Advanced Manufacturing of Rosuvastatin Intermediates: A Chromatography-Free Approach for Global API Supply
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-value cardiovascular therapeutics, and Patent CN101591302A represents a significant technological leap in the synthesis of Rosuvastatin Calcium intermediates. This patent addresses critical bottlenecks found in earlier methodologies, specifically the reliance on labor-intensive and non-scalable column chromatography. By refining the Wittig condensation reaction parameters and revolutionizing the purification strategy through crystallization, this technology enables the production of high-purity heptenoic acid ester derivatives suitable for commercial API manufacturing. The innovation lies not merely in chemical transformation but in the holistic engineering of the process to maximize yield while minimizing waste and operational complexity. For global supply chain stakeholders, this represents a transition from fragile laboratory protocols to rugged industrial chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin intermediates, as disclosed in foundational patents like EP0521471, suffered from severe scalability issues inherent to batch processing with extensive purification requirements. The conventional approach typically necessitated a large excess of the phosphorus ylide reagent, often exceeding 1.5 equivalents relative to the aldehyde substrate, which drove up raw material costs significantly given the high value of the chiral side chain. Furthermore, the reaction solvent loads were excessively high, often greater than ten times the mass of the reagents, leading to inefficient reactor utilization and massive energy consumption during solvent recovery. Most critically, the resulting crude products were often oils or syrups that could not be purified by crystallization, forcing manufacturers to rely on column chromatography. This dependency on silica gel separation is a major barrier to industrialization, as it introduces batch-to-batch variability, limits throughput, and generates substantial solid waste, making it economically unviable for multi-ton production campaigns.
The Novel Approach
The methodology described in CN101591302A fundamentally re-engineers the reaction thermodynamics and kinetics to favor direct isolation of the product. By precisely controlling the stoichiometry of the ylide reagent to a range of 0.5 to 1.5 equivalents, and optimally near 1.0 equivalent, the process minimizes the presence of unreacted starting materials and side products that interfere with crystallization. Simultaneously, the solvent load is drastically reduced to between 0.1 and 5 times the mass of the reagent, which accelerates reaction rates and improves impurity profiles. This strategic adjustment allows the intermediate enone compound to be purified via simple recrystallization, effectively removing triphenylphosphine oxide without chromatography. The subsequent desilylation step is similarly optimized to ensure the final dihydroxy ester precipitates as a high-purity solid, achieving overall yields exceeding 75% over two steps while completely bypassing the need for column separation.
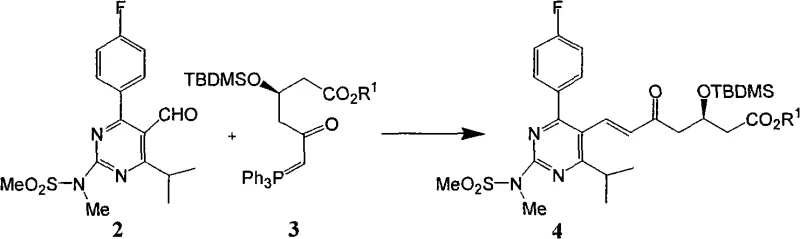
Mechanistic Insights into Optimized Wittig Condensation and Crystallization
The core mechanistic breakthrough in this patent involves the delicate balance between reaction conversion and downstream purification physics. In traditional Wittig reactions, driving the reaction to completion often requires excess ylide, but this patent reveals that residual ylide and its decomposition products act as potent inhibitors of crystallization, trapping the product in an amorphous oil state. By limiting the ylide charge, the process ensures that any remaining aldehyde, which has a higher melting point and better crystallization properties than the ylide residues, does not prevent the product from solidifying. The reduction in solvent volume plays a dual role: it increases the concentration of reactants to accelerate the nucleophilic attack of the ylide on the aldehyde carbonyl, and it creates a supersaturated environment upon cooling that favors the nucleation of the desired product crystal lattice over the precipitation of amorphous impurities. This understanding transforms the purification from a separation problem into a crystallization engineering challenge.
Furthermore, the desilylation step utilizing hydrofluoric acid is meticulously controlled to prevent degradation of the sensitive beta-keto ester moiety while ensuring complete removal of the silyl protecting group. The patent details a specific workup procedure where the reaction mixture is neutralized and subjected to a phase separation that allows for the efficient recovery of acetonitrile, a key cost-saving measure in large-scale operations. The final crystallization from ether or mixed ether solvents is designed to exclude diastereomeric impurities, leveraging the differences in solubility between the target (3R, 5S) configuration and potential stereoisomers. This level of stereochemical control is vital for meeting the stringent regulatory requirements for chiral pharmaceutical intermediates, ensuring that the final API possesses the necessary biological activity without the toxicity associated with incorrect enantiomers.
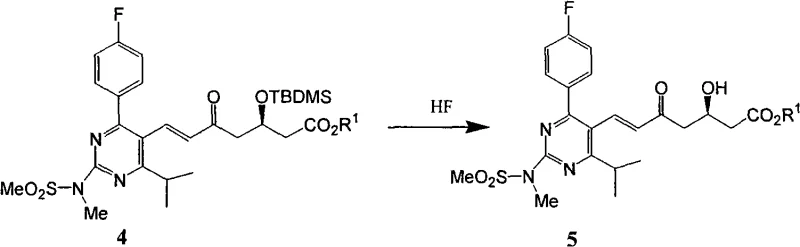
How to Synthesize Rosuvastatin Intermediate Efficiently
The synthesis protocol outlined in this patent provides a clear roadmap for transitioning from laboratory discovery to commercial manufacturing. It emphasizes the importance of strict parameter control, particularly regarding temperature profiles during the exothermic addition of reagents and the cooling ramps during crystallization. The elimination of chromatography simplifies the equipment train, requiring only standard reactors, filters, and dryers, which reduces capital expenditure and maintenance overhead. Detailed standardized synthetic steps see the guide below.
- Perform Wittig condensation between pyrimidine aldehyde and phosphorus ylide using reduced solvent load and optimized stoichiometry to form the enone intermediate.
- Remove triphenylphosphine oxide byproduct via recrystallization rather than chromatographic separation.
- Execute desilylation using hydrofluoric acid followed by controlled crystallization to isolate the high-purity dihydroxy heptenoic acid ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthesis route offers profound strategic advantages beyond simple chemical yield. The primary value driver is the drastic reduction in manufacturing costs achieved through the elimination of expensive consumables like silica gel and the reduction of high-cost chiral reagents. By operating with near-stoichiometric amounts of the ylide reagent, the material cost per kilogram of the intermediate is significantly lowered, directly improving the margin structure for the final API. Additionally, the removal of column chromatography shortens the cycle time per batch, increasing the overall capacity of existing manufacturing facilities without the need for new construction. This efficiency gain translates into a more responsive supply chain capable of meeting fluctuating market demands for cardiovascular medications.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of chromatographic purification, which is notoriously expensive due to the cost of stationary phases and the labor required for packing and running columns. By replacing this with crystallization, the variable costs associated with consumables are virtually eliminated. Furthermore, the optimized stoichiometry reduces the consumption of the complex phosphorus ylide reagent, which is one of the most expensive inputs in the synthesis. The ability to recover and recycle solvents like acetonitrile and hexane further compounds these savings, creating a lean manufacturing process that maximizes resource utilization and minimizes waste disposal costs.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is often compromised by complex purification steps that are prone to failure or variability. The simplified workflow of this patent, which relies on robust unit operations like filtration and distillation, significantly reduces the risk of batch failures. The process is less sensitive to minor fluctuations in raw material quality compared to chromatography-dependent methods, ensuring consistent output. This stability allows for more accurate forecasting and inventory planning, reducing the need for safety stock and minimizing the risk of supply disruptions that could impact the production of the finished Rosuvastatin Calcium drug product.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this technology aligns perfectly with green chemistry principles. The reduction in solvent usage lowers the facility's volatile organic compound (VOC) emissions, simplifying compliance with increasingly strict environmental regulations. The absence of silica gel waste reduces the burden on solid waste management systems. Moreover, the process is inherently scalable; crystallization is a unit operation that scales linearly from pilot plant to multi-ton reactors, unlike chromatography which often requires complex scaling strategies. This makes the technology ideal for long-term commercial production, ensuring a stable supply of high-quality intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific improvements detailed in the patent documentation, focusing on purity, yield, and operational feasibility. Understanding these aspects is crucial for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: How does this patent improve upon the original EP0521471 synthesis route?
A: The patent CN101591302A significantly improves the process by optimizing the stoichiometry of the Wittig reagent and reducing solvent usage. Crucially, it eliminates the need for column chromatography, which is difficult to scale, by enabling direct crystallization of the intermediates.
Q: What is the impact of reducing the ylide reagent excess on product purity?
A: Reducing the excess of the expensive ylide reagent (Formula 3) prevents the formation of oily impurities that hinder crystallization. This allows the final product (Formula 5) to be isolated as a solid with purity exceeding 99% through simple recrystallization.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scale-up. By removing column chromatography and optimizing solvent recovery, the method offers a robust, cost-effective pathway for manufacturing high-quality Rosuvastatin intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex cardiovascular drugs depends on the reliability and quality of the supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101591302A are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality assurance means that we can consistently deliver Rosuvastatin intermediates that meet the exacting standards required by global regulatory agencies, providing our partners with peace of mind and supply security.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these advanced synthesis technologies. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall cost of goods sold.
