Advanced Biocatalytic Synthesis of Paroxetine Intermediates Using Ionic Liquids for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing critical antidepressant intermediates, and patent CN101974604B represents a significant breakthrough in this domain. This intellectual property discloses a sophisticated method for preparing paroxetine intermediates through enzymatic resolution within an ionic liquid medium, specifically targeting the production of (3S, 4R)-4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine. Unlike traditional synthetic routes that rely heavily on hazardous organic solvents and harsh chemical conditions, this biocatalytic approach utilizes the unique physicochemical properties of ionic liquids to create a "green" reaction environment. The technology addresses the growing global demand for paroxetine, a leading serotonin reuptake inhibitor, by offering a pathway that combines high stereoselectivity with environmental responsibility. For R&D directors and process chemists, this patent offers a compelling alternative to legacy methods, promising not only superior product quality with optical purity reaching up to 98 percent but also a streamlined workflow that minimizes downstream purification burdens. The integration of biocatalysis with advanced solvent engineering marks a pivotal shift towards more sustainable API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of paroxetine intermediates has been plagued by significant technical and environmental challenges that hinder scalable and cost-effective production. Early methods, such as those described in patents US3912743 and US4007196, relied on Grignard reactions using arecoline, a raw material known for its strong irritancy and toxicity, making it unsuitable for modern large-scale manufacturing due to safety concerns and supply chain instability. Subsequent attempts at chemical resolution, detailed in patents like EP0223334 and US4902801, utilized (-)-di-p-toluoyl tartaric acid in acetone or toluene systems. These processes suffered from inherently low efficiency, with total yields often stagnating between 34 percent and 40 percent, necessitating the consumption of vast quantities of organic solvents and resolving agents. Furthermore, the reliance on volatile organic compounds (VOCs) like toluene and acetonitrile in earlier enzymatic attempts introduced severe occupational health risks and environmental liabilities. The cumulative effect of these limitations is a manufacturing process characterized by high waste generation, elevated raw material costs, and complex regulatory compliance hurdles regarding solvent residues and worker safety.
The Novel Approach
The innovative methodology presented in CN101974604B fundamentally reengineers the synthesis landscape by replacing volatile organic solvents with designer ionic liquids. This novel approach leverages the tunable nature of ionic liquids, such as [BMIm][PF6] or [BMIm][BF4], which act as both solvent and stabilizer for the biocatalyst. By conducting the transesterification reaction of racemic 4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine with an acyl donor like vinyl acetate in this medium, the process achieves mild reaction conditions typically ranging from 15°C to 60°C. This shift eliminates the need for flammable and toxic solvents, drastically reducing the environmental footprint of the operation. Moreover, the ionic liquid matrix enhances the operational stability and stereoselectivity of the lipase enzyme, allowing for higher conversion rates and superior optical purity compared to organic solvent systems. The ability to recycle both the enzyme and the ionic liquid medium further distinguishes this method, offering a circular economy model that significantly lowers the cost of goods sold (COGS) over the lifecycle of the production campaign while ensuring a reliable supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Lipase-Catalyzed Transesterification in Ionic Liquids
The core of this technological advancement lies in the intricate interplay between the lipase enzyme and the ionic liquid medium, which creates a highly favorable microenvironment for stereoselective catalysis. In traditional organic solvents, enzymes often suffer from denaturation or reduced flexibility, leading to diminished activity and selectivity. However, ionic liquids possess a unique network of hydrogen bonds and electrostatic interactions that can stabilize the tertiary structure of the lipase, preserving its active conformation even under prolonged reaction conditions. The mechanism involves the lipase selectively recognizing and acylating one enantiomer of the racemic substrate, specifically targeting the hydroxyl group to form an ester, while leaving the desired (3S, 4R)-enantiomer unreacted or reacting it at a significantly different rate depending on the specific kinetic resolution strategy employed. The ionic liquid facilitates this by solubilizing the hydrophobic substrate effectively while maintaining a hydration shell around the enzyme that is critical for its catalytic function. This balance ensures that the mass transfer limitations often seen in heterogeneous biocatalytic systems are minimized, allowing for efficient contact between the substrate, the acyl donor, and the catalytic sites.
Furthermore, the control of impurities and byproducts is intrinsically linked to the specificity of the enzymatic reaction and the stability of the medium. Chemical resolution methods often generate diastereomeric salts that require energy-intensive crystallization and neutralization steps, introducing opportunities for impurity carryover. In contrast, the enzymatic route in ionic liquids produces byproducts that are easily separable due to the distinct polarity differences between the ionic phase and the extracted product phase. The high optical purity of up to 98 percent ee reported in the patent is a direct result of the enzyme's rigid active site geometry, which sterically hinders the incorrect enantiomer from binding effectively. Additionally, the absence of reactive chemical resolving agents eliminates the formation of salt-based impurities, simplifying the downstream purification process. The use of vinyl acetate as an acyl donor further drives the reaction equilibrium forward by generating acetaldehyde as a byproduct, which does not interfere with the enzyme, thus ensuring high conversion efficiency without compromising the stereochemical integrity of the final paroxetine intermediate.
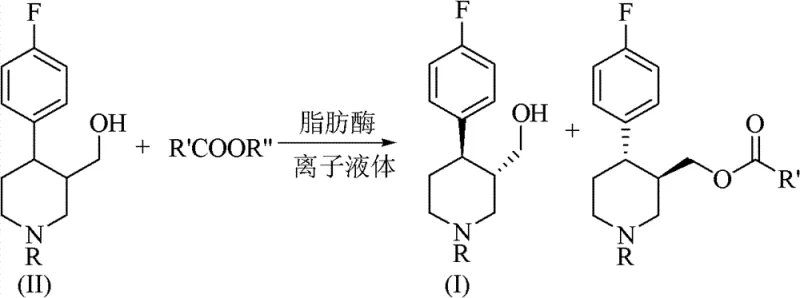
How to Synthesize (3S, 4R)-4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the ionic liquid system. The process begins with the preparation of a homogeneous reaction mixture where the racemic substrate is dissolved in the selected ionic liquid at concentrations optimized for mass transfer, typically between 10 mmol/L and 150 mmol/L. The choice of lipase is critical, with Candida antarctica Lipase B demonstrating superior performance in terms of both activity and enantioselectivity in this specific medium. Once the acyl donor is introduced, the system is maintained under controlled agitation to ensure uniform suspension of the biocatalyst, although the ionic liquid often allows for a quasi-homogeneous phase that enhances reaction kinetics. Monitoring the reaction progress via HPLC is essential to determine the optimal endpoint, balancing conversion rate with optical purity, as over-reaction can lead to decreased enantiomeric excess. The detailed standardized synthesis steps, including specific workup procedures and extraction protocols using n-hexane and isopropanol, are outlined below to ensure reproducibility and compliance with GMP standards.
- Prepare the reaction system by dissolving racemic 4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine in an ionic liquid medium such as [BMIm][PF6] at a concentration of 10-150 mmol/L.
- Add the acyl donor, preferably vinyl acetate, at a molar ratio of 1: 1 to 1:20 relative to the substrate, and introduce the lipase catalyst, such as Candida antarctica Lipase B.
- Maintain the reaction mixture at 15-60°C with stirring at 100-250 rpm for 2-24 hours, then separate the enzyme by centrifugation and extract the product using n-hexane and isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-based biocatalytic process translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of toxic and flammable organic solvents like toluene and acetonitrile significantly reduces the regulatory burden associated with hazardous material storage, transport, and disposal. This shift not only mitigates the risk of supply chain disruptions caused by environmental compliance audits but also lowers the overall cost of waste management, as ionic liquids can be recycled and reused multiple times without significant degradation. The mild reaction conditions, operating at temperatures as low as 30°C, reduce energy consumption compared to high-temperature chemical processes, contributing to a lower carbon footprint and reduced utility costs. Furthermore, the high stereoselectivity of the enzyme minimizes the need for extensive chromatographic purification or recrystallization steps, leading to higher overall throughput and reduced cycle times. These factors collectively enhance the reliability of the supply chain by creating a more robust and resilient manufacturing process that is less susceptible to raw material volatility and regulatory changes.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reusability of the catalytic system and the reduction in solvent consumption. Unlike traditional chemical resolution which consumes stoichiometric amounts of expensive resolving agents, the enzymatic method uses catalytic amounts of lipase which can be recovered. Additionally, the ionic liquid medium does not evaporate, allowing for near-quantitative recovery and reuse, which drastically cuts down on raw material procurement costs over time. The simplified downstream processing, resulting from high selectivity and easy phase separation, further reduces labor and equipment usage, leading to substantial cost savings in the overall production budget without compromising on quality.
- Enhanced Supply Chain Reliability: Dependence on hazardous solvents often introduces fragility into the supply chain due to strict transportation regulations and potential shortages of specialty chemicals. By transitioning to a greener solvent system, manufacturers can mitigate these risks and ensure a more consistent flow of materials. The stability of the ionic liquid also means that the reaction is less sensitive to minor fluctuations in environmental conditions, ensuring consistent batch-to-batch quality. This reliability is crucial for meeting the stringent delivery schedules of multinational pharmaceutical clients who require uninterrupted supply of critical intermediates for their own API production lines, thereby strengthening long-term partnerships.
- Scalability and Environmental Compliance: Scaling biocatalytic processes can often be challenging, but the use of ionic liquids simplifies this by providing a stable medium that supports enzyme activity even at larger volumes. The non-volatile nature of the solvent eliminates the need for complex solvent recovery distillation columns, reducing capital expenditure for plant expansion. From an environmental perspective, the process aligns perfectly with increasingly stringent global regulations on VOC emissions and waste discharge. This proactive compliance ensures that the manufacturing facility remains operational without the threat of shutdowns due to environmental violations, securing the long-term viability of the production asset and protecting the brand reputation of the supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to established industry standards. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this green chemistry approach into their existing manufacturing portfolios. The answers highlight the specific advantages in terms of purity, safety, and operational efficiency that distinguish this patent from prior art.
Q: How does the ionic liquid method improve optical purity compared to traditional chemical resolution?
A: Traditional chemical resolution using tartaric acid derivatives often yields only 34%-40% total recovery with significant solvent waste. The enzymatic method in ionic liquids achieves optical purity up to 98% ee by leveraging the high stereoselectivity of lipases in a stable, non-volatile medium that preserves enzyme activity.
Q: What are the environmental advantages of using ionic liquids over organic solvents like toluene?
A: Unlike toluene or acetonitrile, which are toxic, flammable, and volatile, ionic liquids have negligible vapor pressure and are non-flammable. This drastically reduces VOC emissions and fire hazards, while allowing for the recycling of both the solvent and the biocatalyst, aligning with green chemistry principles.
Q: Can the lipase and ionic liquid be reused in this process?
A: Yes, a key advantage of this protocol is the reusability of the reaction components. The ionic liquid provides a stable environment that maintains lipase operational stability, allowing for multiple cycles of catalysis without significant loss of activity, thereby reducing long-term material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paroxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic resolution technology described in CN101974604B for the production of high-value antidepressant intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of ionic liquid chemistry, including specialized containment and recycling systems that maximize the economic and environmental benefits of this process. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (3S, 4R)-4-(4-fluorophenyl)-3-hydroxymethyl-1-R-piperidine meets the highest international standards for optical purity and chemical identity. Our commitment to green chemistry aligns with the global pharmaceutical industry's push for sustainability, making us an ideal partner for forward-thinking drug developers.
We invite you to collaborate with our technical team to explore how this advanced biocatalytic route can optimize your supply chain and reduce your overall manufacturing costs. By engaging with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you secure a reliable, high-quality, and sustainable source of paroxetine intermediates that will support your clinical and commercial goals with confidence and precision.
