Optimizing Adenosine Manufacturing: A Novel Zinc-Catalyzed Route for High-Purity API Production
Optimizing Adenosine Manufacturing: A Novel Zinc-Catalyzed Route for High-Purity API Production
The global demand for high-purity Adenosine, a critical active pharmaceutical ingredient (API) used in treating arrhythmias and as a precursor for ATP synthesis, necessitates robust and scalable manufacturing processes. Patent CN111808157B introduces a transformative chemical synthesis method that addresses long-standing inefficiencies in Adenosine production. Unlike traditional fermentation or older chemical routes that suffer from low yields and difficult purification, this novel approach utilizes a strategic combination of sterically hindered silyl protecting groups and a unique zinc-based catalytic system. By shifting the physical state of key intermediates from oils to solids, this technology offers a paradigm shift in process control, ensuring that high-purity Pharmaceutical Intermediates can be produced with greater consistency and reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
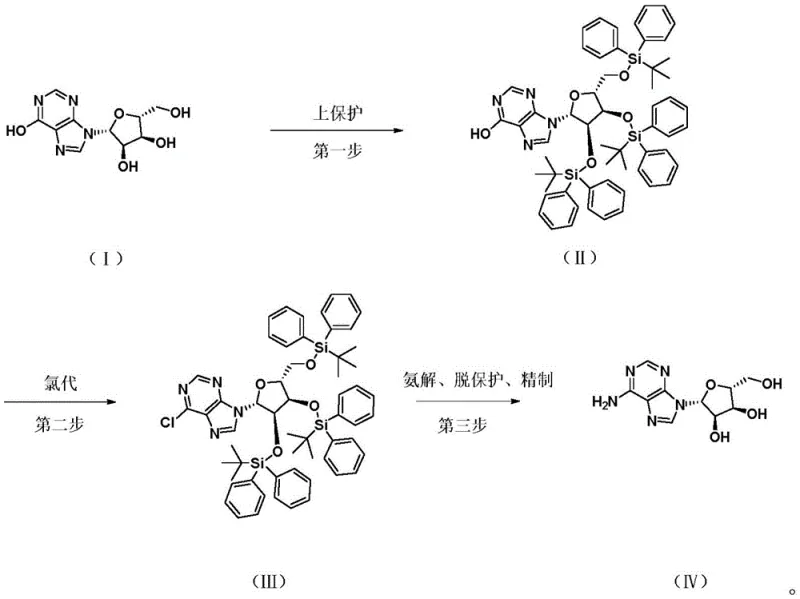
Historically, the chemical synthesis of Adenosine from Inosine relied heavily on acetyl protection strategies, such as those disclosed in earlier patents like CN1406946A. These conventional methods typically involve acetylating the hydroxyl groups of the ribose moiety using acetic anhydride, followed by chlorination and ammonolysis. However, a critical bottleneck in these legacy processes is the physical nature of the key intermediate, chlorotriacetylinosine, which exists as a viscous oil. This oily state presents severe challenges for industrial scale-up, as oils are notoriously difficult to purify via crystallization, often requiring complex column chromatography or extensive washing that leads to significant product loss. Furthermore, these oily intermediates are chemically less stable and prone to generating complex impurity profiles during the high-pressure ammonolysis step, ultimately compromising the quality of the final API and increasing the cost of goods sold due to low overall recovery rates.
The Novel Approach
In stark contrast, the methodology described in CN111808157B employs tert-butyldiphenylchlorosilane (TBDPSCl) as a protecting group, fundamentally altering the physical properties of the synthesis pathway. The introduction of the bulky tert-butyldiphenylsilyl (TBDPS) group converts the reaction intermediates into crystalline solids rather than oils. This physical transformation is a game-changer for commercial scale-up of complex Pharmaceutical Intermediates, as solid compounds can be easily purified through standard recrystallization and filtration techniques, eliminating the need for expensive and time-consuming chromatographic separations. Additionally, the TBDPS group offers superior stability against acid and base conditions compared to acetyl groups, ensuring that the molecular integrity of the ribose ring is maintained throughout the harsh chlorination and ammonolysis steps, thereby delivering a final product with exceptional purity profiles suitable for sensitive cardiovascular applications.
Mechanistic Insights into Zinc-Catalyzed Silylation and Vilsmeier Chlorination
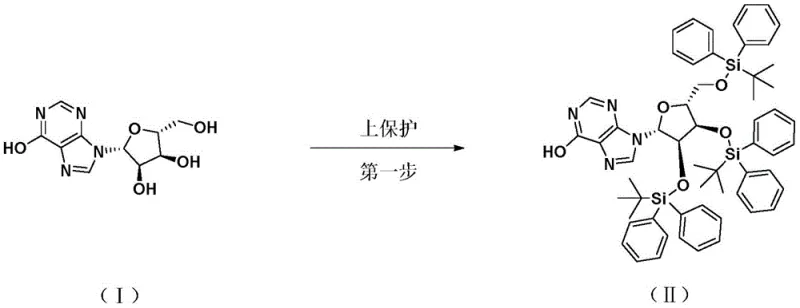
The core innovation of this synthesis lies in the first step: the protection of the hydroxyl groups on the Inosine ribose ring. While TBDPSCl is a powerful protecting agent, its large steric bulk typically results in slow reaction kinetics and poor yields when reacting with crowded nucleophiles like Inosine. To overcome this, the patent discloses a dual-catalyst system comprising Zinc powder and Zinc Chloride (ZnCl2). Mechanistically, the Zinc Chloride acts as a Lewis acid, coordinating with the chlorine atom of the TBDPSCl to generate a highly electrophilic silicon species. This activated complex is far more reactive towards the hydroxyl nucleophiles of Inosine, effectively bypassing the steric hindrance that usually plagues silylation reactions. Simultaneously, the Zinc powder serves to scavenge the hydrochloric acid byproduct generated during the reaction, regenerating Zinc Chloride in situ and driving the equilibrium forward. This synergistic catalytic cycle ensures rapid conversion at moderate temperatures (30°C to 40°C), achieving yields as high as 94.7% with purity exceeding 99.97%.
Following the protection step, the synthesis proceeds via a Vilsmeier-Haack type chlorination to convert the 6-hydroxyl group of the purine ring into a 6-chloro group, a prerequisite for the subsequent amination. The use of Vilsmeier reagent, generated in situ from DMF and thionyl chloride, allows for mild yet effective chlorination conditions. Because the ribose hydroxyls are robustly protected by the acid-resistant TBDPS groups, the sugar moiety remains intact despite the presence of thionyl chloride. The resulting 6-chloro intermediate (Compound III) retains its solid state, allowing for further purification via mixed solvent crystallization (isopropanol/n-hexane) before the final ammonolysis. This rigorous control over impurity generation at every stage ensures that the final ammonolysis step, conducted with aqueous ammonia at 120°C to 130°C, proceeds cleanly to yield Adenosine with HPLC purity levels reaching 99.99%, demonstrating the efficacy of this mechanistic design in reducing lead time for high-purity Pharmaceutical Intermediates.
How to Synthesize Adenosine Efficiently
The synthesis of Adenosine via this patented route involves a streamlined three-step sequence that prioritizes operational simplicity and high yield. The process begins with the zinc-catalyzed silylation of Inosine, followed by Vilsmeier chlorination, and concludes with high-temperature ammonolysis. Each step is designed to maximize the recovery of solid intermediates, minimizing material loss. For detailed laboratory protocols and specific reaction parameters, please refer to the standardized synthesis guide below.
- Protect Inosine hydroxyl groups using tert-butyldiphenylchlorosilane with Zinc/Zinc Chloride catalysts to form solid Compound II.
- React Compound II with Vilsmeier reagent (DMF/SOCl2) to introduce the 6-chloro group, yielding solid Compound III.
- Perform ammonolysis on Compound III using aqueous ammonia under heat and pressure, followed by recrystallization to obtain pure Adenosine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from oily acetyl-intermediates to solid silyl-intermediates represents a significant opportunity for cost reduction in Pharmaceutical Intermediates manufacturing. The ability to purify intermediates via simple crystallization rather than chromatography drastically reduces solvent consumption and processing time, directly lowering utility and waste disposal costs. Furthermore, the use of inexpensive and readily available catalysts like Zinc powder and Zinc Chloride eliminates the reliance on precious metal catalysts or exotic reagents, stabilizing the raw material cost structure and insulating the supply chain from volatile market fluctuations associated with specialized catalytic metals.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps is the primary driver of cost savings in this process. By engineering intermediates that crystallize spontaneously, the manufacturing workflow is simplified to basic unit operations like filtration and drying. This reduction in processing complexity not only lowers labor and equipment overhead but also significantly decreases the volume of organic solvents required for purification, leading to substantial savings in both raw material procurement and environmental compliance costs associated with solvent recovery and disposal.
- Enhanced Supply Chain Reliability: The robustness of the TBDPS protecting group enhances the stability of the intermediates, extending their shelf-life and reducing the risk of degradation during storage or transport between manufacturing sites. This chemical stability, combined with the use of commodity chemicals like Inosine, Zinc, and DMF, ensures a resilient supply chain that is less susceptible to disruptions. The high yields reported in the patent (over 90% for the first two steps) further guarantee consistent output volumes, allowing for more accurate production planning and reliable fulfillment of large-scale API orders without the unpredictability often associated with low-yielding fermentation processes.
- Scalability and Environmental Compliance: Scaling a process that relies on solid intermediates is inherently safer and more predictable than scaling one dependent on handling viscous oils, which can cause pumping and mixing issues in large reactors. The solid-state nature of Compounds II and III facilitates easier handling, weighing, and transfer in GMP environments. Additionally, the avoidance of heavy metal catalysts and the reduction in solvent intensity align with modern green chemistry principles, simplifying the regulatory approval process for new drug master files (DMFs) and ensuring long-term compliance with increasingly stringent environmental regulations regarding industrial effluent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-catalyzed Adenosine synthesis route. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on how this technology compares to incumbent methods.
Q: Why is the zinc-catalyzed silylation method superior to traditional acetylation?
A: Traditional acetylation produces oily intermediates that are difficult to purify and prone to impurities. The novel zinc-catalyzed silylation method using TBDPS groups yields solid intermediates, enabling simple crystallization and filtration, which drastically improves purity and process control.
Q: How does the use of Zinc powder and Zinc Chloride improve the reaction?
A: Tert-butyldiphenylchlorosilane has significant steric hindrance. The Zn/ZnCl2 system forms an electrophilic transition state that activates the silylating agent, overcoming steric barriers, reducing reaction time, and significantly increasing yield compared to uncatalyzed methods.
Q: What are the purity specifications achievable with this route?
A: This optimized route allows for intermediate purities exceeding 99.95% and final Adenosine API purity reaching 99.99% (HPLC), meeting stringent requirements for cardiovascular drug manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adenosine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the zinc-catalyzed Adenosine synthesis can be seamlessly transferred from the laboratory to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Adenosine meets the highest international standards for potency and impurity control.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this solid-intermediate process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a secure and efficient supply of high-quality Adenosine for your cardiovascular drug formulations.
