Optimized Industrial Synthesis of Rosuvastatin Calcium Intermediates for Global Pharma Supply
Optimized Industrial Synthesis of Rosuvastatin Calcium Intermediates for Global Pharma Supply
The global demand for high-performance lipid-lowering agents continues to drive innovation in the manufacturing of key pharmaceutical intermediates, specifically within the statin class. Patent CN102219749A discloses a robust and highly efficient preparation method for Rosuvastatin Calcium, a blockbuster medication widely prescribed for managing hypercholesterolemia and preventing cardiovascular disease. This technical disclosure represents a significant advancement over prior art by offering a streamlined synthetic pathway that balances high stereoselectivity with operational simplicity. For R&D directors and procurement specialists evaluating supply chain resilience, this methodology offers a compelling alternative to traditional routes that often suffer from complex purification requirements or reliance on prohibitively expensive chiral catalysts. The process leverages a strategic combination of Wittig olefination, boron-mediated stereoselective reduction, and nucleophilic aromatic substitution to construct the complex pyrimidine-heptenoic acid backbone with exceptional precision.
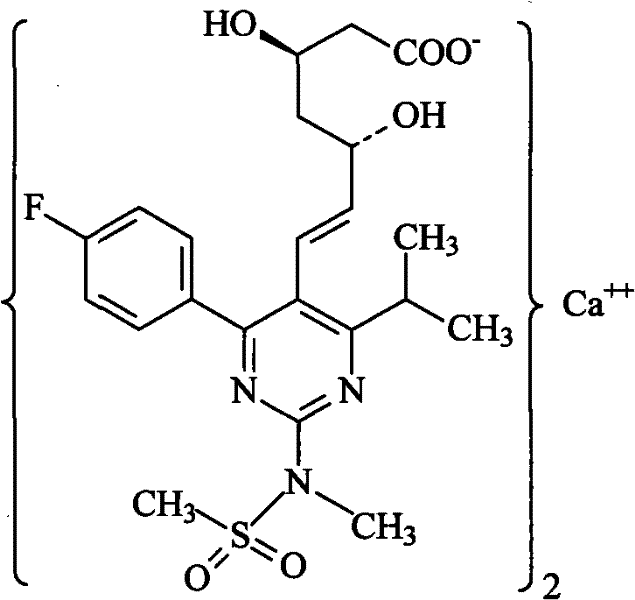
From a commercial manufacturing perspective, the stability of yields and the accessibility of raw materials described in this patent are paramount. The synthesis avoids the use of hazardous reagents where possible and employs solvent systems that are standard in the fine chemical industry, such as acetonitrile, tetrahydrofuran (THF), and dimethyl sulfoxide (DMSO). This alignment with standard industrial practices reduces the barrier to entry for contract development and manufacturing organizations (CDMOs) looking to scale production. Furthermore, the explicit focus on mild reaction conditions for sensitive functional groups, such as the preservation of the E-alkene geometry during reduction steps, ensures that the final active pharmaceutical ingredient (API) meets stringent purity specifications required by regulatory bodies like the FDA and EMA. This report analyzes the technical nuances of this pathway to demonstrate its viability as a cornerstone for reliable Rosuvastatin Calcium supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin intermediates has been plagued by several inefficiencies that impact both cost and throughput. Many legacy processes rely on early-stage introduction of the sulfonamide moiety or utilize coupling reactions that require expensive transition metal catalysts, which subsequently necessitate rigorous and costly heavy metal removal steps to meet safety standards. Conventional routes often struggle with controlling the stereochemistry at the C3 and C5 positions of the heptenoic acid side chain, leading to mixtures of diastereomers that are difficult and wasteful to separate via chromatography on a large scale. Additionally, older methodologies frequently employ harsh acidic or basic conditions that can compromise the integrity of the fluorophenyl-pyrimidine core or lead to isomerization of the critical double bond. These factors cumulatively result in lower overall yields, increased waste generation, and extended production cycles, creating significant bottlenecks for supply chain heads aiming to secure consistent volumes of high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN102219749A introduces a divergent strategy that decouples the construction of the side chain from the final functionalization of the pyrimidine ring, thereby optimizing each transformation independently. A key innovation is the use of a specific chiral phosphorus ylide reagent in a Wittig condensation with the pyrimidine aldehyde, which efficiently establishes the carbon skeleton with the correct E-alkene geometry. Following this, the process employs a sophisticated deprotection and reduction sequence that installs the vital diol functionality with high fidelity. By delaying the introduction of the N-methyl-N-methanesulfonamide group until after the stereochemical centers are established, the route minimizes side reactions and simplifies purification. This logical sequencing allows for the use of cheaper, commodity-grade reagents while maintaining the structural integrity required for biological activity. The result is a process that is not only chemically elegant but also economically superior, offering substantial cost reduction in pharmaceutical intermediates manufacturing through improved atom economy and reduced downstream processing.
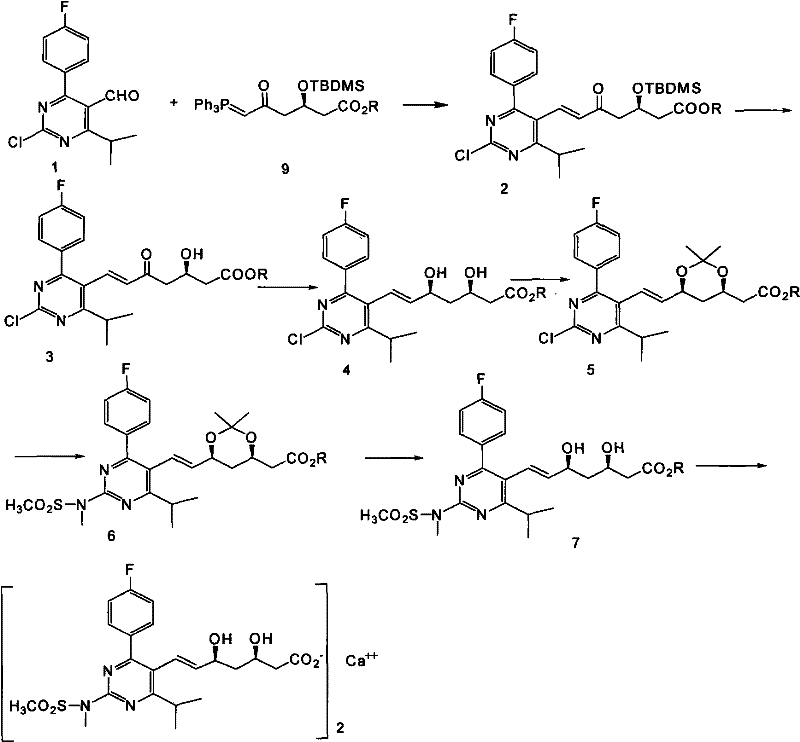
Mechanistic Insights into Boron-Mediated Stereoselective Reduction
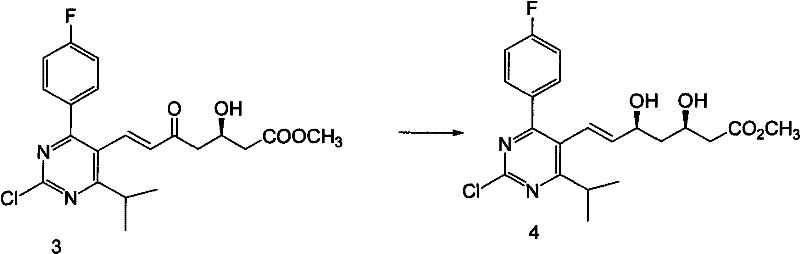
The cornerstone of this synthetic route's success lies in the stereoselective reduction step, which transforms the beta-keto ester intermediate into the crucial syn-diol motif found in the final drug substance. This transformation utilizes an organoboron reagent, specifically diethyl methoxy borane, in conjunction with sodium borohydride at controlled low temperatures ranging from -40°C to -60°C. Mechanistically, the Lewis acidic boron species coordinates with the carbonyl oxygen of the ketone and the adjacent ether oxygen, forming a rigid, cyclic transition state. This chelation effectively shields one face of the carbonyl group, directing the hydride attack from sodium borohydride exclusively from the less hindered face. This precise spatial control ensures the formation of the (3R, 5S) configuration with exceptional diastereomeric excess, effectively eliminating the need for difficult chiral separations later in the process. For R&D teams, understanding this mechanism is vital for troubleshooting and scaling, as strict temperature control is identified as a critical process parameter (CPP) to maintain this high level of stereochemical purity.
Furthermore, the subsequent protection of the newly formed diol as an acetonide serves a dual purpose: it locks the stereochemistry in place and protects the hydroxyl groups during the subsequent nucleophilic substitution on the pyrimidine ring. The substitution reaction itself involves the displacement of a chlorine atom by N-methylmethanesulfonamide in a polar aprotic solvent like DMSO, facilitated by a base such as potassium carbonate. The electron-withdrawing nature of the pyrimidine ring activates the C2 position for nucleophilic attack, allowing this reaction to proceed smoothly at moderate temperatures (80°C to 100°C). The final hydrolysis steps are designed to be orthogonal, removing the acetonide protecting group under acidic conditions and hydrolyzing the methyl ester under basic conditions without affecting the sensitive alkene or the sulfonamide linkage. This orthogonality is a hallmark of a well-designed synthetic route, ensuring that each functional group is manipulated independently without compromising the rest of the molecule.
How to Synthesize Rosuvastatin Calcium Intermediates Efficiently
The synthesis of these complex intermediates requires a disciplined approach to reaction monitoring and workup procedures to ensure maximum yield and purity. The process begins with the condensation of the pyrimidine aldehyde and the phosphorus ylide, followed by a carefully orchestrated sequence of deprotection, reduction, and protection steps. Each stage builds upon the previous one, requiring strict adherence to stoichiometry and temperature profiles to prevent the formation of impurities that could carry through to the final API. The patent details specific solvent choices and molar ratios that have been optimized to balance reaction rate with selectivity. For example, the use of anhydrous conditions during the boron reduction is critical to prevent the decomposition of the reactive borane species. Detailed standardized operating procedures for these steps are essential for technology transfer.
- Perform Wittig condensation between 4-(4-fluorophenyl)-6-isopropyl-2-chloropyrimidine-5-carbaldehyde and a chiral phosphorus ylide reagent in acetonitrile or THF under reflux conditions.
- Execute acidic deprotection of the silyl ether followed by stereoselective reduction using an organoboron reagent and sodium borohydride at low temperatures (-40°C to -60°C) to establish the critical 3R,5S diol configuration.
- Conduct nucleophilic substitution with N-methylmethanesulfonamide in DMSO, followed by acetonide deprotection, ester hydrolysis, and final calcium salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the economic implications of this synthetic route are profound. The shift towards using readily available starting materials and avoiding proprietary or scarce catalysts directly translates to a more resilient supply chain. By relying on bulk chemicals like acetonitrile, THF, and common inorganic salts, manufacturers can mitigate the risk of raw material shortages that often plague the pharmaceutical industry. Moreover, the high yields reported in key steps, such as the 95% yield in the deprotection step and nearly 99% in the reduction step, significantly reduce the amount of starting material required per kilogram of final product. This efficiency lowers the overall cost of goods sold (COGS) and reduces the environmental footprint associated with waste disposal, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity solvents drastically simplify the cost structure of the manufacturing process. Traditional routes often require palladium or other precious metals for cross-coupling reactions, which not only adds direct material costs but also necessitates expensive scavenging resins to remove metal residues to ppm levels. By utilizing a Wittig reaction and nucleophilic substitution instead, this route bypasses those costs entirely. Additionally, the high stereoselectivity of the reduction step minimizes the loss of material to unwanted isomers, ensuring that a greater proportion of the input mass is converted into saleable product, thereby driving down the unit cost significantly.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents enhances the predictability of the supply chain. Reagents like diethyl methoxy borane and sodium borohydride are produced at a global scale and are less susceptible to the geopolitical or logistical disruptions that can affect specialized chiral ligands or enzymes. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and concentrations without significant degradation in performance, further ensures that production schedules can be met consistently. This reliability is crucial for maintaining continuous supply to downstream formulation partners and preventing stockouts of the final medication.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as reflux, extraction, and crystallization that are standard in multi-purpose chemical plants. The avoidance of cryogenic conditions below -60°C (the reaction operates comfortably at -40°C to -60°C) reduces the energy load on cooling systems compared to processes requiring liquid nitrogen temperatures. Furthermore, the aqueous workups and the ability to recycle solvents like THF and DMSO contribute to a lower E-factor (mass of waste per mass of product). This makes the process more attractive from a regulatory and sustainability standpoint, facilitating easier approval of environmental permits for new manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders assess the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: How is stereochemical control achieved in this Rosuvastatin synthesis route?
A: The process utilizes a specialized organoboron-mediated reduction step (using diethyl methoxy borane and sodium borohydride) at controlled low temperatures (-40°C to -60°C). This creates a rigid cyclic transition state that ensures high stereoselectivity for the desired (3R, 5S) diol configuration, minimizing the formation of unwanted diastereomers.
Q: What are the key cost drivers in this manufacturing process?
A: The process relies on commercially available and relatively inexpensive reagents such as acetonitrile, THF, and common inorganic bases like potassium carbonate. By avoiding exotic transition metal catalysts and utilizing high-yielding steps (often exceeding 90% yield in key transformations), the overall cost of goods is significantly optimized compared to routes requiring precious metal catalysis.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial production. The reaction conditions utilize standard solvents and manageable temperature ranges (from cryogenic reduction to moderate reflux), and the workup procedures involve standard extraction and crystallization techniques that are easily transferable from laboratory to multi-ton manufacturing scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in securing the global supply of life-saving medications. Our team of expert chemists has extensively analyzed the methodology presented in CN102219749A and possesses the technical capability to execute this route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity pharmaceutical intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling to ensure compliance with international pharmacopeia standards. We understand that the quality of the intermediate dictates the quality of the final API, and we are committed to delivering excellence at every batch.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. Whether you require a Customized Cost-Saving Analysis comparing this route to your current incumbent process or need specific COA data to validate our capabilities, we are ready to assist. We encourage you to request route feasibility assessments to explore how our manufacturing expertise can enhance your production efficiency and reduce time-to-market for your generic or branded formulations. Let us be your strategic partner in navigating the complexities of fine chemical manufacturing.
