Advanced Synthesis of N-(Cyanoalkyl) Benzamides for Scalable Agrochemical Production
Advanced Synthesis of N-(Cyanoalkyl) Benzamides for Scalable Agrochemical Production
The global demand for high-efficiency insecticides and fungicides continues to drive innovation in the synthesis of complex heterocyclic intermediates. A pivotal advancement in this sector is detailed in Chinese Patent CN102020634B, which discloses a novel preparation method for N-(cyanoalkyl) benzamide compounds. These compounds, exemplified by structures such as 3-bromo-N-(4-chloro-2-(2-cyanopropyl-2-ylcarboxamido)-6-methylphenyl)-1-(3-chloro-2-pyridyl)-1H-pyrazole-5-carboxamide, exhibit potent biological activity against agricultural pests. The patent introduces a transformative synthetic strategy that replaces the traditional, multi-step nitro-reduction pathway with a more direct condensation involving 2-thionylaminobenzoyl chloride. This shift not only streamlines the manufacturing workflow but also addresses critical pain points regarding yield optimization and environmental compliance, making it a highly attractive route for industrial scale-up.
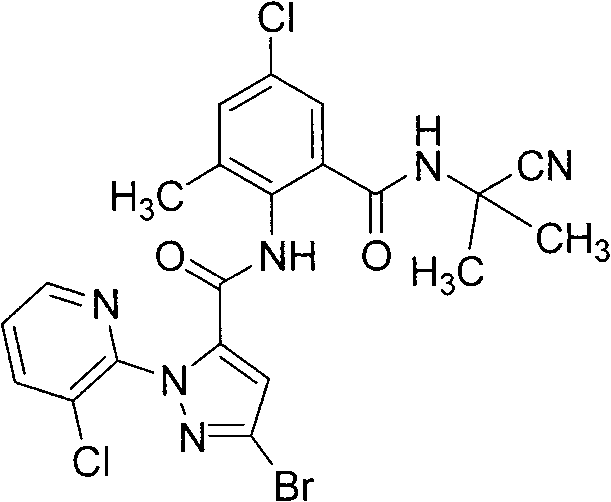
For R&D directors and process chemists, the structural integrity and purity of these intermediates are paramount. The disclosed method ensures the production of high-purity N-(cyanoalkyl) benzamides by leveraging the unique reactivity of the thionyl-amino group. Unlike conventional routes that require harsh reducing agents to convert nitro groups to amines—often introducing metallic impurities—this new approach utilizes a condensation reaction that inherently generates the free amine functionality required for the subsequent acylation. This mechanistic elegance reduces the impurity profile significantly, facilitating easier purification and ensuring that the final active pharmaceutical or agrochemical ingredient meets stringent regulatory specifications without extensive recrystallization or chromatography.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-(cyanoalkyl) benzamide derivatives relied heavily on starting materials like o-nitrobenzoic acid. As documented in prior art such as WO2008/134969A1, the traditional pathway involves acyl chlorination of o-nitrobenzoic acid, followed by condensation with cyanoalkylamine to form an N-(cyanoalkyl) o-nitrobenzamide. Crucially, this intermediate then requires a reduction step to convert the nitro group into the necessary amino group before the final coupling can occur. This three-step sequence is inherently inefficient, with reported total yields ranging merely from 47% to 60%. Furthermore, the reduction step often necessitates the use of transition metal catalysts or stoichiometric reducing agents, which introduce heavy metal contaminants and generate substantial hazardous waste, complicating both cost management and environmental safety protocols in large-scale manufacturing facilities.
The Novel Approach
The methodology presented in CN102020634B represents a paradigm shift by utilizing 2-thionylaminobenzoyl chloride as the key starting material. This innovative route allows for the direct condensation with cyanoalkylamine to produce the N-(cyanoalkyl) o-aminobenzamide intermediate in a single, high-yielding operation. By bypassing the nitro-reduction step entirely, the process reduces the number of unit operations and minimizes the exposure of sensitive functional groups to harsh reducing conditions. The subsequent reaction with pyrazole acyl halides is conducted under neutral conditions without the need for acid-binding agents, a feature that drastically simplifies the workup procedure. This approach not only enhances the overall atom economy but also aligns perfectly with modern green chemistry principles, offering a robust solution for the cost reduction in agrochemical intermediate manufacturing.
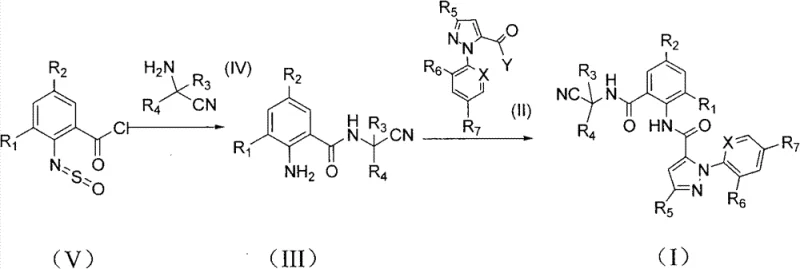
Mechanistic Insights into Thionyl-Amino Mediated Condensation
The core of this technological breakthrough lies in the unique behavior of the 2-thionylaminobenzoyl chloride moiety. In standard amide couplings, the presence of a free amine usually requires protection or careful pH control to prevent self-reaction or polymerization. However, in this specific system, the thionyl-amino group acts as a latent amine source. During the condensation with the cyanoalkylamine, the reaction conditions facilitate the transformation of the N=S=O functionality into the free primary amine (NH2) found in the intermediate (III). This transformation is highly selective and proceeds with remarkable efficiency, avoiding the formation of side products typically associated with nitro-group reductions. The mechanistic pathway suggests a concerted displacement or hydrolysis-like event during the workup that unveils the reactive aniline nitrogen, ready for the subsequent acylation step.
Furthermore, the final coupling step between the anthranilamide intermediate and the pyrazole acyl halide is engineered to proceed without exogenous bases. In traditional peptide or amide bond formations, tertiary amines like triethylamine or pyridine are added to scavenge the generated hydrogen chloride. However, this patent demonstrates that the nucleophilicity of the ortho-amino group in the intermediate is sufficient to drive the reaction forward even in the absence of such additives. This base-free condition is critical for impurity control; it prevents the formation of quaternary ammonium salts and eliminates the need for aqueous washes to remove excess base, thereby reducing the volume of organic solvents and water required for purification. This results in a cleaner reaction profile and a more straightforward isolation of the final crystalline product.
How to Synthesize N-(Cyanoalkyl) Benzamide Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the thionyl-amino chemistry. The process begins with the preparation of the 2-thionylaminobenzoyl chloride, which can be derived from the corresponding anthranilic acid using thionyl chloride. This activated species is then reacted with the cyanoalkylamine in a solvent such as tetrahydrofuran or dichloromethane. The reaction temperature is carefully managed, typically ranging from -10°C to reflux, to ensure complete conversion while minimizing thermal degradation. Following the isolation of the amino-intermediate, the final coupling is performed by mixing the intermediate with the pyrazole acyl halide in a polar aprotic solvent like acetonitrile. The mixture is heated to reflux, allowing the evolved hydrogen chloride gas to escape or be trapped, driving the equilibrium towards the product without the need for solid bases.
- Condense 2-thionylaminobenzoyl chloride with cyanoalkylamine in a suitable solvent at -10°C to reflux to form the N-(cyanoalkyl) anthranilamide intermediate.
- React the resulting anthranilamide intermediate with pyrazole acyl halide in a solvent such as acetonitrile or dichloromethane.
- Heat the mixture to reflux for 1-10 hours without adding any acid-binding agents to obtain the final N-(cyanoalkyl) benzamide product with high purity.
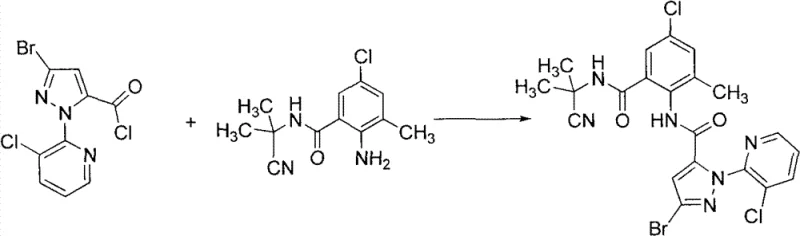
To visualize the practical application of this method, consider the synthesis of Compound 14 as described in the patent examples. The reaction of 2-amino-5-chloro-N-(2-cyanopropyl-2-yl)-3-methylbenzamide with 3-bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carbonyl chloride proceeds smoothly in acetonitrile. The absence of base allows for a clean reaction profile, yielding the final product with an HPLC purity of 96% and an isolated yield of 93%. This specific example underscores the robustness of the protocol across different substitution patterns on the aromatic rings, confirming its versatility for generating a library of analogues for biological testing.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers compelling economic and logistical benefits. The elimination of the nitro-reduction step removes a significant bottleneck in the production schedule. Reduction reactions often require specialized equipment, such as hydrogenation reactors, and pose safety risks associated with high-pressure hydrogen or pyrophoric catalysts. By circumventing this step, manufacturers can utilize standard glass-lined or stainless steel reactors, reducing capital expenditure and maintenance costs. Additionally, the higher overall yield means that less raw material is consumed per kilogram of final product, directly translating to substantial cost savings in the bill of materials. This efficiency is crucial for maintaining competitive pricing in the volatile agrochemical market.
- Cost Reduction in Manufacturing: The most significant financial advantage stems from the simplified process flow. By removing the reduction step and the need for acid-binding agents in the final coupling, the consumption of auxiliary chemicals is drastically reduced. Traditional methods require stoichiometric amounts of bases and reducing agents, which add to the raw material cost and generate expensive waste streams that require treatment. The new method's ability to operate without these additives lowers the variable cost of production. Furthermore, the high yields reported (often exceeding 90%) minimize the loss of valuable intermediates, ensuring that the maximum amount of input material is converted into saleable product, thereby optimizing the return on investment for every batch produced.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by complex, multi-step syntheses that depend on a long list of specialized reagents. This novel method relies on readily available starting materials like anthranilic acid derivatives and thionyl chloride, which are commodity chemicals with stable supply lines. The reduction in process steps also shortens the manufacturing lead time, allowing for faster response to market demand fluctuations. With fewer unit operations, there are fewer opportunities for process deviations or batch failures, leading to more consistent delivery schedules. This reliability is essential for downstream formulators who depend on a steady supply of high-quality intermediates to maintain their own production timelines for finished pesticide products.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental challenges, particularly regarding waste disposal. The base-free condensation step significantly reduces the salt load in the wastewater, easing the burden on effluent treatment plants. Moreover, avoiding heavy metal catalysts used in nitro reductions eliminates the risk of metal contamination in the final product and the environment. This aligns with increasingly stringent global environmental regulations, reducing the risk of regulatory shutdowns or fines. The process is inherently safer and cleaner, making it easier to obtain the necessary permits for commercial scale-up of complex agrochemical intermediates, thus ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding the practicality, safety, and quality aspects of the patented method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of using 2-thionylaminobenzoyl chloride over o-nitrobenzoic acid?
A: Using 2-thionylaminobenzoyl chloride eliminates the need for a separate nitro-reduction step, significantly simplifying the synthetic route and improving overall yield compared to traditional methods.
Q: Why is the absence of an acid acceptor beneficial in the final condensation step?
A: Operating without an acid acceptor reduces the generation of salt by-products, simplifies downstream purification, lowers wastewater treatment costs, and enhances the environmental profile of the manufacturing process.
Q: What yields can be expected from this novel preparation method?
A: The patent reports unexpectedly high yields, with specific examples demonstrating isolated yields ranging from 86% to 96% for the intermediates and final products, surpassing the 47%-60% range of conventional three-step routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(Cyanoalkyl) Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern agrochemical industry. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN102020634B and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this high-yield, base-free method are realized at an industrial level. Our state-of-the-art facilities are designed to handle complex heterocyclic chemistry with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards.
We invite potential partners to collaborate with us to leverage this advanced technology for their product pipelines. Whether you require custom synthesis of specific analogues or large-scale supply of the core intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patent can enhance your supply chain efficiency and reduce your overall manufacturing costs.
