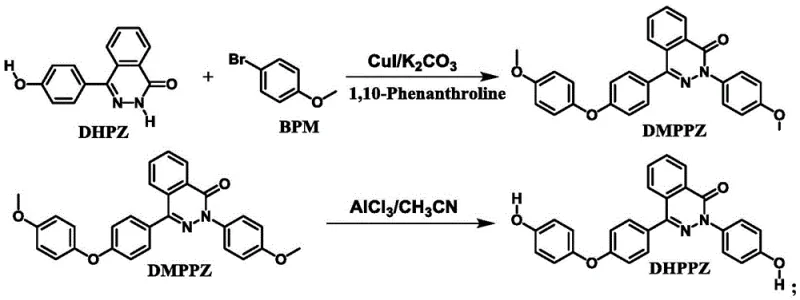
The integration of these two steps results in a streamlined workflow that minimizes purification burdens. The patent data indicates that the target monomer can be achieved with a purity of 98% and yields ranging from 85% to 90%, which is exceptional for such a complex heterocyclic structure. For technical teams, understanding this mechanism is key to troubleshooting potential scale-up issues, such as catalyst loading optimization or solvent selection. The robustness of the CuI/phenanthroline system suggests that it can tolerate minor variations in feedstock quality, a desirable trait for industrial manufacturing. Moreover, the specificity of the AlCl3 reduction ensures that the carbonyl group on the naphthalenone ring remains intact, preserving the electronic properties required for the monomer's intended function in high-performance resins.
How to Synthesize DHPPZ Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent to ensure reproducibility and safety. The process begins with the salt formation of DHPZ, followed by the addition of the coupling partners under inert atmosphere to prevent catalyst oxidation. Detailed operational parameters, including the specific molar ratios of DHPZ to BPM (1:2.5-3.5) and the precise timing for temperature ramps, are critical for maximizing the yield of the DMPPZ intermediate. Subsequent reduction steps demand careful handling of Lewis acids and moisture-sensitive reagents, although the patent notes that rigorous solvent dehydration is not a prerequisite, simplifying the setup. For a comprehensive guide on executing this protocol in a GMP-compliant environment, please refer to the standardized operating procedures below.
- Perform a one-pot salt formation and Ullmann coupling reaction using DHPZ, K2CO3, BPM, CuI catalyst, and 1,10-phenanthroline ligand at 130-160°C to obtain DMPPZ intermediate.
- Purify the DMPPZ intermediate via recrystallization in DMAc solvent to ensure high purity before the reduction step.
- Execute Lewis acid reduction using AlCl3 and KI in acetonitrile at 60-90°C to convert DMPPZ into the final DHPPZ bisphenol monomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain directors focused on cost reduction in advanced material manufacturing. The elimination of solvent dehydration pretreatment is a major operational win, as it removes the need for energy-intensive distillation columns or molecular sieve beds prior to reaction. This simplification not only lowers utility costs but also reduces the turnaround time between batches, allowing for higher throughput in existing reactor trains. Furthermore, the raw materials utilized, such as p-bromoanisole and potassium carbonate, are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that often plague specialty chemical production. This reliability ensures a consistent flow of goods to downstream polymer manufacturers.
- Cost Reduction in Manufacturing: The process design inherently drives down production expenses by utilizing a mature and cost-effective catalytic system. The use of copper iodide, rather than precious metals like palladium or platinum, significantly lowers the catalyst cost per kilogram of product. Additionally, the high yield and purity reported in the patent reduce the burden on downstream purification units, such as chromatography or extensive recrystallization cycles, which are typically the most expensive parts of fine chemical processing. By minimizing waste generation and maximizing atom economy, the overall cost of goods sold (COGS) is optimized, making the final DHPPZ monomer more competitive in the marketplace against alternative high-performance intermediates.
- Enhanced Supply Chain Reliability: Securing a reliable polymer intermediate supplier is critical for maintaining continuous production lines in the electronics and aerospace sectors. This synthesis route enhances supply security by relying on widely available reagents and avoiding proprietary or restricted catalysts that might be subject to export controls or geopolitical volatility. The mild reaction conditions also imply lower stress on reactor vessels and ancillary equipment, leading to reduced maintenance downtime and longer asset life. For supply chain heads, this translates to a more predictable delivery schedule and the ability to scale orders rapidly in response to market demand without encountering technical bottlenecks associated with hazardous or unstable chemistries.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify, the environmental profile of a chemical process becomes a key decision factor. This method demonstrates excellent scalability potential due to its straightforward workup procedures, which involve simple precipitation and filtration rather than complex extractions. The avoidance of harsh demethylating agents like boron tribromide reduces the generation of corrosive waste streams, simplifying effluent treatment and ensuring compliance with stringent environmental standards. Consequently, manufacturers can expand capacity from pilot scale to commercial tonnage with greater confidence, knowing that the process aligns with modern green chemistry principles and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the DHPPZ monomer. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains. Understanding these nuances is essential for making informed decisions about material selection and vendor qualification.
Q: What are the key advantages of the DHPPZ monomer structure?
A: The DHPPZ monomer features a twisted, non-coplanar naphthalenone structure that significantly enhances the solubility and heat resistance of resulting polyarylene ether materials compared to traditional semi-crystalline polymers.
Q: Does this synthesis require expensive anhydrous solvents?
A: No, a major benefit of this patented process is that it does not require dehydration pretreatment of solvents, which drastically simplifies the operational procedure and reduces raw material costs.
Q: What is the reported purity and yield of the target monomer?
A: According to the patent data, the target DHPPZ monomer achieves a purity of 98% as tested by LC-MS, with a reaction yield ranging between 85% and 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DHPPZ Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the development of next-generation polymeric materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global clients. We are committed to delivering DHPPZ and related advanced intermediates with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex heterocyclic syntheses, such as the naphthalenone derivatives described in CN109851563B, positions us as a strategic partner for companies seeking to innovate in the fields of high-performance engineering plastics and specialty coatings.
We invite you to collaborate with us to optimize your material supply chain and accelerate your product development timelines. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can add value to your operations.
