Advanced Stereospecific Synthesis of Paroxetine Intermediates for Commercial Scale-Up
Advanced Stereospecific Synthesis of Paroxetine Intermediates for Commercial Scale-Up
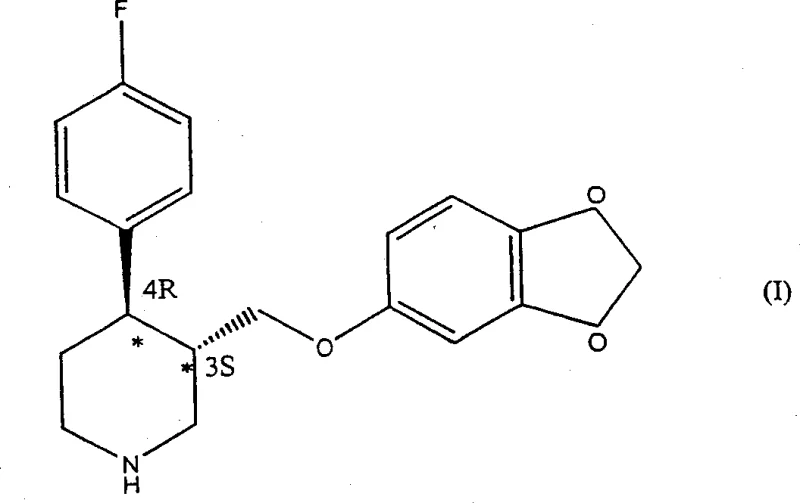
The pharmaceutical industry constantly seeks robust, scalable, and economically viable routes for the production of high-value Active Pharmaceutical Ingredients (APIs), particularly those requiring precise stereochemical control. Paroxetine, a widely prescribed selective serotonin reuptake inhibitor (SSRI), exemplifies this challenge due to its two chiral centers at the 3 and 4 positions of the piperidine ring. Among the four possible stereoisomers, only the (3S, 4R) configuration, also known as 4R-trans-paroxetine, possesses the desired pharmacological activity. Patent CN1341113A introduces a groundbreaking methodology that bypasses the inefficiencies of traditional resolution techniques by employing a highly stereospecific catalytic hydrogenation strategy. This approach ensures the direct formation of intermediates enriched with the critical 4R-isomeric component, which are subsequently converted into paroxetine in quantitative yields, representing a significant leap forward in process chemistry efficiency.
Historically, the synthesis of paroxetine has been plagued by the inherent limitations of resolving racemic mixtures. Conventional methods often involve the formation of intermediate derivatives which must then be separated into optical isomers through crystallization with optically active salts or enzymatic resolution. For instance, previous art describes separating derivatives of formula (II) into two optical isomers, requiring independent synthetic routes for each, which drastically complicates the manufacturing workflow. Furthermore, enzymatic methods, while stereospecific, suffer from the high cost and instability of enzymes, alongside slow reaction rates that demand precise pH and temperature control. These legacy processes inevitably result in the discard or separate reprocessing of approximately half of the synthesized material, leading to substantial economic loss and reduced overall throughput for manufacturers.
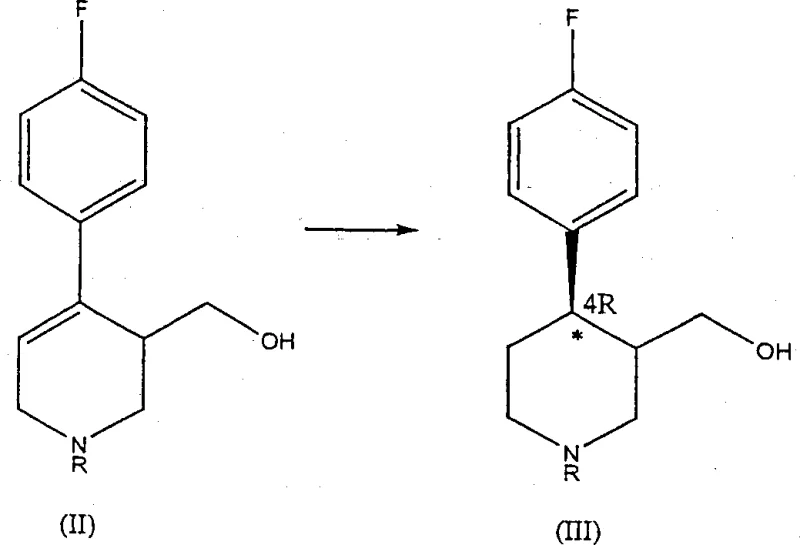
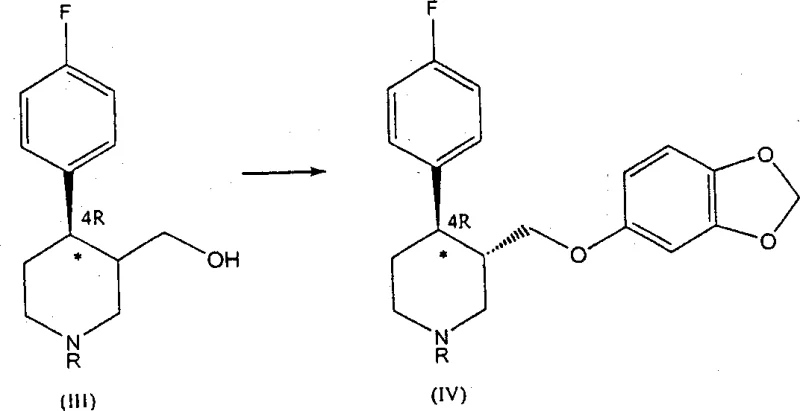
In stark contrast, the novel approach detailed in the patent data utilizes a transition metal-catalyzed asymmetric hydrogenation to establish the crucial chirality early in the synthesis. By treating N-substituted derivatives of 4-(p-fluorophenyl)-3-hydroxymethyl-1,2,3,6-tetrahydropyridine with a chiral catalyst, the process directly yields compounds enriched in the 4R-enantiomer. This eliminates the need for tedious resolution steps and allows for the utilization of both cis and trans isomers of the hydrogenated product in subsequent steps, as the stereochemistry at the 4-position is securely locked. The reaction sequence proceeds through hydrogenation, followed by derivatization of the hydroxyl group and nucleophilic substitution with sesamol, finally concluding with N-dealkylation. This streamlined pathway not only simplifies the operational complexity but also maximizes atom economy by retaining the desired stereochemical configuration throughout the synthesis.
Mechanistic Insights into Ru-BINAP Catalyzed Asymmetric Hydrogenation
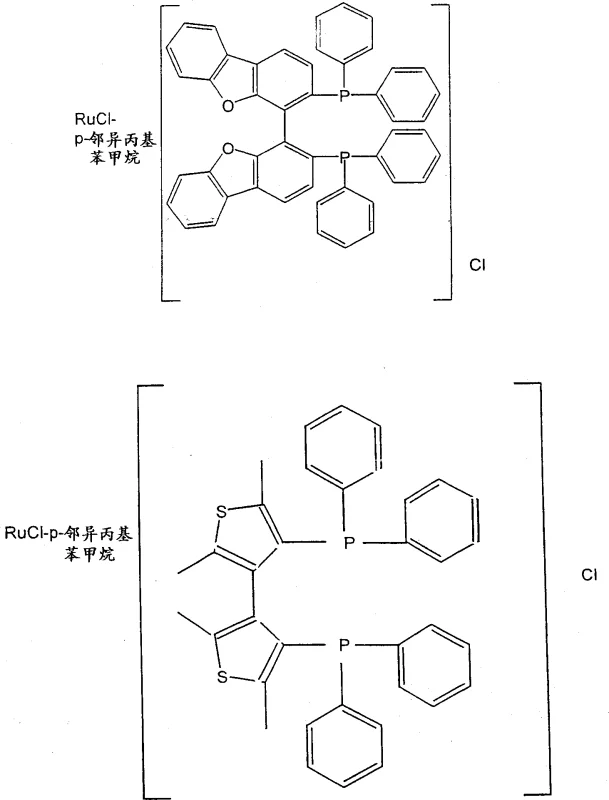
The core of this technological advancement lies in the sophisticated coordination chemistry employed during the hydrogenation step. The catalysts utilized belong to the class of transition metal complexes featuring chiral diphosphine ligands, with Ruthenium, Iridium, and Rhodium being the preferred metals. Specifically, complexes such as {RuCl(p-cymene)[(S)-BINAP]}Cl coordinate the metal center with the chiral ligand to create a rigid, chiral environment around the catalytic site. When the substrate, a tetrahydropyridine derivative, approaches this complex, the steric and electronic properties of the ligand dictate the face of the double bond that undergoes reduction. This interaction ensures that the hydrogen atoms are added in a highly stereospecific manner, favoring the formation of the 4R-absolute configuration at the piperidine ring's 4-position carbon atom. The result is a product mixture where the total enantiomeric excess (ee) can exceed 95%, and in optimized conditions using solvents like isopropanol, values greater than 99% are achievable.
Following the establishment of chirality, the synthesis proceeds through a carefully controlled derivatization and substitution sequence to install the sesamol moiety. The hydroxymethyl group on the 4R-enriched piperidine intermediate is first activated by converting it into a good leaving group, such as a mesylate or tosylate, using reagents like methanesulfonyl chloride in the presence of a base. This activation is critical for the subsequent nucleophilic displacement. The activated intermediate is then reacted with sesamol (3,4-methylenedioxyphenol) in an alkaline medium, typically using an alcoholic solvent like 4-methylpentan-2-ol. This nucleophilic substitution occurs with retention of the stereochemical integrity at the chiral centers, yielding the trans-diastereomer enriched in the 4R-enantiomer. The process is designed such that even if a mixture of cis and trans isomers enters this stage, the reaction conditions favor the formation and isolation of the pharmacologically active trans-form, further purifying the stream before the final deprotection step.
How to Synthesize 4R-Paroxetine Intermediate Efficiently
The synthesis of high-purity paroxetine intermediates via this route requires precise control over reaction parameters to maintain the high enantiomeric excess achieved during the initial hydrogenation. The process begins with the preparation of the tetrahydropyridine starting material, which can be efficiently synthesized via a Prins reaction involving 1-alkyl-4-(p-fluorophenyl)-1,2,5,6-tetrahydropyridine and formaldehyde. Once the substrate is prepared, the critical hydrogenation step is conducted under moderate hydrogen pressure (5-15 atmospheres) and elevated temperatures (100-130°C) in a solvent system that supports catalyst stability and selectivity, such as isopropanol or dichloromethane. The choice of the specific chiral ligand enantiomer, such as (S)-BINAP, is determined through screening to ensure it correlates with the desired 4R-product configuration. Following hydrogenation, the crude mixture undergoes activation and substitution without the need for intermediate isomer separation, leveraging the robustness of the catalytic step to deliver a stream ready for final dealkylation.
- Perform asymmetric hydrogenation of the tetrahydropyridine precursor using a chiral Ruthenium-BINAP complex catalyst to generate the 4R-enriched piperidine derivative.
- Convert the hydroxymethyl group into a leaving group (such as mesylate or tosylate) followed by nucleophilic substitution with sesamol to form the ether linkage.
- Execute N-dealkylation of the resulting intermediate to remove the protecting group and yield the final pharmacologically active paroxetine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this stereospecific catalytic process offers transformative benefits regarding cost structure and supply reliability. Traditional resolution-based methods inherently cap the maximum theoretical yield at 50% for the desired isomer unless complex recycling loops are implemented, which adds significant operational expenditure. By shifting to a catalytic asymmetric synthesis, the process effectively doubles the potential yield from the same amount of starting raw materials, as the chirality is induced rather than separated. This fundamental shift in process chemistry translates directly into substantial cost reductions in API manufacturing, as the consumption of expensive chiral resolving agents and the disposal costs associated with unwanted isomers are virtually eliminated. Furthermore, the use of robust transition metal catalysts that function at moderate pressures reduces the capital expenditure required for specialized high-pressure reactor infrastructure.
Supply chain reliability is significantly enhanced by the scalability and robustness of this synthetic route. The reaction conditions described, operating at temperatures between 100°C and 130°C and pressures around 10 atmospheres, are well within the standard operating envelopes of most multipurpose chemical manufacturing facilities. This compatibility means that the process can be scaled from pilot plant quantities to multi-ton commercial production without requiring bespoke engineering solutions or exotic equipment. Additionally, the catalysts employed, such as Ruthenium-BINAP complexes, can often be used at low catalyst-to-substrate ratios while maintaining high turnover numbers, reducing the dependency on scarce precious metals and mitigating supply risks associated with volatile metal markets. The ability to source common solvents like isopropanol and toluene further stabilizes the supply chain against raw material fluctuations.
From an environmental and regulatory compliance perspective, this methodology aligns perfectly with modern green chemistry principles and stringent waste disposal regulations. By avoiding the generation of large quantities of unwanted stereoisomers, the process drastically reduces the chemical load on waste treatment facilities. The elimination of enzymatic steps removes the variability and biological waste associated with fermentation or biocatalysis, simplifying the validation and quality control protocols required for regulatory approval. The high selectivity of the reaction ensures that the impurity profile of the final intermediate is clean and predictable, facilitating easier purification and reducing the risk of batch failures due to out-of-specification impurities. This predictability is crucial for maintaining continuous supply to downstream formulation partners and ensuring uninterrupted market availability of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced paroxetine synthesis route. These insights are derived directly from the patented experimental data and process descriptions, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding the nuances of catalyst selection, solvent effects, and downstream processing is essential for R&D teams planning technology transfer and for procurement teams assessing the long-term viability of this supply source.
Q: How does this process improve upon traditional resolution methods?
A: Unlike traditional methods that require resolving racemic mixtures and discarding up to 50% of unwanted isomers, this catalytic asymmetric hydrogenation directly generates the desired 4R-enantiomer with high enantiomeric excess (ee > 95%), significantly reducing material waste and processing steps.
Q: What catalysts are suitable for this stereospecific hydrogenation?
A: The process utilizes transition metal complexes of chiral diphosphine ligands, specifically Ruthenium complexes such as {RuCl(p-cymene)[(S)-BINAP]}Cl or RuHCl[(S)-BINAP]2, which provide the necessary chiral environment for high stereoselectivity.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction conditions operate at moderate pressures (5-15 atm) and temperatures (100-130°C) using common solvents like isopropanol, making it highly suitable for commercial scale-up without requiring extreme high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paroxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic chemistry described in patent CN1341113A can be successfully translated into a robust industrial process. We understand that high-purity pharmaceutical intermediates demand rigorous quality control; therefore, our facilities are equipped with state-of-the-art analytical instrumentation and stringent purity specifications to guarantee that every batch meets the exacting standards required for global regulatory submissions. Our commitment to technical excellence ensures that the high enantiomeric excess achieved in the lab is maintained consistently at scale.
We invite you to collaborate with us to optimize your supply chain for paroxetine and related antidepressant intermediates. By leveraging our expertise in asymmetric catalysis and process intensification, we can help you achieve significant efficiencies and cost savings. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us engineer the bottleneck out of your supply chain together.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
