Advanced Synthetic Route for l-Betaxolol Hydrochloride: Enhancing Purity and Scalability
Advanced Synthetic Route for l-Betaxolol Hydrochloride: Enhancing Purity and Scalability
The pharmaceutical landscape for ophthalmic agents demands rigorous standards for both chemical and optical purity, particularly for beta-blockers like l-betaxolol hydrochloride. Patent CN101665441A introduces a transformative methodology for the preparation of this critical active pharmaceutical ingredient, addressing long-standing inefficiencies in chiral synthesis. This innovation pivots away from hazardous, high-cost reagents towards a streamlined, selective protection strategy that ensures exceptional enantiomeric excess. By utilizing p-hydroxyphenylethanol as a starting material and employing a novel sequence of selective esterification and etherification, the process achieves a total yield of approximately 62% with chemical and optical purities exceeding 99.0%. This technical breakthrough not only mitigates the risks associated with traditional synthesis but also establishes a robust framework for the reliable l-betaxolol hydrochloride supplier seeking to optimize their production lines.
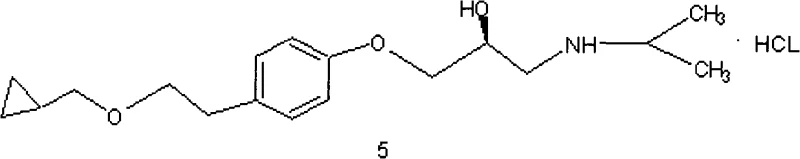
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of l-betaxolol has been plagued by significant operational hazards and stereochemical instability. Prior art, such as U.S. Patent 6989465, relies heavily on the use of zinc ethyl and methylene iodide for cyclization steps. These reagents are notoriously pyrophoric, posing severe combustion risks upon contact with air or moisture, thereby necessitating stringent anhydrous conditions and specialized equipment that drive up capital expenditure. Furthermore, alternative routes disclosed in patents like CN101012175A involve heating chiral intermediates at elevated temperatures (60°C) for prolonged periods, which frequently induces racemization at the chiral center. This loss of optical purity compromises the therapeutic efficacy of the final drug product, requiring costly and energy-intensive recrystallization processes to restore enantiomeric purity. Additionally, the reliance on unstable intermediates like bromoethyl cyclopropane introduces supply chain volatility and handling difficulties due to its high irritancy and pungency.
The Novel Approach
In stark contrast, the methodology outlined in CN101665441A offers a paradigm shift by implementing a selective protection-deprotection strategy that inherently safeguards the chiral integrity of the molecule. The core innovation lies in the initial selective tosylation of the aliphatic hydroxyl group of p-hydroxyphenylethanol, which effectively masks this site while leaving the phenolic hydroxyl available for subsequent etherification. This strategic maneuvering allows for the introduction of the chiral epoxide moiety under mild basic conditions without risking the stability of the alcohol chain. The subsequent steps involve the displacement of the tosyl group with cyclopropylmethanol, a reaction that proceeds with high regioselectivity and yield. By avoiding high-temperature racemization triggers and eliminating the need for dangerous organometallic reagents, this novel approach drastically simplifies the manufacturing workflow. The result is a process that is not only safer and more environmentally benign but also delivers a final product with superior purity profiles suitable for direct pharmaceutical formulation.
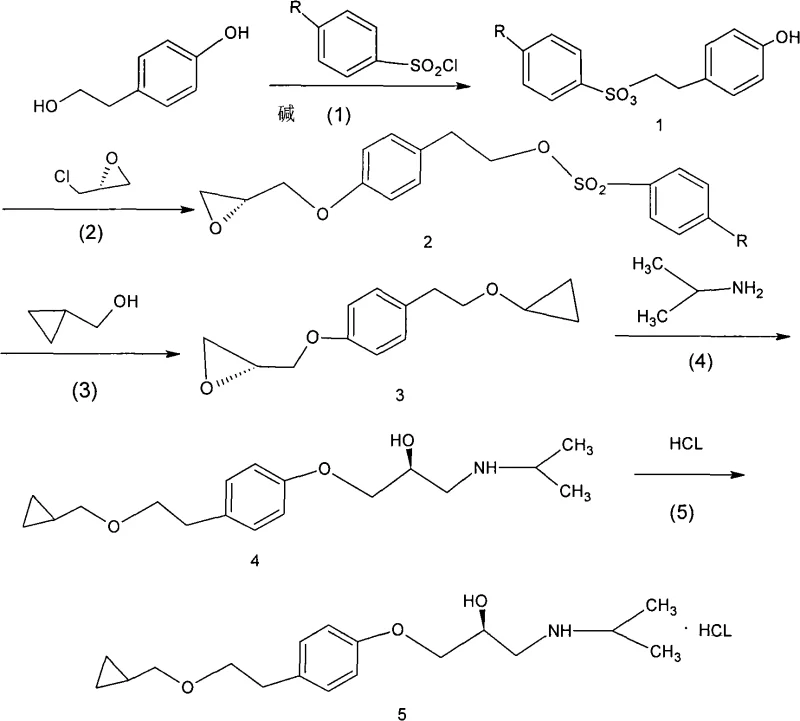
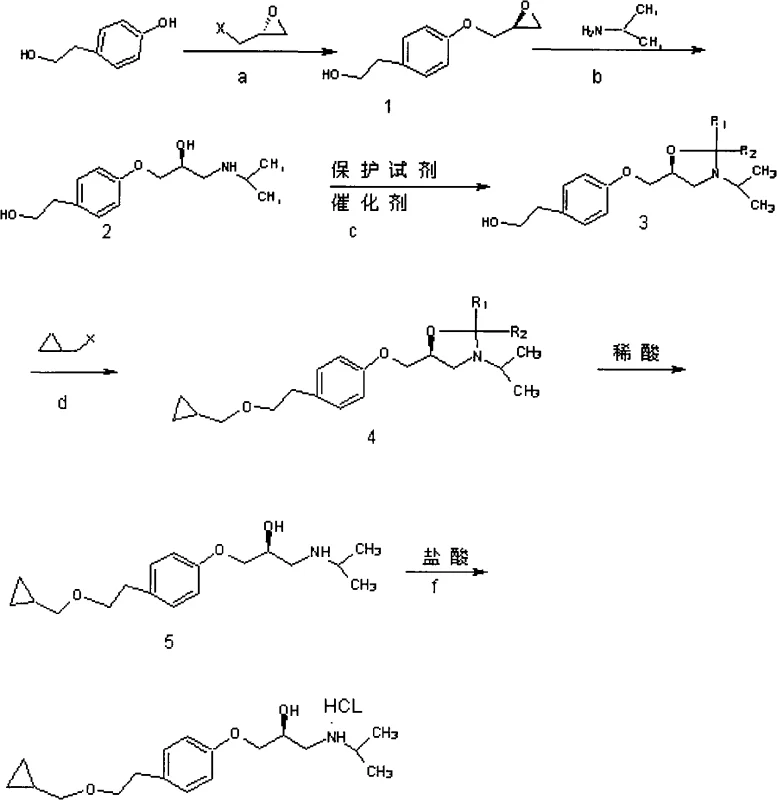
Mechanistic Insights into Selective Tosylation and Epoxide Ring Opening
The success of this synthetic route hinges on the precise control of chemoselectivity during the initial functionalization of p-hydroxyphenylethanol. In the first step, the reaction with tosyl chloride is conducted in pyridine at controlled temperatures ranging from -15°C to 60°C. Under these conditions, the aliphatic hydroxyl group exhibits higher nucleophilicity compared to the phenolic hydroxyl, leading to the preferential formation of the tosylate ester. This temporary protecting group serves a dual purpose: it prevents unwanted side reactions at the aliphatic position during the subsequent phenolic etherification and acts as a superior leaving group for the final installation of the cyclopropylmethoxy side chain. The use of R-epoxyhalopropane in the second step allows for the construction of the chiral backbone via an SN2 mechanism, where the phenoxide ion attacks the less hindered carbon of the epoxide ring. This step is critical as it sets the stereochemistry early in the synthesis, and the mild reaction conditions ensure that the newly formed chiral center remains intact without epimerization.
Following the formation of the chiral epoxide intermediate, the synthesis proceeds through a nucleophilic substitution that defines the final structural architecture of the drug. The tosylate group is displaced by the sodium salt of cyclopropylmethanol, a reaction that benefits from the excellent leaving group ability of the tosyl moiety. This transformation occurs efficiently in polar aprotic solvents like DMF or acetonitrile, facilitating the SN2 attack by the alkoxide nucleophile. The final amination step involves the ring-opening of the epoxide by isopropylamine. This reaction is highly regioselective, attacking the terminal carbon of the epoxide to generate the secondary amine functionality characteristic of beta-blockers. The mechanistic elegance of this pathway lies in its linearity and the avoidance of branching side reactions; each step builds directly upon the previous one with minimal byproduct formation. This clean reaction profile significantly reduces the burden on downstream purification units, allowing for simpler isolation techniques such as cooling crystallization rather than complex chromatographic separations.
How to Synthesize l-Betaxolol Hydrochloride Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize the benefits of the selective protection strategy. The process begins with the dissolution of p-hydroxyphenylethanol in pyridine, followed by the slow addition of tosyl chloride to manage exotherms and ensure mono-substitution. Subsequent steps involve standard workup procedures including aqueous washing and solvent removal under reduced pressure, which are easily adaptable to existing pilot plant infrastructure. The key to success lies in maintaining the integrity of the chiral epoxide intermediate during the etherification with cyclopropylmethanol sodium; strict temperature control between -5°C and 30°C is recommended to prevent thermal degradation. For detailed operational parameters, stoichiometric ratios, and specific workup protocols, please refer to the standardized synthesis guide below which outlines the exact procedural steps derived from the patent embodiments.
- Selective esterification of the aliphatic hydroxyl group of p-hydroxyphenylethanol using tosyl chloride to form the protected intermediate.
- Etherification of the phenolic hydroxyl group with R-epoxyhalopropane under basic conditions to introduce the chiral epoxide moiety.
- Nucleophilic ring opening of the epoxide with cyclopropylmethanol sodium salt, followed by amination with isopropylamine and final salification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By transitioning away from exotic and hazardous reagents like zinc ethyl and bromoethyl cyclopropane, the process significantly reduces the cost of goods sold (COGS) associated with raw material procurement and hazardous waste disposal. The elimination of pyrophoric materials removes the need for specialized inert atmosphere reactors and extensive safety monitoring systems, thereby lowering capital investment requirements for new production lines. Furthermore, the use of stable, commodity-grade starting materials ensures a consistent supply stream, mitigating the risk of production stoppages due to raw material shortages. The high yield and purity achieved in this process also mean that less material is wasted during purification, leading to substantial cost savings in solvent usage and energy consumption for distillation and crystallization steps.
- Cost Reduction in Manufacturing: The replacement of expensive, unstable reagents with cost-effective alternatives like tosyl chloride and cyclopropylmethanol drives down direct material costs. Additionally, the simplified purification workflow, which avoids energy-intensive freezing crystallization and high-pressure hydrogenation, results in significant operational expenditure reductions. The high overall yield minimizes the amount of starting material required per kilogram of final API, further enhancing economic efficiency.
- Enhanced Supply Chain Reliability: Sourcing stable intermediates reduces the vulnerability of the supply chain to market fluctuations and regulatory restrictions on hazardous chemicals. The robustness of the synthetic route allows for flexible manufacturing scheduling, as the intermediates possess sufficient stability for short-term storage if necessary. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The avoidance of heavy metal catalysts and toxic solvents aligns with increasingly stringent environmental regulations, reducing the compliance burden and potential liability for manufacturers. The generation of less hazardous waste simplifies effluent treatment processes, contributing to a greener manufacturing footprint and improved corporate sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this technology. Understanding these nuances is essential for assessing the feasibility of adopting this method for commercial scale-up of complex pharmaceutical intermediates.
Q: How does this synthesis method prevent racemization compared to prior art?
A: Unlike previous methods that heat chiral centers at 60°C for extended periods, this route utilizes a selective tosylation strategy at lower temperatures (-15°C to 60°C), preserving optical purity above 99.0%.
Q: What are the safety advantages of this process over traditional routes?
A: This method eliminates the use of pyrophoric reagents like zinc ethyl and unstable, irritant compounds like bromoethyl cyclopropane, significantly improving operational safety and reducing hazard management costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses stable, commercially available raw materials and avoids complex purification steps like freezing crystallization or high-pressure hydrogenation, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable l-Betaxolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like CN101665441A to maintain competitiveness in the global ophthalmic API market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity l-betaxolol hydrochloride that meets stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our expertise in chiral synthesis and selective protection strategies allows us to consistently produce APIs with superior optical purity, catering to the exacting demands of modern pharmaceutical formulations.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By partnering with our technical procurement team, you can gain access to specific COA data and comprehensive route feasibility assessments that demonstrate the tangible benefits of this innovative synthesis. Let us help you optimize your supply chain and reduce lead time for high-purity pharmaceutical intermediates, ensuring your projects remain on schedule and within budget.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
