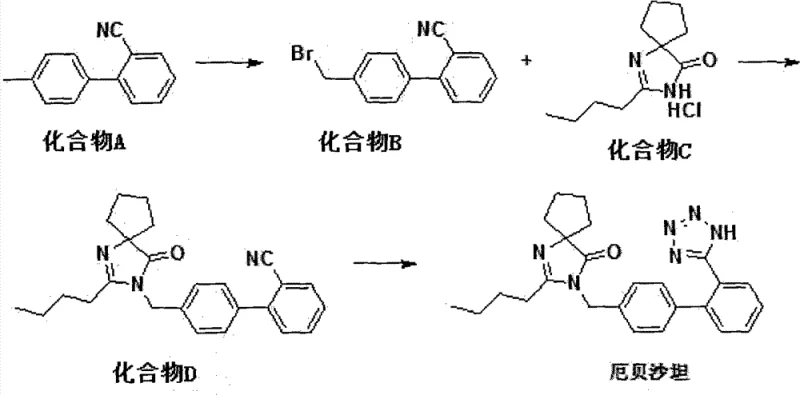
Advanced 3-Step Irbesartan Synthesis: Technical Upgrade for Commercial Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency, safety, and cost-effectiveness, particularly for high-volume antihypertensive agents like Irbesartan. Patent CN102898420A introduces a transformative three-step synthetic route that addresses critical bottlenecks found in legacy manufacturing processes. By leveraging a unique inorganic salt oxidation system for the initial bromination and a zinc-catalyzed tetrazole formation, this methodology eliminates the reliance on hazardous organotin reagents and expensive palladium catalysts. For R&D directors and supply chain managers, this represents a significant opportunity to streamline production while adhering to stringent environmental regulations. The process utilizes readily available inorganic oxidants and reductants, specifically sodium bromate and sodium dithionite, to generate reactive bromine species in situ, thereby enhancing operational safety and reducing raw material volatility. This technical insight report analyzes the mechanistic advantages and commercial viability of this approach for large-scale API intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Irbesartan has been plagued by significant safety and economic challenges inherent to earlier patented methodologies. For instance, US Patent US5270317 relies heavily on tributyltin azide for the crucial tetrazole ring formation, a reagent known for its high toxicity and the difficulty associated with removing trace tin residues from the final active pharmaceutical ingredient. Furthermore, alternative routes such as those described in US5629331 utilize expensive aprotic dipolar solvents like N-methylpyrrolidone, which are not only costly but also present substantial difficulties in solvent recovery and waste treatment due to their high boiling points and tendency to emulsify. Other approaches involving Suzuki coupling, as seen in WO2004065383, introduce expensive palladium catalysts that drastically inflate the cost of goods sold, making them less attractive for generic manufacturing. Additionally, methods employing trityl protecting groups, such as CN1668612, increase the molecular weight of intermediates unnecessarily, leading to lower atom economy and requiring additional hydrolysis steps that reduce overall yield and complicate the purification process.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel route disclosed in CN102898420A offers a streamlined three-step process that maximizes atom economy and minimizes hazardous waste generation. The core innovation lies in the first step, where 2-cyano-4'-methylbiphenyl is converted to the bromomethyl intermediate using a safe, in-situ generated bromine source derived from sodium bromate and sodium dithionite. This eliminates the need for handling elemental bromine or purchasing expensive brominating agents. The subsequent coupling reaction utilizes a biphasic system with tetrabutylammonium bromide as a phase transfer catalyst, ensuring efficient reaction kinetics without the need for exotic solvents. Finally, the tetrazole ring construction employs zinc chloride instead of toxic tin reagents, coupled with a precise pH control strategy that preserves the integrity of the sensitive spiro-cycle. This holistic approach results in a cleaner reaction profile, higher purity intermediates, and a significantly reduced environmental footprint, making it ideal for modern green chemistry standards in pharmaceutical manufacturing.
Mechanistic Insights into Inorganic Salt Oxidation and Zinc-Catalyzed Cyclization
The mechanistic elegance of this synthesis begins with the redox reaction between sodium bromate and sodium dithionite in a dichloromethane-water biphasic system. This interaction generates elemental bromine in situ, which immediately reacts with the methyl group of the biphenyl starting material to form the reactive bromomethyl species. This in-situ generation avoids the accumulation of free bromine, thereby mitigating safety risks associated with exothermic bromination reactions. The use of a phase transfer catalyst in the second step facilitates the nucleophilic substitution between the bromomethyl intermediate and the spiro-ketone hydrochloride salt. The inorganic base, preferably sodium hydroxide, neutralizes the hydrochloride salt to release the free amine, which then attacks the benzylic carbon. This biphasic setup allows for easy separation of the organic product from inorganic byproducts, simplifying the workup procedure and enhancing the purity of the resulting Compound D without the need for complex chromatographic purification.
A critical aspect of this route is the control of impurities during the final tetrazole formation step, particularly the prevention of spiro-ring opening. Under strongly alkaline conditions, the five-membered spiro-ring is susceptible to hydrolysis, leading to the formation of Impurity A, which compromises the quality of the final API. 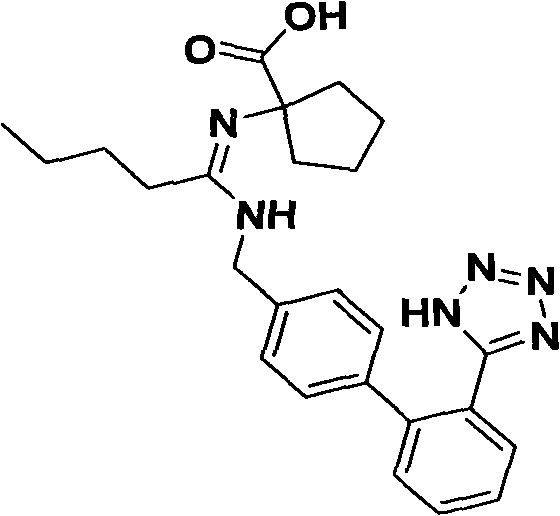 The patent discloses that using zinc chloride as a Lewis acid catalyst moderates the reactivity of the azide ion, allowing the cyclization to proceed under milder conditions. Furthermore, the workup procedure involves a careful pH adjustment sequence where the organic phase is acidified to extract the product into the aqueous phase, followed by neutralization to precipitate the pure Irbesartan. This specific pH control prevents the exposure of the sensitive spiro-lactam structure to harsh alkaline environments for prolonged periods, effectively suppressing the formation of ring-opened byproducts and ensuring the final product meets stringent purity specifications required for regulatory approval.
The patent discloses that using zinc chloride as a Lewis acid catalyst moderates the reactivity of the azide ion, allowing the cyclization to proceed under milder conditions. Furthermore, the workup procedure involves a careful pH adjustment sequence where the organic phase is acidified to extract the product into the aqueous phase, followed by neutralization to precipitate the pure Irbesartan. This specific pH control prevents the exposure of the sensitive spiro-lactam structure to harsh alkaline environments for prolonged periods, effectively suppressing the formation of ring-opened byproducts and ensuring the final product meets stringent purity specifications required for regulatory approval.
How to Synthesize Irbesartan Efficiently
The synthesis of Irbesartan via this patented route is designed for scalability and operational simplicity, making it highly suitable for industrial implementation. The process begins with the preparation of the bromomethyl intermediate in a standard reactor equipped with a dropping funnel for the controlled addition of the reductant solution. Following the isolation of the brominated species, the coupling reaction is performed in a similar biphasic setup, leveraging the efficiency of phase transfer catalysis to drive the reaction to completion within a few hours at ambient temperature. The final cyclization step requires reflux conditions in toluene but benefits from the use of non-toxic zinc salts. Detailed standardized operating procedures, including specific molar ratios and temperature controls optimized through extensive experimentation, are essential to replicate the high yields reported in the patent examples. For a comprehensive guide on the exact parameters and safety protocols, please refer to the structured synthesis instructions below.
- React 2-cyano-4'-methylbiphenyl with sodium bromate and sodium dithionite in a dichloromethane-water biphasic system to generate 2-cyano-4'-bromomethylbiphenyl.
- Couple the brominated intermediate with 2-butyl-1,3-diazaspiro[4.4]nonan-1-en-4-one hydrochloride using tetrabutylammonium bromide and sodium hydroxide.
- Convert the cyano-spiro intermediate to Irbesartan using sodium azide and zinc chloride in toluene, followed by acid-base workup.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial strategic advantages by decoupling production from volatile commodity markets and hazardous material supply chains. The replacement of expensive palladium catalysts and toxic organotin reagents with common inorganic salts like sodium bromate, sodium dithionite, and zinc chloride significantly stabilizes the cost structure of the manufacturing process. These inorganic reagents are globally available in bulk quantities, ensuring a reliable supply chain that is less susceptible to geopolitical disruptions or supplier monopolies often associated with specialized organometallic catalysts. Furthermore, the elimination of high-boiling solvents like NMP reduces the complexity of solvent recovery systems, lowering energy consumption and waste disposal costs. This translates to a more predictable cost of goods sold and improved margin stability for long-term supply contracts.
- Cost Reduction in Manufacturing: The substitution of high-cost reagents with inexpensive inorganic alternatives directly impacts the bottom line by reducing raw material expenses. By avoiding the use of tributyltin azide and palladium catalysts, manufacturers eliminate the need for expensive heavy metal scavenging steps and complex purification protocols, which are both capital and operationally intensive. The simplified workup procedures, characterized by straightforward phase separations and crystallizations, reduce labor hours and utility usage per kilogram of product. Additionally, the high atom economy achieved by avoiding bulky protecting groups like trityl means less waste is generated per unit of product, further driving down disposal costs and enhancing overall process efficiency.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as sodium hydroxide, sodium azide, and zinc chloride ensures that the supply chain remains robust and resilient against market fluctuations. Unlike specialized catalysts that may have limited suppliers and long lead times, these inorganic salts are produced by multiple vendors worldwide, providing procurement teams with greater negotiating power and flexibility. The reduced dependency on hazardous materials also simplifies logistics and storage requirements, as the transportation and handling of non-toxic inorganic salts are subject to fewer regulatory restrictions compared to organotin compounds. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-liter reactors with consistent yields, indicating readiness for ton-scale production. The biphasic reaction systems facilitate heat dissipation and mixing, which are critical factors when scaling up exothermic reactions safely. From an environmental compliance standpoint, the absence of tin residues and the use of recyclable solvents like dichloromethane and toluene align with increasingly strict global environmental regulations. The generation of inorganic salt waste, which is easier to treat and dispose of compared to heavy metal contaminated waste streams, simplifies the permitting process for manufacturing facilities. This eco-friendly profile enhances the corporate sustainability metrics of the supply chain, appealing to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Irbesartan synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, focusing on safety, purity, and operational feasibility. Understanding these nuances is vital for technical teams evaluating the transition from legacy methods to this improved process. The answers highlight the specific mechanisms by which this route overcomes historical challenges in antihypertensive drug manufacturing.
Q: How does this route improve safety compared to traditional tin-based methods?
A: This method replaces toxic tributyltin azide with non-toxic zinc chloride and sodium azide, eliminating heavy metal residues and significantly reducing environmental hazards.
Q: What are the cost advantages of using inorganic salts for bromination?
A: Using sodium bromate and sodium dithionite generates bromine in-situ, avoiding the procurement and handling costs of expensive elemental bromine or specialized brominating reagents.
Q: How is the spiro-ring integrity maintained during tetrazole formation?
A: The use of zinc chloride as a catalyst combined with controlled pH adjustment prevents the alkaline-induced ring-opening of the five-membered spiro structure, ensuring high product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Irbesartan Supplier
The technical potential of this three-step synthesis route underscores the importance of partnering with a CDMO that possesses deep expertise in process optimization and scale-up. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure that the theoretical benefits of this patent are fully realized in a commercial setting. Our facility is equipped with state-of-the-art rigorous QC labs capable of monitoring trace impurities and ensuring stringent purity specifications are met for every batch. We understand that the transition to a new synthetic route requires meticulous validation, and our team is dedicated to managing the complexities of in-situ reagent generation and pH-sensitive crystallization to deliver consistent, high-quality Irbesartan intermediates.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this inorganic salt-based methodology. We encourage you to contact us to obtain specific COA data from our pilot runs and detailed route feasibility assessments tailored to your production volume requirements. Let us collaborate to secure a sustainable and cost-effective supply of high-purity pharmaceutical intermediates for your global market needs.
