Scalable Manufacturing of 5-N-Formalardeemin: A High-Yield Route for MDR Reversal Agents
Scalable Manufacturing of 5-N-Formalardeemin: A High-Yield Route for MDR Reversal Agents
The pharmaceutical industry faces a persistent challenge in the form of multidrug resistance (MDR), a phenomenon where tumor cells develop immunity to chemotherapeutic agents, leading to treatment failure in over 90% of cancer-related deaths. Addressing this critical issue requires potent MDR reversal agents, among which 5-N-formalardeemin has emerged as a star molecule with superior efficacy compared to traditional reversers like verapamil. Patent CN102276615B discloses a groundbreaking method for preparing 5-N-formalardeemin that overcomes the limitations of natural extraction and previous synthetic routes. This technical insight report analyzes the robust 8-step synthesis detailed in the patent, highlighting its potential for reliable supply chain integration and cost-effective manufacturing of this high-purity pharmaceutical intermediate.
The significance of this technology lies in its ability to bypass the bottlenecks associated with isolating ardeemin derivatives from fungal fermented filtrates, a process historically plagued by low yields and high complexity. By establishing a concise chemical synthesis pathway, the patent enables the mass production necessary for extensive pharmacodynamic research and eventual clinical application. For R&D directors and procurement managers, understanding this route is essential for securing a stable supply of this critical oncology intermediate. The following analysis dissects the chemical innovation, operational feasibility, and commercial advantages inherent in this novel manufacturing process.
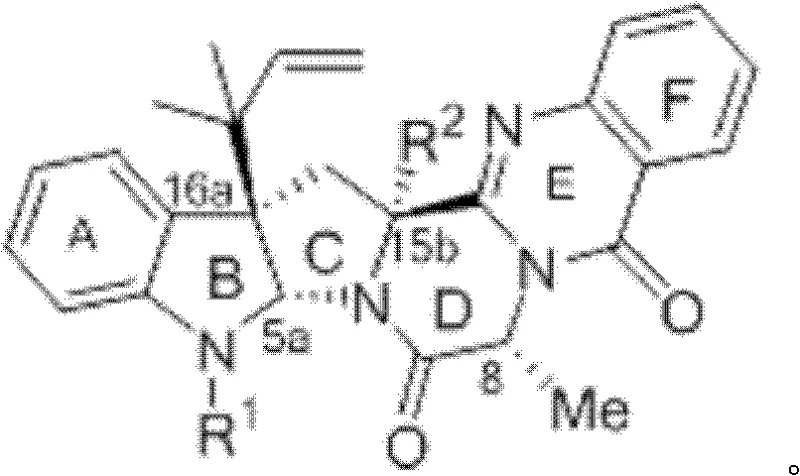
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of ardeemin derivatives relied heavily on extraction from the fungus Aspergillus fischerii, a method fraught with inefficiency and supply chain volatility. The separation of acetyl ardeemin from fungal mycelium is technically demanding, requiring complex purification protocols that result in disappointingly low productive rates. Furthermore, semi-synthetic approaches starting from naturally extracted precursors are constrained by the scarcity of the starting material, creating a fundamental bottleneck for large-scale drug development. Previous total synthesis methods, such as those reported by Qin et al., while scientifically valid, often involve tedious multi-step sequences with cumulative yield losses that render them economically unviable for industrial adoption. These conventional pathways fail to meet the urgent demand for kilogram-to-ton quantities required for preclinical and clinical trials.
The Novel Approach
The patented method introduces a streamlined, linear synthetic strategy that drastically simplifies the construction of the complex pentacyclic skeleton of 5-N-formalardeemin. As illustrated in the comprehensive reaction scheme below, the route proceeds through eight distinct but high-yielding transformations, starting from a readily accessible bromo-indoline derivative. This approach eliminates the dependency on biological fermentation, ensuring a consistent and controllable supply of the active pharmaceutical ingredient (API) intermediate. The strategic design of the synthesis allows for the late-stage installation of the critical formyl group, minimizing side reactions and maximizing overall throughput.
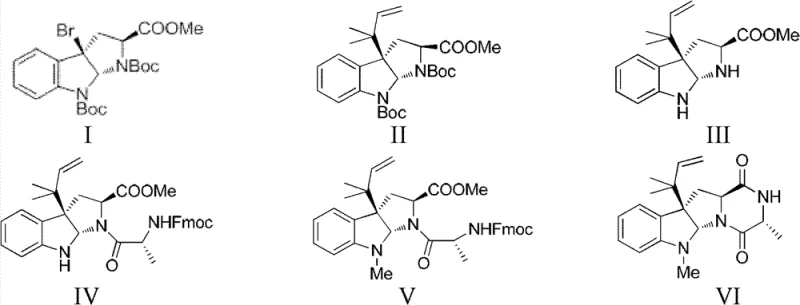
By utilizing robust chemical transformations such as silver-mediated coupling and selective oxidation, the new method achieves a total recovery rate exceeding 30%, a substantial improvement over prior art. This efficiency translates directly into reduced waste generation and lower raw material consumption, addressing both economic and environmental concerns. For supply chain heads, this shift from extraction to synthesis represents a move towards a more predictable and scalable manufacturing model, reducing lead times and mitigating the risks associated with biological variability.
Mechanistic Insights into Silver-Mediated Coupling and Selective Oxidation
The cornerstone of this synthetic success is the initial construction of the quaternary carbon center and the allyl side chain via a silver-promoted substitution reaction. In Step 1, the bromo-indoline precursor reacts with tributyl(3-methyl-2-butenyl)tin in the presence of cesium carbonate and a silver salt, such as silver perchlorate. This transformation is mechanistically distinct from standard palladium-catalyzed Stille couplings; the silver species likely acts as a halophile, facilitating the departure of the bromide ion and generating a reactive cationic intermediate that is subsequently trapped by the organotin nucleophile. The use of cesium carbonate as a mild inorganic base ensures compatibility with the sensitive Boc-protected amine functionality, preventing premature deprotection or racemization.
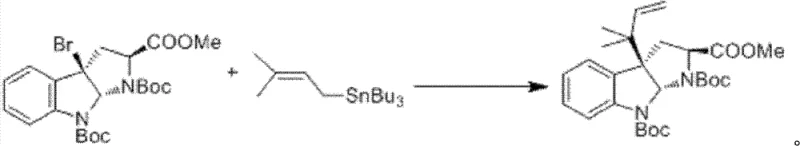
This step is critical for establishing the stereochemistry and structural integrity of the core scaffold. The high yield of 87% reported in the embodiments underscores the efficiency of this silver-mediated protocol. Following this, the synthesis proceeds through a series of functional group manipulations, including Boc deprotection, peptide coupling with Fmoc-alanine, and N-methylation of the indole nitrogen. Each step is optimized to maintain high purity, with the use of reagents like iodotrimethylsilane for deprotection offering a cleaner alternative to harsh acidic conditions that might degrade the sensitive indoline ring system.
The final transformation involves the selective oxidation of the N-methyl group on the quinazolinone core to the N-formyl group, a chemically challenging conversion that defines the biological activity of the molecule. As shown in the reaction scheme for Step 8, the process employs pyridinium chlorochromate (PCC) or activated manganese dioxide as the oxidant. This reaction likely proceeds through the formation of an iminium ion intermediate, which is then hydrolyzed or further oxidized to the stable formamide. The ability to perform this oxidation selectively without affecting the adjacent alkene or amide functionalities demonstrates the high chemoselectivity of the chosen conditions. This precise control over the final oxidation state is vital for ensuring the potency of the final 5-N-formalardeemin product.
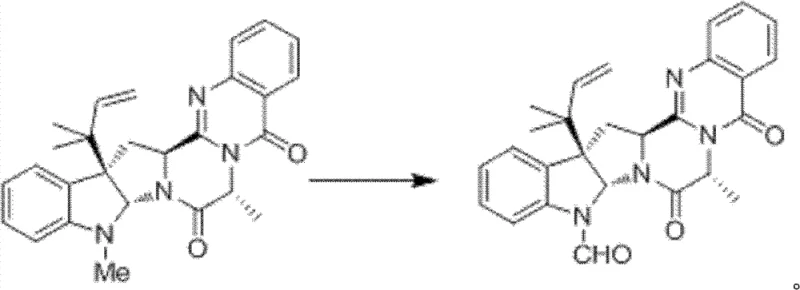
How to Synthesize 5-N-Formalardeemin Efficiently
The execution of this synthesis requires careful attention to reaction conditions, particularly temperature control during the silver-mediated coupling and the final oxidation steps. The process is designed to be operationally simple, utilizing common organic solvents like dichloromethane and tetrahydrofuran, which facilitates easy solvent recovery and recycling in a plant setting. The standardized protocol outlined in the patent provides a clear roadmap for process chemists to replicate the high yields observed in the laboratory examples. Detailed standard operating procedures for each transformation, including specific molar ratios and workup techniques, are essential for maintaining batch-to-batch consistency.
- Perform silver-mediated allylation of the bromo-indoline precursor using tributyl(3-methyl-2-butenyl)tin and cesium carbonate to form the protected intermediate.
- Execute Boc deprotection followed by peptide coupling with Fmoc-Ala-OH and subsequent N-methylation of the indole nitrogen.
- Complete the synthesis via intramolecular cyclization to form the quinazolinone core, followed by selective oxidation of the N-methyl group to the N-formyl functionality.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this synthetic route offers profound strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of production from biological sources, which inherently stabilizes the supply chain against seasonal or fermentation-based fluctuations. By relying on commodity chemicals and standard reagents, the manufacturing process becomes less susceptible to the volatility often seen in specialized biological extracts. This reliability is crucial for long-term project planning and ensures that clinical trial materials can be produced on demand without lengthy lead times associated with fungal cultivation and extraction.
- Cost Reduction in Manufacturing: The synthetic route significantly lowers the cost of goods sold (COGS) by eliminating the expensive and low-yield extraction processes previously required. The use of inexpensive bases like cesium carbonate and widely available oxidants reduces raw material costs substantially. Furthermore, the high individual step yields (often exceeding 80-90%) minimize the loss of valuable intermediates, thereby reducing the overall material input required per kilogram of final product. This efficiency translates into direct cost savings that can be passed down the supply chain, making the development of MDR reversal therapies more economically viable.
- Enhanced Supply Chain Reliability: The reliance on synthetic chemistry rather than natural extraction ensures a consistent and scalable supply of 5-N-formalardeemin. The starting materials, such as the bromo-indoline derivative and organotin reagents, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base enhances resilience against market disruptions. Additionally, the robustness of the reaction conditions allows for flexible manufacturing scheduling, enabling rapid response to increased demand from downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The avoidance of exotic catalysts and the use of standard organic solvents simplify the engineering requirements for scale-up. From an environmental perspective, the high atom economy of the key coupling steps and the ability to recycle solvents contribute to a greener manufacturing footprint. The reduction in waste generation compared to extraction methods aligns with modern sustainability goals, facilitating easier regulatory approval and compliance with increasingly stringent environmental regulations in chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-N-formalardeemin. These answers are derived directly from the technical specifications and experimental data provided in patent CN102276615B, ensuring accuracy and relevance for industry stakeholders. Understanding these details is key for R&D teams evaluating the feasibility of incorporating this intermediate into their drug discovery pipelines.
Q: What is the total yield of the patented 5-N-formalardeemin synthesis?
A: According to patent CN102276615B, the optimized synthetic route achieves a total yield of over 30%, specifically demonstrated at 32.6% in experimental embodiments, which is significantly higher than previous extraction or synthesis methods.
Q: Why is 5-N-formalardeemin preferred over ardeemin for MDR reversal?
A: Research indicates that 5-N-formalardeemin exhibits stronger inhibitory activity against tumor cells and superior sensitization effects when combined with antitumor drugs like vincristine compared to its acetyl counterpart, making it a more potent candidate for overcoming multidrug resistance.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the process utilizes readily available raw materials and reagents such as cesium carbonate and standard oxidants, avoiding complex natural extraction processes, thereby making it highly suitable for industrial-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-N-Formalardeemin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation oncology therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are committed to delivering 5-N-formalardeemin with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our facility is equipped to handle the specific reagents and conditions required for this silver-mediated synthesis, guaranteeing a reliable supply for your research and development efforts.
We invite you to collaborate with us to optimize this synthesis for your specific requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our manufacturing capabilities can reduce your overall project costs. Please contact us today to request specific COA data and route feasibility assessments, and let us support your mission to overcome multidrug resistance in cancer therapy with a dependable supply of this vital pharmaceutical intermediate.
