Scalable Manufacturing of High-Purity l-Betaxolol Hydrochloride via Novel Chiral Synthesis
Scalable Manufacturing of High-Purity l-Betaxolol Hydrochloride via Novel Chiral Synthesis
The pharmaceutical landscape for ophthalmic treatments demands rigorous adherence to purity standards and process safety, particularly for beta-blockers like l-betaxolol hydrochloride used in glaucoma management. Patent CN101665441B introduces a transformative synthetic methodology that addresses critical bottlenecks in traditional manufacturing, offering a pathway to enantiomerically pure products with exceptional chemical integrity. This technical insight analyzes the proprietary five-step sequence that begins with p-hydroxyphenylethanol, strategically employing selective tosylation to differentiate hydroxyl reactivity without resorting to hazardous organometallic reagents. By circumventing the use of unstable bromotoluene cyclopropane and pyrophoric zinc species, this innovation not only enhances operator safety but also streamlines the purification workflow, resulting in a total yield that supports robust commercial viability. For procurement and supply chain leaders, understanding the mechanistic superiority of this route is essential for securing a reliable l-betaxolol hydrochloride supplier capable of meeting stringent regulatory specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for levobetaxolol have been plagued by significant operational hazards and efficiency losses that hinder cost reduction in ophthalmic API manufacturing. Prior art, such as the method described in US Patent 6989465, relies heavily on diethylzinc and methylene iodide for key cyclization steps, reagents that are not only prohibitively expensive but also pose severe combustion risks upon contact with air or moisture. Furthermore, alternative pathways involving benzyl protection groups necessitate high-pressure catalytic hydrogenation using palladium on carbon, a process that introduces complex filtration challenges and potential heavy metal contamination risks requiring extensive downstream purification. These conventional methodologies often suffer from low first-step yields, sometimes as low as 48%, and require prolonged reaction times under nitrogen protection, drastically inflating production cycles and energy consumption. The reliance on unstable intermediates like bromotoluene cyclopropane further compromises supply chain continuity, as these materials degrade rapidly and exhibit high irritancy, complicating logistics and storage.
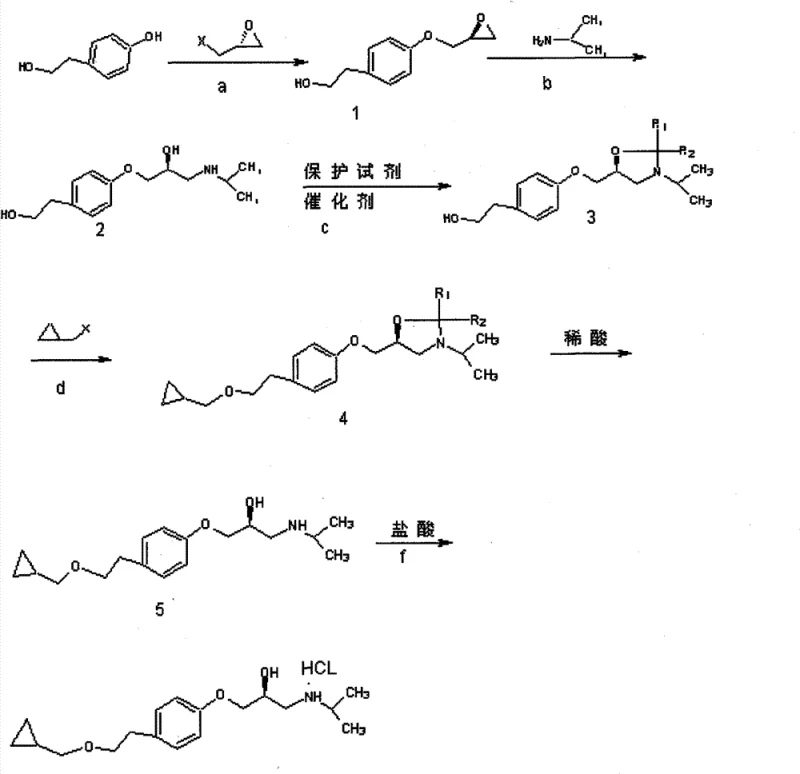
The Novel Approach
In stark contrast, the novel approach delineated in CN101665441B leverages a chemically elegant strategy that prioritizes selectivity and stability at every transformation stage. By initiating the synthesis with the selective esterification of the aliphatic hydroxyl group using tosyl chloride, the process effectively masks the primary alcohol while leaving the phenolic hydroxyl available for subsequent etherification, thereby eliminating the need for bulky benzyl protecting groups. This strategic differentiation allows for the direct coupling of R-epoxyhalopropane under mild basic conditions, preserving the chiral integrity of the epoxide ring which is critical for the final biological activity of the drug. The subsequent displacement of the tosylate with cyclopropylmethanol proceeds smoothly without the need for cryogenic temperatures or exotic catalysts, significantly simplifying the reactor requirements. This streamlined sequence not only reduces the total number of unit operations but also ensures that intermediates are solid or easily crystallizable oils, facilitating superior impurity profiling and consistent batch-to-batch quality.
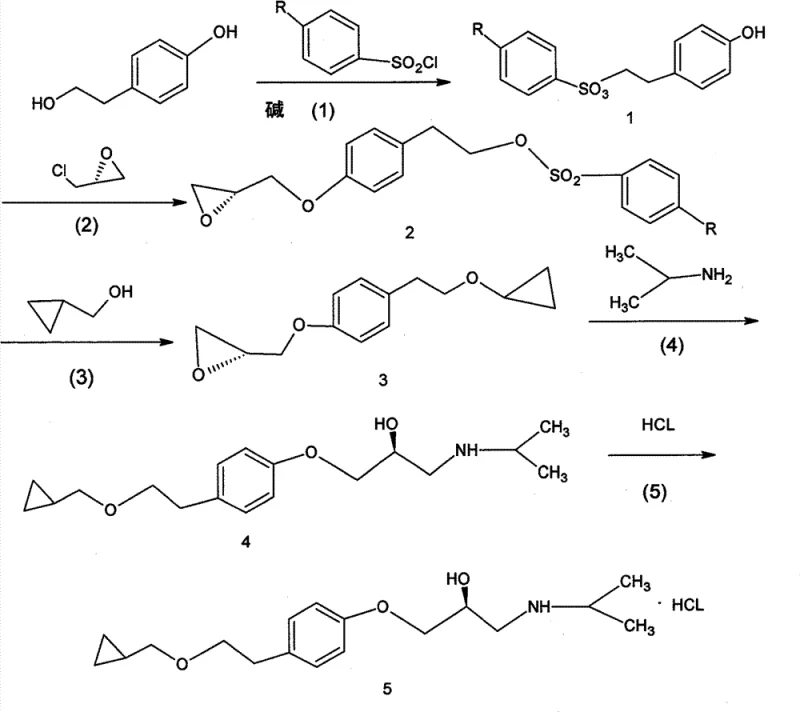
Mechanistic Insights into Selective Tosylation and Epoxide Coupling
The core of this technological advancement lies in the precise control of regioselectivity during the initial functionalization of p-hydroxyphenylethanol. In the first critical step, the reaction with tosyl chloride is conducted at controlled temperatures ranging from -15°C to 60°C, ensuring that the sulfonyl group attaches exclusively to the aliphatic hydroxyl rather than the phenolic oxygen. This selectivity is paramount because it generates a stable leaving group on the side chain while maintaining the nucleophilicity of the phenol for the subsequent attack on the epoxide ring. The formation of the key intermediate, (2S)-3-(4-tolysulfonyloxy ethyl phenoxy)-1,2-propylene oxide, represents a pivotal junction where the stereochemical fate of the molecule is secured. By utilizing R-epoxyhalopropane in the presence of mild bases such as potassium carbonate or sodium alkoxides, the process avoids the racemization pitfalls associated with high-temperature heating seen in older patents. The resulting epoxy intermediate is remarkably stable, allowing for isolation and rigorous quality control before proceeding to the ring-opening steps, thus acting as a crucial checkpoint for maintaining the >99.0% optical purity required for clinical efficacy.
Furthermore, the impurity control mechanism inherent in this design significantly reduces the burden on downstream purification systems. Traditional routes often generate complex byproduct mixtures due to non-selective alkylation or decomposition of unstable cyclopropyl species, necessitating multiple chromatographic separations that are impractical at scale. In this novel pathway, the use of cyclopropylmethanol sodium salt for the nucleophilic substitution ensures a clean displacement of the tosylate group, minimizing the formation of elimination byproducts. The final ring-opening with isopropylamine is executed under reflux conditions that are gentle enough to prevent epimerization yet vigorous enough to drive the reaction to completion with yields exceeding 90% in individual steps. This high level of chemical fidelity means that the final salification with hydrochloric acid yields a product with chemical purity exceeding 99.5%, drastically reducing the need for energy-intensive recrystallization cycles and solvent recovery operations.
How to Synthesize l-Betaxolol Hydrochloride Efficiently
Implementing this synthesis route requires a disciplined approach to reaction parameter control, specifically regarding temperature modulation and stoichiometric precision during the tosylation and etherification phases. The process begins with the dissolution of p-hydroxyphenylethanol in a suitable organic solvent such as pyridine or dichloromethane, followed by the slow addition of tosyl chloride to manage exothermicity and ensure complete conversion to the monotosylate. Detailed standardized operating procedures for each reaction stage, including workup protocols involving aqueous washing and vacuum distillation, are critical to maintaining the high purity profile documented in the patent embodiments. Operators must adhere strictly to the specified molar ratios, particularly the 1:1 to 1:5 ratio between the tosylate intermediate and the epoxide reagent, to prevent oligomerization. The following guide outlines the critical process parameters derived from the patent data to assist technical teams in replicating this high-efficiency pathway.
- Selective esterification of p-hydroxyphenylethanol with tosyl chloride to protect the aliphatic hydroxyl group.
- Etherification of the phenolic hydroxyl with R-epoxyhalopropane to form the chiral epoxy intermediate.
- Nucleophilic substitution with cyclopropylmethanol sodium salt followed by isopropylamine ring opening and salification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, adopting this manufacturing protocol offers substantial cost savings and risk mitigation benefits that extend far beyond simple raw material pricing. The elimination of pyrophoric reagents like diethylzinc removes the necessity for specialized handling equipment and inert atmosphere reactors, which translates directly into lower capital expenditure and reduced insurance premiums for production facilities. Moreover, the avoidance of high-pressure hydrogenation steps simplifies the plant infrastructure requirements, allowing for production in standard glass-lined or stainless steel reactors that are more readily available and easier to maintain. This operational simplicity enhances supply chain reliability by reducing the dependency on niche catalyst suppliers and minimizing the risk of production stoppages due to equipment failure or safety incidents. The robust nature of the intermediates also allows for flexible inventory management, as key compounds can be stockpiled without significant degradation, ensuring continuity of supply even during raw material fluctuations.
- Cost Reduction in Manufacturing: The qualitative economic benefits of this route are driven primarily by the simplification of the purification train and the reduction in hazardous waste disposal costs. By avoiding heavy metal catalysts and toxic alkylating agents, the expense associated with wastewater treatment and residual solvent removal is drastically simplified, leading to a leaner cost structure per kilogram of active ingredient. The higher overall yield reported in the patent embodiments implies less raw material waste per unit of output, which compounds into significant savings when scaled to commercial tonnage. Additionally, the use of commodity chemicals like tosyl chloride and potassium carbonate instead of bespoke organometallic reagents stabilizes the bill of materials against market volatility.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials ensures that the production schedule is not held hostage by the lead times of exotic reagents. Unlike methods requiring freshly prepared bromotoluene cyclopropane, which has a short shelf life and high transport hazard classification, the reagents in this process are stable solids or liquids that can be sourced from multiple global vendors. This diversification of the supply base mitigates the risk of single-source bottlenecks and allows procurement managers to negotiate more favorable terms. The ease of intermediate isolation further supports a modular manufacturing approach, where different stages can be outsourced or produced at different sites without compromising quality.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry initiatives, as it minimizes the generation of halogenated waste and avoids the use of ozone-depleting solvents where possible. The absence of heavy metals simplifies the regulatory filing process for drug master files, as residual metal testing becomes less critical, accelerating time-to-market for generic formulations. Scalability is inherently supported by the exothermic nature of the reactions being manageable through standard cooling jackets, removing the need for cryogenic loops that limit batch size in other processes. This makes the technology ideal for commercial scale-up of complex ophthalmic intermediates, ensuring that supply can meet growing global demand for glaucoma medications without environmental compromise.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and validation of this synthesis pathway, drawing directly from the experimental data and comparative analysis provided in the patent documentation. These answers are designed to clarify the operational feasibility and quality advantages for technical directors evaluating this technology for adoption. Understanding these nuances is critical for making informed decisions about process validation and regulatory strategy.
Q: How does this synthesis route improve optical purity compared to conventional methods?
A: By utilizing R-epoxyhalopropane early in the sequence and avoiding high-temperature racemization steps common in older benzyl-protection routes, this method maintains >99.0% optical purity throughout the process.
Q: What safety advantages does this process offer over zinc-mediated cyclization?
A: This protocol eliminates the need for pyrophoric diethylzinc and methylene iodide, significantly reducing operational hazards and removing the requirement for expensive heavy metal scavenging steps.
Q: Is the process suitable for large-scale commercial production?
A: Yes, the avoidance of high-pressure hydrogenation and cryogenic crystallization, combined with robust intermediate isolation, makes this route highly adaptable for multi-ton annual manufacturing capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable l-Betaxolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR instrumentation to verify that every batch meets the >99.0% optical purity benchmark. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated process solutions that integrate seamlessly into your pharmaceutical manufacturing workflow.
We invite you to engage with our technical procurement team to discuss how this optimized route can enhance your product portfolio and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this methodology impacts your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage in the ophthalmic market with a supply chain built on safety, purity, and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
