Advanced Synthesis of N-(Cyanoalkyl) Benzamides for High-Efficiency Agrochemical Production
The global demand for high-efficacy agrochemicals continues to drive innovation in intermediate synthesis, particularly for classes of compounds exhibiting potent insecticidal and fungicidal properties. Patent CN102020634A introduces a groundbreaking methodology for the preparation of N-(cyanoalkyl) benzamide compounds, a critical scaffold in modern crop protection chemistry. This technical disclosure addresses long-standing inefficiencies in the production of these bioactive molecules by proposing a route that bypasses traditional bottlenecks associated with nitro-group manipulation. For R&D directors and process chemists, the shift from conventional nitro-reduction pathways to a thionyl-mediated activation strategy represents a significant leap forward in synthetic elegance and operational safety. By leveraging 2-thionylaminobenzoyl chloride as a key starting material, the inventors have unlocked a pathway that not only streamlines the reaction sequence but also dramatically improves the overall mass balance of the process. This report analyzes the technical merits of this invention, highlighting its potential to redefine supply chain standards for reliable agrochemical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those detailed in WO2008/134969A1, typically rely on a multi-step sequence beginning with o-nitrobenzoic acid derivatives. In these traditional routes, the carboxylic acid must first be activated to an acid chloride, followed by condensation with a cyanoalkylamine to form an N-(cyanoalkyl) o-nitrobenzamide. The critical bottleneck arises in the subsequent step, where the nitro group must be chemically reduced to an amino group to yield the necessary N-(cyanoalkyl) anthranilamide intermediate. This reduction step is notoriously problematic, often requiring hazardous reagents, transition metal catalysts, or high-pressure hydrogenation equipment, all of which introduce significant safety risks and environmental burdens. Furthermore, the cumulative yield of this three-step sequence is frequently suboptimal, reported in the range of 47% to 60%, leading to substantial material loss and increased cost of goods sold. The subsequent coupling of this intermediate with a pyrazole acid halide traditionally requires the presence of an acid-binding agent to neutralize generated hydrogen halides, adding further complexity to the workup and purification stages.
The Novel Approach
The inventive method disclosed in CN102020634A fundamentally reimagines this synthetic landscape by introducing 2-thionylaminobenzoyl chloride as a superior precursor. This unique reagent allows for the direct formation of the N-(cyanoalkyl) anthranilamide intermediate in a single condensation step, effectively merging the activation and deprotection phases. As illustrated in the general reaction scheme below, the process circumvents the need for a discrete nitro reduction entirely. 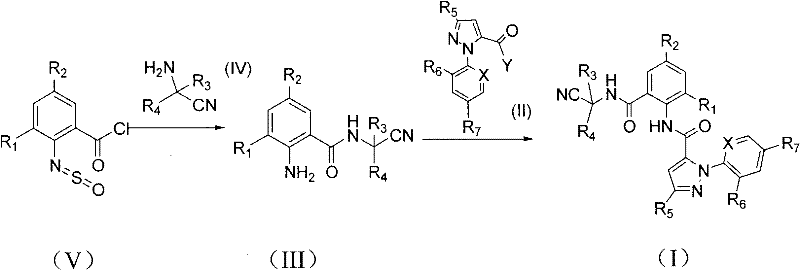 . Moreover, the second stage of the synthesis, involving the coupling of the anthranilamide intermediate with the pyrazole acid halide, is achieved under remarkably mild conditions without the addition of any acid-binding agent. This base-free protocol not only simplifies the reaction mixture but also eliminates the generation of salt by-products, thereby facilitating easier isolation of the final high-purity product. The result is a robust, high-yielding process that offers a compelling alternative for cost reduction in insecticide manufacturing.
. Moreover, the second stage of the synthesis, involving the coupling of the anthranilamide intermediate with the pyrazole acid halide, is achieved under remarkably mild conditions without the addition of any acid-binding agent. This base-free protocol not only simplifies the reaction mixture but also eliminates the generation of salt by-products, thereby facilitating easier isolation of the final high-purity product. The result is a robust, high-yielding process that offers a compelling alternative for cost reduction in insecticide manufacturing.
Mechanistic Insights into Thionyl-Mediated Amidation
The core chemical innovation lies in the dual functionality of thionyl chloride when reacting with 2-aminobenzoic acid derivatives. In the initial phase, thionyl chloride serves to convert the carboxylic acid moiety into a highly reactive acid chloride while simultaneously transforming the ortho-amino group into a thionylamino species (N=S=O). This transient modification acts as a protecting group that stabilizes the molecule during the activation phase. Upon introduction of the cyanoalkylamine nucleophile, the acid chloride undergoes rapid amidation. Crucially, under the reaction conditions employed—typically involving solvents like dichloromethane, THF, or toluene at temperatures ranging from -10°C to reflux—the thionylamino moiety is labile. It undergoes hydrolysis or nucleophilic displacement during the workup or reaction progression, regenerating the free ortho-amino group to yield the desired N-(cyanoalkyl) anthranilamide. This mechanism avoids the harsh conditions associated with nitro reduction, preserving sensitive functional groups elsewhere on the molecular scaffold.
In the subsequent coupling step, the regenerated ortho-amino group acts as a nucleophile attacking the pyrazole acid halide. The absence of an external base is particularly noteworthy from a mechanistic standpoint. It suggests that the inherent basicity of the anthranilamide nitrogen or the specific solvation environment is sufficient to drive the reaction forward, or that the leaving group (halide) is expelled efficiently without immediate neutralization, possibly forming a soluble complex that does not precipitate or interfere with the reaction kinetics. This subtle mechanistic feature is vital for impurity control, as it prevents the formation of N-acylated by-products that can occur when excess base promotes over-reaction. The specific synthesis of the key intermediate shown in Example 1 demonstrates this precision, where the conversion from the thionyl-species to the free amine occurs seamlessly. 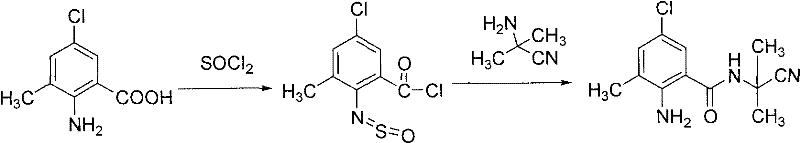 . This level of control ensures that the final agrochemical intermediates meet stringent purity specifications required for biological efficacy.
. This level of control ensures that the final agrochemical intermediates meet stringent purity specifications required for biological efficacy.
How to Synthesize N-(Cyanoalkyl) Benzamides Efficiently
Implementing this novel synthesis route requires careful attention to stoichiometry and solvent selection to maximize the benefits of the thionyl-mediated pathway. The process is designed to be scalable, utilizing common industrial solvents such as toluene, acetonitrile, or dichloromethane, which are readily available and easily recovered. The molar ratios are optimized to ensure complete conversion of the valuable pyrazole acid halide, typically employing a slight excess of the anthranilamide intermediate. Reaction temperatures are flexible, allowing operators to choose between ambient conditions for sensitive substrates or reflux conditions to accelerate kinetics for more sterically hindered analogues. The detailed standardized synthesis steps are provided in the guide below.
- Condense 2-thionylaminobenzoyl chloride with cyanoalkylamine in a suitable solvent to form the N-(cyanoalkyl) anthranilamide intermediate.
- React the resulting anthranilamide intermediate with pyrazole acid halide under reflux conditions.
- Isolate the final N-(cyanoalkyl) benzamide product without the need for additional acid-binding agents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond mere chemical curiosity. The elimination of the nitro reduction step removes a significant bottleneck from the production schedule, reducing the number of unit operations and the associated labor and utility costs. Furthermore, the avoidance of transition metal catalysts used in traditional reductions means there is no need for expensive and time-consuming heavy metal scavenging processes, which are often required to meet regulatory limits for residual metals in agrochemical products. This simplification directly contributes to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The most significant economic driver is the removal of the acid-binding agent in the final coupling step. In large-scale production, the cost of bases such as triethylamine or inorganic carbonates, along with the disposal of the resulting salt waste, constitutes a non-trivial portion of variable costs. By operating under base-free conditions, the process drastically reduces raw material consumption and waste treatment expenses. Additionally, the high yields reported in the patent examples indicate a more efficient use of starting materials, minimizing the financial impact of material loss and improving the overall atom economy of the synthesis.
- Enhanced Supply Chain Reliability: The reliance on commercially available or easily synthesized starting materials, such as substituted anthranilic acids and thionyl chloride, ensures a stable supply chain. Unlike specialized reduction catalysts which may face supply constraints or price volatility, the reagents for this process are commodity chemicals. This stability allows for better long-term planning and reduces the risk of production delays caused by raw material shortages. The robustness of the reaction conditions also implies a lower rate of batch failures, ensuring consistent delivery schedules to downstream formulators.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. Avoiding nitro reduction eliminates the generation of potentially explosive intermediates and the use of high-pressure hydrogenation equipment, lowering the facility's risk profile. The base-free coupling further reduces the chemical oxygen demand (COD) of the wastewater stream by preventing salt accumulation. These factors facilitate easier regulatory compliance and support sustainability goals, making the commercial scale-up of complex agrochemical intermediates more feasible and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is essential for assessing the fit of this technology within existing production infrastructure.
Q: How does this method improve upon the traditional nitro-reduction route?
A: This novel approach eliminates the hazardous and low-yielding nitro reduction step found in prior art (WO2008/134969A1), utilizing a thionylamino precursor that directly yields the anthranilamide intermediate with significantly higher efficiency.
Q: Is an acid-binding agent required for the final coupling step?
A: No, a distinct advantage of this patented process is the ability to perform the final condensation between the anthranilamide and pyrazole acid halide without any acid-binding agent, simplifying purification and reducing waste.
Q: What represents the primary cost-saving driver in this synthesis?
A: The primary cost drivers are reduced through the telescoping of the activation and protection steps, the removal of expensive reduction catalysts or reagents, and the elimination of base scavengers in the final coupling stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(Cyanoalkyl) Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the competitive agrochemical market. Our team of expert process chemists has extensively evaluated the technology described in CN102020634A and possesses the capability to translate this laboratory-scale innovation into commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and simplified workup are fully realized in a GMP-compliant manufacturing environment. Our rigorous QC labs and stringent purity specifications guarantee that every batch of N-(cyanoalkyl) benzamide intermediates meets the exacting standards required for final pesticide formulation.
We invite you to collaborate with us to leverage this advanced chemistry for your product pipeline. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your supply chain.
