Revolutionizing Adenosine Cyclophosphate Production: A Scalable 2-Step Synthetic Strategy
Introduction to Advanced Nucleoside Synthesis
The landscape of nucleoside chemistry is constantly evolving, driven by the demand for high-purity intermediates essential for molecular biology and pharmaceutical applications. A significant breakthrough in this domain is detailed in patent CN109206465B, which discloses a highly efficient method for synthesizing adenosine cyclophosphate (cyclic AMP). This molecule, chemically known as adenosine-3',5'-cyclic phosphate (CAS: 60-92-4), serves as a critical second messenger in numerous physiological processes and is a hotspot in research regarding diseases such as psoriasis, coronary heart disease, and asthma. The disclosed technology represents a paradigm shift from legacy multi-step procedures to a streamlined, two-step continuous reaction sequence. By leveraging a direct acylation-cyclization strategy, this method circumvents the historical bottlenecks of low yields, excessive solvent consumption, and hazardous reagent usage. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance and commercial viability of this route is paramount for securing a stable supply chain of this vital biochemical.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
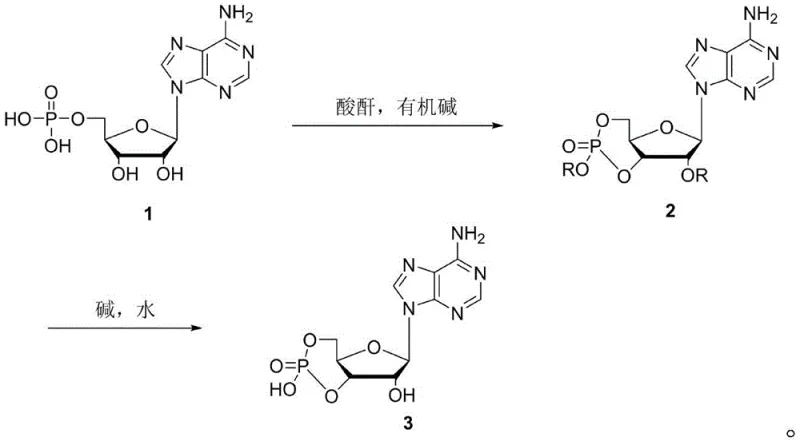
Historically, the industrial production of adenosine cyclophosphate has been plagued by significant technical and economic inefficiencies. The earliest reported methods, such as the alkaline hydrolysis of Adenosine Triphosphate (ATP) described by Lipkin et al., rely on extremely expensive raw materials that are difficult to source in bulk quantities. Furthermore, these processes often require harsh conditions, such as boiling barium hydroxide solutions at 100°C, which pose severe safety risks and complicate downstream purification. Alternative routes involving DCC dehydration or active ester methods introduce their own set of challenges; for instance, the Taguchi method necessitates the use of large volumes of boiling pyridine, a solvent notorious for its strong odor and stringent environmental disposal requirements. Additionally, methods employing metallic sodium, while theoretically viable, generate substantial amounts of dicyclohexylurea (DCU) waste and require rigorous safety protocols for handling reactive metals, making them unsuitable for modern green chemistry standards. The phosphorus oxychloride route, although using mild conditions, suffers from catastrophic solvent inefficiency, requiring hundreds of times the volume of solvent relative to the raw material, which drastically inflates production costs and environmental footprint.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach outlined in the patent data introduces a concise two-step synthesis that fundamentally redesigns the manufacturing logic. The first step involves the direct reaction of adenosine with an acid anhydride (preferably acetic anhydride) and an organic base to form an acyl-protected intermediate. This is immediately followed by a second step of alkaline hydrolysis in water to yield the final adenosine cyclophosphate. This strategy effectively bypasses the need for expensive activated nucleotides like ATP or hazardous dehydrating agents like DCC. By utilizing commodity chemicals such as acetic anhydride and triethylamine, the process significantly lowers the barrier to entry for raw material sourcing. Moreover, the reaction conditions are remarkably mild, typically operating between 70-80°C, which reduces energy consumption compared to high-temperature hydrolysis methods. The elimination of massive solvent volumes and the avoidance of toxic pyridine or reactive sodium metal position this method as a superior choice for cost reduction in pharmaceutical intermediates manufacturing, offering a cleaner, safer, and more economically sustainable pathway to high-purity cyclic nucleotides.
Mechanistic Insights into Acylation-Cyclization Cascade
The core of this synthetic innovation lies in the tandem acylation and intramolecular cyclization mechanism. In the first stage, the organic base, such as triethylamine or diisopropylethylamine, acts as a proton scavenger, activating the hydroxyl groups of the adenosine ribose moiety. The acid anhydride then reacts with these activated positions to form acyl esters. Crucially, under the heated conditions of 70-80°C in solvents like dichloroethane or acetonitrile, the phosphoryl group undergoes an intramolecular nucleophilic attack on the activated sugar carbons, facilitating the formation of the cyclic phosphate ester bond. This one-pot transformation effectively protects the sensitive amine and hydroxyl groups while simultaneously closing the phosphate ring, a feat that traditionally required separate protection and cyclization steps. The choice of solvent plays a pivotal role here; chlorinated solvents like 1,2-dichloroethane provide the optimal polarity to dissolve the polar nucleoside while maintaining the stability of the acylating agent, ensuring high conversion rates without the need for exotic catalysts.
Following the formation of the acyl-protected intermediate, the second stage involves a controlled alkaline hydrolysis. The addition of an inorganic base, such as sodium hydroxide or potassium carbonate, in an aqueous environment selectively cleaves the acyl protecting groups without disrupting the newly formed cyclic phosphate bond. This selectivity is critical for maintaining the structural integrity of the cyclic monophosphate while restoring the free hydroxyl groups necessary for biological activity. The reaction is conducted at room temperature (20-25°C), which minimizes the risk of ring-opening hydrolysis of the cyclic phosphate itself—a common side reaction in harsher basic conditions. The final product is isolated by precise pH adjustment, precipitating the zwitterionic cyclic AMP from the aqueous solution. This mechanism ensures that impurities such as linear AMP or unreacted adenosine are effectively separated during the crystallization phase, resulting in a final product with HPLC purity exceeding 99.0%, meeting the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Adenosine Cyclophosphate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins with the suspension of adenosine in a suitable organic solvent, followed by the sequential addition of the organic base and acid anhydride. Maintaining the reaction temperature between 70-80°C is essential to drive the cyclization to completion within a reasonable timeframe of 4 to 6 hours. Once the intermediate is formed, the workup involves a simple filtration to remove any unreacted starting material, followed by concentration. The subsequent hydrolysis step demands precise pH control; the reaction mixture is treated with aqueous base at ambient temperature, and the pH is carefully adjusted first to neutrality and then to acidic conditions (pH 4) to induce precipitation. For detailed operational parameters, stoichiometric ratios, and specific workup procedures validated on a kilogram scale, please refer to the standardized synthesis guide below.
- React adenosine with acetic anhydride and an organic base (e.g., triethylamine) in a solvent like dichloroethane at 70-80°C to form the acyl-protected intermediate.
- Treat the crude acyl-protected intermediate with an inorganic base (e.g., sodium hydroxide) in water at room temperature to effect cyclization and deprotection.
- Adjust pH to precipitate the product, followed by recrystallization from water to achieve high purity (>99%).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the supply chain for raw materials. By replacing scarce and expensive reagents like ATP or specialized coupling agents with ubiquitous commodities like acetic anhydride and triethylamine, manufacturers can insulate themselves from volatile market pricing and supply disruptions associated with niche fine chemicals. This shift not only stabilizes the cost of goods sold (COGS) but also enhances the overall resilience of the supply network, ensuring consistent production schedules even during global raw material shortages. Furthermore, the reduction in solvent usage directly translates to lower logistics costs for solvent transport and significantly reduced expenses for hazardous waste disposal, contributing to a leaner and more environmentally compliant operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of high-cost inputs and the streamlining of unit operations. Traditional methods often involve multiple isolation steps, extensive chromatography, or the use of precious metal catalysts that require expensive removal processes. In contrast, this two-step protocol relies on simple filtration and recrystallization for purification, removing the need for costly chromatographic columns or specialized scavenger resins. The use of acetic anhydride, a bulk chemical available at a fraction of the cost of ATP or DCC, fundamentally alters the cost structure of the final API intermediate. Additionally, the ability to recycle solvents like dichloroethane or acetonitrile further amplifies these savings, creating a manufacturing model that is inherently cost-competitive without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex reagents. This synthesis route mitigates that risk by utilizing a broad spectrum of commercially available solvents, including dichloromethane, chloroform, acetonitrile, and toluene. This flexibility allows production facilities to switch solvents based on regional availability and pricing without revalidating the entire process, providing a robust buffer against logistical bottlenecks. Moreover, the process has been successfully verified on a 10-kilogram scale, demonstrating that the kinetics and thermodynamics observed at the bench level translate effectively to pilot and commercial scales. This scalability assurance means that procurement teams can confidently place larger orders, knowing that the supplier has the technical capability to ramp up production volume rapidly to meet surging market demand.
- Scalability and Environmental Compliance: In an era of increasing regulatory scrutiny, the environmental profile of a chemical process is as critical as its yield. This method significantly reduces the E-factor (mass of waste per mass of product) by minimizing solvent volumes and avoiding the generation of heavy metal waste or difficult-to-degrade urea byproducts. The absence of pyridine and metallic sodium removes major occupational health and safety hazards, simplifying facility permitting and reducing insurance premiums. The straightforward aqueous workup in the second step generates a waste stream that is easier to treat compared to the complex organic sludge produced by DCC or phosphorus oxychloride methods. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand value of the final product for eco-conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of adenosine cyclophosphate using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, aiming to clarify the operational benefits and quality assurances associated with this route. Understanding these details is crucial for technical buyers evaluating potential partners for long-term supply agreements.
Q: How does this new method compare to traditional ATP hydrolysis routes?
A: Unlike traditional methods that rely on expensive Adenosine Triphosphate (ATP) and harsh barium hydroxide conditions at 100°C, this novel route utilizes cheap, commodity-grade adenosine and acetic anhydride. It eliminates the need for expensive starting materials and reduces energy consumption by operating at moderate temperatures (70-80°C).
Q: What are the safety advantages over metal sodium methods?
A: Previous methods utilizing metallic sodium posed significant safety risks regarding storage and handling, along with generating difficult-to-treat DCU waste. This acylation-cyclization protocol avoids reactive metals entirely, using stable organic bases like triethylamine, thereby enhancing operational safety and simplifying waste treatment.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly verifies the process on a 10-kilogram scale (Example 8), demonstrating robustness parallel to bench-scale results. The use of common solvents like dichloroethane and acetonitrile, combined with simple filtration and recrystallization workups, makes it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adenosine Cyclophosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex nucleosides like adenosine cyclophosphate requires not just chemical expertise, but a deep commitment to quality and scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards. Our capability to implement the efficient two-step acylation-cyclization route allows us to offer a product that is not only chemically superior but also commercially competitive, aligning perfectly with the needs of global pharmaceutical innovators.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new drug development programs, we are ready to provide the support you need. Contact us today to request samples and discover how our advanced manufacturing capabilities can drive efficiency and reliability in your nucleoside supply chain.
