Advanced Rosuvastatin Calcium Synthesis: High-Purity Intermediates and Scalable Manufacturing
Introduction to Next-Generation Rosuvastatin Calcium Synthesis
The pharmaceutical landscape for HMG-CoA reductase inhibitors continues to evolve, driven by the demand for higher purity and more efficient manufacturing processes. Patent CN110642790B, published in early 2021, introduces a transformative approach to the preparation of Rosuvastatin Calcium, a blockbuster lipid-lowering agent. This technology addresses long-standing challenges in the field by integrating an Evans auxiliary structure into the synthetic pathway, fundamentally altering the physical properties of key intermediates. Unlike traditional methods that rely on difficult-to-handle oily substances, this novel route generates solid intermediates, specifically compounds of Formula 5 and Formula 6, which are far easier to purify and characterize. 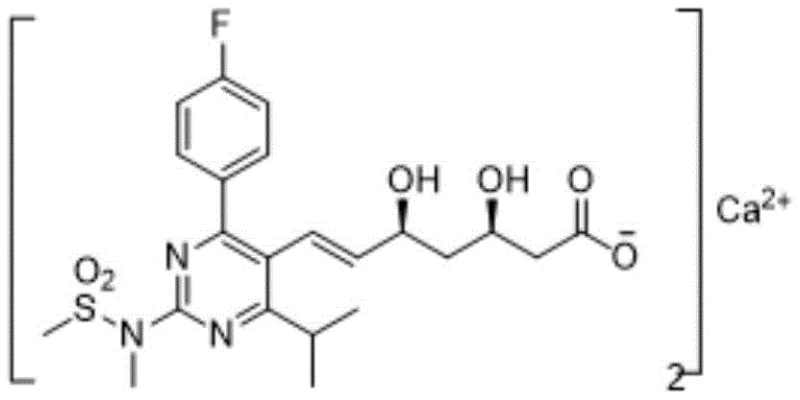 For procurement and supply chain leaders, this shift represents a significant opportunity to enhance supply reliability. The process utilizes a Julia olefination strategy that offers superior stereocontrol compared to conventional Wittig reactions, ensuring high E/Z selectivity without the need for extreme cryogenic conditions. By leveraging this intellectual property, manufacturers can achieve a final product purity of up to 99.9%, meeting the stringent quality standards required by global regulatory bodies for cardiovascular medications.
For procurement and supply chain leaders, this shift represents a significant opportunity to enhance supply reliability. The process utilizes a Julia olefination strategy that offers superior stereocontrol compared to conventional Wittig reactions, ensuring high E/Z selectivity without the need for extreme cryogenic conditions. By leveraging this intellectual property, manufacturers can achieve a final product purity of up to 99.9%, meeting the stringent quality standards required by global regulatory bodies for cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin Calcium has been plagued by several technical bottlenecks that impact both cost and quality. Many established routes, such as those disclosed in earlier patents like EP0521471A1 and WO0049014A1, rely heavily on Wittig reactions to construct the critical carbon-carbon double bond. A major drawback of the Wittig reaction in this context is its inherent lack of stereocontrol, often producing a mixture of E and Z isomers that requires tedious and yield-reducing separation steps. Furthermore, recent prior art, such as the route disclosed in CN201910190264, attempts to address side-chain synthesis but introduces new vulnerabilities. 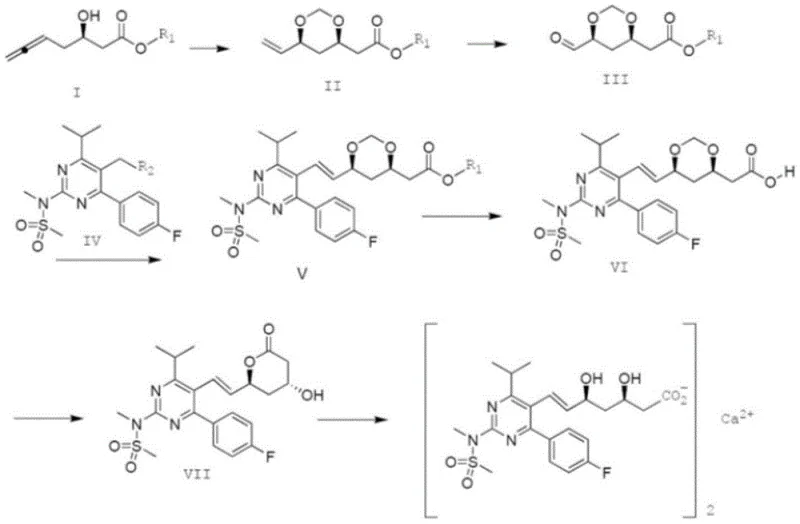 As illustrated in the referenced prior art, these methods often involve the oxidation of olefins to aldehydes, a step that frequently suffers from low yields and the generation of oxidative impurities. Additionally, a pervasive issue across many legacy processes is the physical state of the intermediates; most are oils. Oily intermediates are notoriously difficult to purify via crystallization, often requiring column chromatography which is impractical for large-scale industrial production. This leads to batch-to-batch variability, extended processing times, and increased solvent consumption, all of which drive up the cost of goods sold (COGS) and complicate supply chain logistics.
As illustrated in the referenced prior art, these methods often involve the oxidation of olefins to aldehydes, a step that frequently suffers from low yields and the generation of oxidative impurities. Additionally, a pervasive issue across many legacy processes is the physical state of the intermediates; most are oils. Oily intermediates are notoriously difficult to purify via crystallization, often requiring column chromatography which is impractical for large-scale industrial production. This leads to batch-to-batch variability, extended processing times, and increased solvent consumption, all of which drive up the cost of goods sold (COGS) and complicate supply chain logistics.
The Novel Approach
The methodology outlined in CN110642790B offers a robust solution to these historical inefficiencies by re-engineering the synthetic backbone. The core innovation lies in the introduction of a chiral Evans auxiliary (an oxazolidinone derivative) early in the sequence. This strategic modification transforms the synthesis from a series of oily transformations into a process dominated by solid-state chemistry. 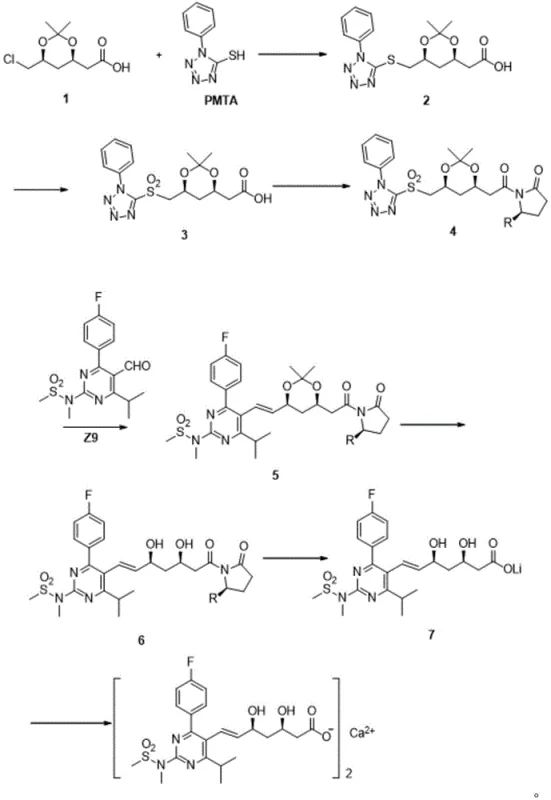 As depicted in the comprehensive reaction scheme, the route proceeds through well-defined solid intermediates (Formula 5 and Formula 6), allowing for simple filtration and washing steps to remove impurities rather than complex chromatographic separations. The replacement of the Wittig reaction with a Julia olefination is another critical improvement. This reaction, coupling the sulfone-bearing side chain (Formula 4) with the pyrimidine aldehyde (Formula Z9), provides exceptional control over the geometry of the resulting alkene. Moreover, the process conditions are remarkably mild; while some steps utilize cooling, the overall thermal profile is manageable, avoiding the extreme cryogenic temperatures often associated with high-selectivity organometallic reactions. This combination of solid intermediates and selective coupling creates a streamlined, scalable pathway ideal for commercial manufacturing.
As depicted in the comprehensive reaction scheme, the route proceeds through well-defined solid intermediates (Formula 5 and Formula 6), allowing for simple filtration and washing steps to remove impurities rather than complex chromatographic separations. The replacement of the Wittig reaction with a Julia olefination is another critical improvement. This reaction, coupling the sulfone-bearing side chain (Formula 4) with the pyrimidine aldehyde (Formula Z9), provides exceptional control over the geometry of the resulting alkene. Moreover, the process conditions are remarkably mild; while some steps utilize cooling, the overall thermal profile is manageable, avoiding the extreme cryogenic temperatures often associated with high-selectivity organometallic reactions. This combination of solid intermediates and selective coupling creates a streamlined, scalable pathway ideal for commercial manufacturing.
Mechanistic Insights into Julia Olefination and Auxiliary Control
At the heart of this technological advancement is the precise application of the Julia olefination mechanism, facilitated by the Evans auxiliary. The reaction begins with the formation of the sulfone intermediate (Formula 4), where the chiral auxiliary is attached to the carboxylic acid precursor using standard coupling agents like DCC and DMAP. This auxiliary serves a dual purpose: it acts as a protecting group for the carboxylic acid functionality and imposes steric constraints that influence the stereochemical outcome of subsequent reactions. When Formula 4 reacts with the aldehyde Formula Z9 in the presence of a base such as potassium tert-butoxide (t-BuOK), a beta-hydroxy sulfone intermediate is formed, which subsequently undergoes elimination to yield the alkene. 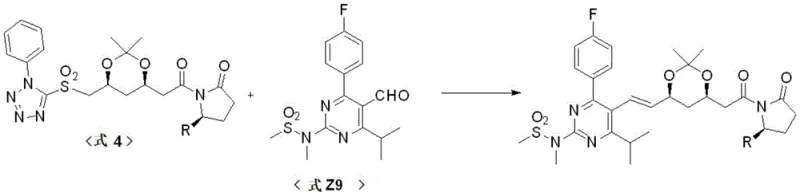 The beauty of this mechanism, as detailed in the patent, is its ability to favor the thermodynamically stable E-isomer, which is the biologically active configuration required for Rosuvastatin. The elimination step is driven by the stability of the resulting conjugated system and the steric bulk of the auxiliary, effectively suppressing the formation of the unwanted Z-isomer. Following the coupling, the auxiliary is removed under mild acidic hydrolysis conditions. Unlike harsh saponification or reduction methods that might epimerize sensitive chiral centers on the heptanoic acid chain, the acidic deprotection described here preserves the integrity of the (3R, 5S) stereocenters. This mechanistic elegance ensures that the final API not only meets chemical purity specifications but also maintains the strict enantiomeric excess required for therapeutic efficacy.
The beauty of this mechanism, as detailed in the patent, is its ability to favor the thermodynamically stable E-isomer, which is the biologically active configuration required for Rosuvastatin. The elimination step is driven by the stability of the resulting conjugated system and the steric bulk of the auxiliary, effectively suppressing the formation of the unwanted Z-isomer. Following the coupling, the auxiliary is removed under mild acidic hydrolysis conditions. Unlike harsh saponification or reduction methods that might epimerize sensitive chiral centers on the heptanoic acid chain, the acidic deprotection described here preserves the integrity of the (3R, 5S) stereocenters. This mechanistic elegance ensures that the final API not only meets chemical purity specifications but also maintains the strict enantiomeric excess required for therapeutic efficacy.
How to Synthesize Rosuvastatin Calcium Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly during the coupling and oxidation phases. The process is designed to be operationally simple, utilizing common solvents like THF, dichloromethane, and MTBE, which facilitates technology transfer from lab to plant. The initial steps involve building the chiral side chain, followed by the critical Julia coupling at controlled temperatures (around -40°C to room temperature) to maximize selectivity. Subsequent hydrolysis and oxidation steps are performed under aqueous conditions that allow for easy phase separation and extraction. For a detailed breakdown of the specific reagent quantities, temperature profiles, and workup procedures required to execute this synthesis according to GMP standards, please refer to the technical guide below.
- Preparation of Sulfone Intermediate (Formula 4): React the protected side-chain acid with an Evans oxazolidinone auxiliary using DCC and DMAP to form the key chiral sulfone precursor.
- Julia Olefination Coupling: React the sulfone intermediate (Formula 4) with the pyrimidine aldehyde (Formula Z9) using t-BuOK in THF/t-BuOH to establish the critical E-alkene bond with high selectivity.
- Deprotection and Salt Formation: Hydrolyze the auxiliary group under mild acidic conditions, oxidize the resulting alcohol to the acid, and finally treat with calcium acetate to precipitate high-purity Rosuvastatin Calcium.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the sourcing and production of cardiovascular APIs, the technical improvements in CN110642790B translate directly into tangible commercial benefits. The shift from oily to solid intermediates is not merely a chemical curiosity; it is a logistical game-changer that impacts inventory management, warehousing, and processing throughput. By adopting this route, manufacturers can mitigate the risks associated with unstable or difficult-to-handle liquid intermediates, ensuring a more consistent supply of high-quality material.
- Cost Reduction in Manufacturing: The economic implications of this route are profound. By eliminating the need for column chromatography—a staple in older, oil-based routes—the process significantly reduces solvent consumption and waste disposal costs. The use of solid intermediates allows for purification via crystallization or simple filtration, which are inherently cheaper and faster unit operations. Furthermore, the avoidance of expensive transition metal catalysts or exotic reagents in favor of cost-effective bases like t-BuOK and oxidants like hydrogen peroxide contributes to a leaner cost structure. The improved E/Z selectivity also means less material is lost to isomer separation, effectively increasing the overall yield of the process and lowering the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex syntheses with low yields or difficult purification steps. This novel method enhances reliability by simplifying the production workflow. The intermediates (Formula 5 and Formula 6) are stable solids that can be easily stored, transported, and quality-controlled before proceeding to the next step. This modularity allows for better inventory buffering and reduces the risk of batch failures propagating through the entire production line. Additionally, the use of readily available, commodity-grade chemicals reduces dependency on specialized suppliers, mitigating the risk of raw material shortages.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers substantial advantages. The reaction conditions are generally mild, avoiding the extreme hazards associated with cryogenic operations or highly pyrophoric reagents often found in alternative lithiation-based routes. The ability to perform key steps at or near room temperature reduces energy consumption for heating and cooling. Moreover, the simplified workup procedures generate less hazardous waste, aligning with modern green chemistry principles and facilitating easier compliance with increasingly strict environmental regulations in major pharmaceutical manufacturing hubs.
Frequently Asked Questions (FAQ)
Understanding the nuances of this synthesis is crucial for technical teams evaluating its adoption. The following questions address common inquiries regarding the stereochemistry, scalability, and purification advantages of this specific patent family. These answers are derived directly from the experimental data and technical disclosures within CN110642790B, providing a factual basis for your feasibility assessments.
Q: How does the Evans auxiliary improve the synthesis of Rosuvastatin Calcium?
A: The introduction of the Evans prosthetic group converts key oily intermediates into solids (specifically Formula 5 and Formula 6). This physical state change drastically simplifies purification processes like filtration and crystallization, leading to significantly higher final product purity compared to traditional oily routes.
Q: What are the advantages of using Julia Olefination over Wittig reactions in this context?
A: Traditional Wittig reactions often suffer from poor E/Z stereoselectivity and require difficult separation of isomers. The Julia olefination method utilized in this patent provides superior E-selectivity for the heptenoic acid side chain, reducing impurity profiles and eliminating the need for complex isomer separation steps.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It avoids harsh low-temperature conditions (operating mostly at room temperature or mild cooling like -40°C rather than cryogenic levels) and utilizes common, cost-effective reagents like t-BuOK and hydrogen peroxide, making it highly viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Supplier
The technical potential of the Evans auxiliary-mediated Julia olefination route is immense, offering a clear path to high-purity Rosuvastatin Calcium that meets global pharmacopeial standards. At NINGBO INNO PHARMCHEM, we specialize in translating such complex patent methodologies into robust, commercial-scale realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of API or intermediate we deliver performs consistently in your downstream formulations.
We invite you to leverage our expertise to optimize your sourcing strategy for this critical cardiovascular medication. Whether you require custom synthesis of the key solid intermediates or full-scale API production, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of this advanced chemistry can drive value for your organization.
