Scalable Synthesis of Rosuvastatin Calcium Intermediates via Optimized Wittig Condensation
The pharmaceutical industry continuously seeks robust manufacturing processes for high-value statins, and patent CN101591302A presents a pivotal advancement in the preparation of heptenoic acid ester derivatives, specifically serving as critical intermediates for Rosuvastatin Calcium. This technology addresses the long-standing bottlenecks associated with the original Shionogi synthetic route disclosed in EP0521471, which, while chemically valid, suffered from poor scalability due to reliance on column chromatography and excessive reagent usage. By fundamentally re-engineering the Wittig condensation step and the subsequent desilylation purification, this patent enables the production of high-purity intermediates with significantly improved yield and cost-efficiency. For global procurement and R&D teams, understanding this shift from laboratory-scale purification to industrial crystallization is essential for securing a reliable supply chain for this blockbuster lipid-lowering agent. The following analysis dissects the technical innovations that make this process viable for multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical benchmark for Rosuvastatin synthesis, established by EP0521471, relies heavily on conditions that are economically and operationally prohibitive for large-scale manufacturing. In the conventional Wittig condensation, the phosphorus ylide reagent is typically used in significant excess, often around 1.5 equivalents relative to the aldehyde, leading to a utilization rate of less than 50% for this expensive starting material. Furthermore, the reaction traditionally requires massive solvent loads, typically exceeding 10 volumes relative to the reagent mass, which drastically increases energy consumption for heating and subsequent distillation. Perhaps most critically, the purification of the resulting intermediate necessitates column chromatography to remove triphenylphosphine oxide and unreacted starting materials, a technique that is virtually impossible to scale efficiently in a GMP environment. These factors combine to create a process with low total recovery, high operational costs, and significant difficulties in maintaining consistent purity profiles across batches.
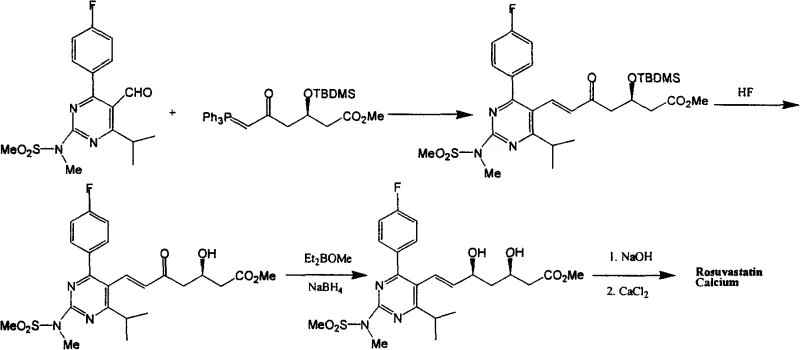
The Novel Approach
In stark contrast, the methodology disclosed in CN101591302A introduces a paradigm shift by optimizing stoichiometry and solvent dynamics to facilitate crystallization-based purification. The inventors discovered that reducing the ylide reagent charge to a range of 0.5 to 1.5 equivalents, with an optimal window of 0.9 to 1.2 equivalents, surprisingly improves the reaction profile rather than hindering it. This precise control minimizes the formation of oily impurities that typically inhibit crystallization, allowing the target intermediate to precipitate directly from the reaction mixture or upon simple workup. Additionally, the solvent load is dramatically reduced to between 0.1 and 5 volumes, with 0.5 volumes being optimal, which accelerates reaction kinetics and simplifies solvent recovery. By replacing column chromatography with recrystallization using solvents like hexane or ether mixtures, the process achieves purity levels exceeding 99.0% while being fully adaptable to industrial reactor setups.
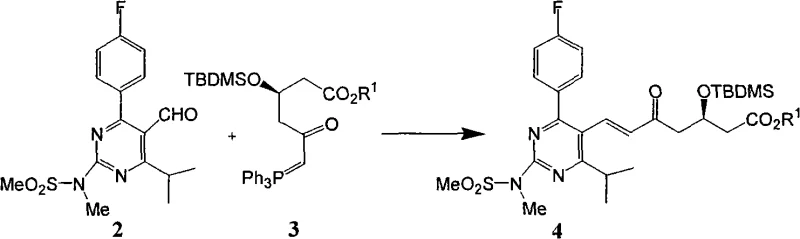
Mechanistic Insights into Optimized Wittig Condensation and Deprotection
The core mechanistic breakthrough lies in the intricate relationship between reagent stoichiometry and the physical state of the crude product. In traditional Wittig reactions, excess ylide reagent and its decomposition products often remain in the mixture as viscous oils or syrups, which act as impurities that prevent the nucleation and growth of crystals for the desired heptenoic acid ester. By strictly limiting the ylide reagent to near-stoichiometric amounts, the process ensures that the byproduct, triphenylphosphine oxide, becomes the dominant solid impurity, which can be effectively removed through selective recrystallization in non-polar solvents like hexane. This leaves the desired intermediate in a state where it can be easily isolated. Furthermore, the subsequent desilylation step using hydrofluoric acid in acetonitrile is optimized to allow for direct phase separation and solvent recycling, avoiding the complex extraction procedures that generate large volumes of mixed waste solvents in older methods.
Impurity control is further enhanced by the specific choice of solvents and temperatures during the workup phases. The patent details how the crude product from the Wittig reaction, containing minimal residual aldehyde and triphenylphosphine oxide, can undergo a direct transformation to the deprotected alcohol without intermediate chromatographic purification. The desilylation is conducted at controlled low temperatures (0-5°C) to prevent side reactions, followed by a careful pH adjustment to neutralize the acid. The resulting product, Formula 5, possesses a crystalline nature that allows for final polishing via recrystallization in ether or acetone/isopropyl ether mixtures. This sequence ensures that diastereomeric impurities and other organic byproducts are excluded from the crystal lattice, resulting in a final intermediate that meets stringent pharmaceutical specifications without the need for preparative HPLC or flash chromatography.
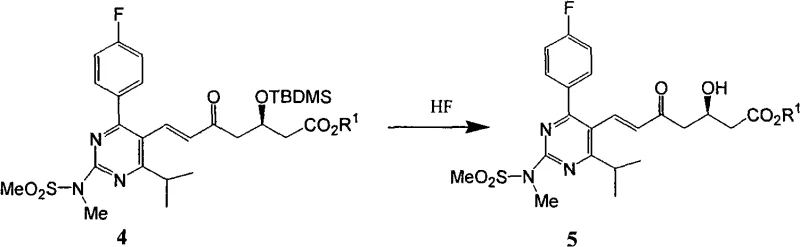
How to Synthesize Rosuvastatin Intermediate Efficiently
The synthesis of the key Rosuvastatin intermediate Formula 5 is achieved through a streamlined two-step sequence that prioritizes operational simplicity and material efficiency. The process begins with the condensation of the pyrimidine aldehyde and the chiral phosphorus ylide under reflux in acetonitrile, utilizing a carefully calculated molar ratio to maximize yield while minimizing waste. Following the reaction, the mixture is concentrated and treated with hexane to precipitate triphenylphosphine oxide, leaving the desired ester in solution or as a solid that can be further purified. The second step involves the removal of the silyl protecting group using an HF-acetonitrile complex, followed by neutralization and a final recrystallization to secure high purity. For detailed standard operating procedures and specific parameter settings, please refer to the guide below.
- Perform Wittig condensation between pyrimidine aldehyde and phosphorus ylide reagent using a stoichiometric ratio (0.9-1.2 eq) in low solvent load (0.5 vol) acetonitrile.
- Remove triphenylphosphine oxide byproduct via hexane recrystallization instead of column chromatography to isolate intermediate Formula 4.
- Execute desilylation using HF-acetonitrile solution followed by pH adjustment and solvent recrystallization to obtain high-purity Formula 5.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN101591302A offers transformative benefits in terms of cost structure and supply reliability. The elimination of column chromatography is perhaps the most significant economic driver, as it removes the need for expensive silica gel, large volumes of elution solvents, and the specialized labor required for batch chromatography operations. This shift to crystallization-based purification not only lowers the direct cost of goods sold but also drastically reduces the cycle time per batch, allowing for higher throughput in existing manufacturing facilities. Furthermore, the optimization of solvent usage means that less energy is required for heating and distillation, contributing to a lower carbon footprint and reduced utility costs, which aligns with modern sustainability goals in chemical manufacturing.
- Cost Reduction in Manufacturing: The strategic reduction of the expensive ylide reagent from excess to near-stoichiometric levels directly lowers the raw material cost per kilogram of the intermediate. By avoiding the losses associated with column chromatography and improving the overall yield through better impurity control, the process delivers substantial cost savings. The ability to recycle solvents like acetonitrile and hexane efficiently further compounds these financial benefits, making the production of this high-value intermediate significantly more economical compared to legacy methods.
- Enhanced Supply Chain Reliability: Reliance on chromatography often creates bottlenecks that limit production capacity and extend lead times, whereas crystallization is a unit operation that scales linearly and predictably. This process modification ensures that manufacturers can meet large-volume demands without the risk of purification bottlenecks, thereby securing a more stable supply of the Rosuvastatin intermediate. The robustness of the crystallization process also implies better batch-to-batch consistency, reducing the risk of quality failures that could disrupt the supply chain.
- Scalability and Environmental Compliance: The reduction in solvent volume and the removal of silica-based purification steps significantly decrease the generation of hazardous solid waste and spent solvent mixtures. This makes the process easier to permit and operate within strict environmental regulations, facilitating smoother scale-up from pilot plant to commercial tonnage. The simplified waste stream management reduces disposal costs and enhances the overall environmental profile of the manufacturing site, a key consideration for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on how this method overcomes previous technical barriers. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own production lines or for sourcing intermediates produced via this superior methodology.
Q: How does this patent improve upon the original EP0521471 synthesis route?
A: The patent CN101591302A significantly optimizes the EP0521471 route by reducing the excess usage of the expensive ylide reagent from 1.5 equivalents to near-stoichiometric amounts (0.9-1.2 eq) and drastically lowering solvent consumption. Crucially, it replaces difficult-to-scale column chromatography purification with efficient recrystallization steps, enabling true industrial manufacturing.
Q: What is the key advantage of the new purification method for Formula 5?
A: The primary advantage is the elimination of column chromatography, which is notoriously difficult to scale and costly. By optimizing the reaction stoichiometry and solvent load, the crude product can be directly purified via solvent recrystallization (using ethers or mixed solvents), achieving purity levels exceeding 99.0% suitable for pharmaceutical applications.
Q: Why is controlling the ylide reagent ratio critical in this synthesis?
A: Controlling the ylide reagent ratio is critical because excessive reagent leads to impurities that exist as oils or syrups, preventing the crystallization of the desired product Formula 5. Reducing the reagent load minimizes these oily impurities and facilitates the removal of triphenylphosphine oxide, ensuring the final product precipitates as a high-purity solid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, scalable synthesis routes for high-demand pharmaceutical intermediates like those used in Rosuvastatin production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101591302A are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for API synthesis, providing our partners with absolute confidence in material quality and consistency.
We invite global pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how our advanced capabilities can support your supply chain needs. By leveraging our expertise in process optimization, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technical excellence translates into tangible value for your organization.
