Advanced Enzymatic Synthesis of Chiral Bridged Ring Compounds for Commercial Scale-up
Introduction to Next-Generation Bridged Ring Synthesis
The pharmaceutical industry constantly seeks more efficient pathways to construct complex chiral scaffolds, particularly bridged ring systems which serve as critical cores in numerous bioactive molecules. Patent CN111194304B introduces a transformative preparation method for these bridged ring compounds, shifting the paradigm from traditional racemic synthesis followed by resolution to a streamlined enzymatic kinetic resolution strategy. This innovation addresses the longstanding challenge of obtaining single-configuration intermediates with high optical purity early in the synthetic sequence. By leveraging specific biocatalysts such as lipases and esterases, the method achieves enantiomeric excess (ee) values exceeding 40%, with optimized embodiments demonstrating ee values greater than 90%. This technical breakthrough not only simplifies the synthetic route but also lays a robust foundation for the cost-effective manufacturing of high-purity pharmaceutical intermediates, ensuring that downstream drug development processes are supported by reliable and scalable supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral bridged ring compounds relied heavily on non-selective chemical transformations that produced racemic mixtures. As illustrated in the prior art, the conventional approach typically involves the double sulfonation of a starting material containing bridged ring fragments, followed by a series of steps to generate a racemic intermediate. 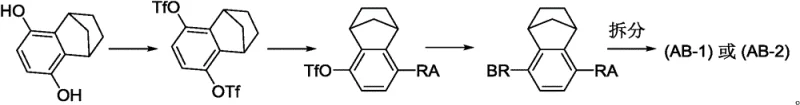 . The critical bottleneck in this traditional workflow is the resolution step, where the racemic compound must be separated into its individual enantiomers. Since resolution theoretically caps the maximum yield at 50%, half of the valuable material is discarded or requires energy-intensive recycling processes. Furthermore, the early introduction of sulfonating groups can lead to unavoidable disubstituted byproducts, complicating purification and increasing the overall cost of goods. These inefficiencies result in significant material loss across multiple steps prior to reaching the final active pharmaceutical ingredient, creating substantial economic and operational burdens for large-scale manufacturing.
. The critical bottleneck in this traditional workflow is the resolution step, where the racemic compound must be separated into its individual enantiomers. Since resolution theoretically caps the maximum yield at 50%, half of the valuable material is discarded or requires energy-intensive recycling processes. Furthermore, the early introduction of sulfonating groups can lead to unavoidable disubstituted byproducts, complicating purification and increasing the overall cost of goods. These inefficiencies result in significant material loss across multiple steps prior to reaching the final active pharmaceutical ingredient, creating substantial economic and operational burdens for large-scale manufacturing.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a highly selective enzymatic hydrolysis to differentiate between enantiomers or pro-chiral centers at the diester stage. Instead of struggling with a racemic mixture late in the synthesis, this approach starts with a diester compound (Formula 02) and employs specific enzymes to selectively hydrolyze one ester group, yielding a chiral monoester (Formula 03) with high optical purity. 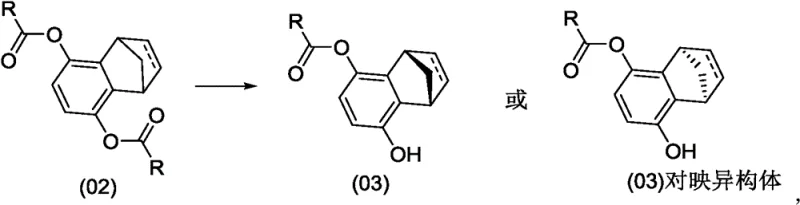 . This strategic shift allows chemists to establish the desired stereochemistry early in the process, thereby avoiding the inherent 50% yield penalty associated with classical resolution. The resulting monoester can then be easily converted into a monosulfonated intermediate, which serves as a versatile building block for coupling reactions with boronic acids or other fragments. By eliminating the need for early-stage racemic synthesis and late-stage separation, this novel approach drastically reduces waste, simplifies the impurity profile, and enhances the overall atom economy of the manufacturing process.
. This strategic shift allows chemists to establish the desired stereochemistry early in the process, thereby avoiding the inherent 50% yield penalty associated with classical resolution. The resulting monoester can then be easily converted into a monosulfonated intermediate, which serves as a versatile building block for coupling reactions with boronic acids or other fragments. By eliminating the need for early-stage racemic synthesis and late-stage separation, this novel approach drastically reduces waste, simplifies the impurity profile, and enhances the overall atom economy of the manufacturing process.
Mechanistic Insights into Enzymatic Kinetic Resolution
The core of this technological advancement lies in the precise selection of biocatalysts that exhibit high stereoselectivity towards the bridged ring substrate. The patent details that the type of enzyme employed has a profound impact on the configuration of the product, enabling access to either enantiomer simply by switching the biocatalyst. For instance, using recombinant Escherichia coli esterase, Candida rugosa lipase, or Pseudomonas fluorescens lipase tends to yield the compound of Formula 03, whereas employing Lipase Novozymes 435, Aspergillus fungal protease, or Bacillus protease preferentially produces the enantiomer of Formula 03. This flexibility is crucial for medicinal chemistry programs where specific stereochemistry is required for biological activity. The reaction mechanism involves the enzyme actively binding to the diester substrate within a buffered aqueous-organic solvent system, where the pH is carefully controlled between 3.0 and 12.0 to maintain enzyme stability and activity. The hydrolysis occurs under mild thermal conditions, typically ranging from 0°C to 80°C, which preserves the integrity of sensitive functional groups on the bridged ring scaffold.
Furthermore, the process demonstrates remarkable robustness regarding impurity control and substrate scope. The enzymatic reaction tolerates a wide variety of ester groups (R groups), including straight-chain alkyls, cycloalkyls, aryls, and even halogen-substituted chains, without compromising the enantiomeric excess. Experimental data within the patent shows that ee values can consistently exceed 60%, 70%, or even 80% depending on the specific enzyme-substrate pairing. For example, the hydrolysis of specific diesters using Thermomyces lanuginosa lipase yielded products with ee values as high as 97%. This high level of stereocontrol minimizes the formation of unwanted diastereomers and enantiomeric impurities, which are often difficult to remove in later stages of drug synthesis. The ability to achieve such high optical purity directly from the enzymatic step significantly reduces the burden on downstream purification technologies like chromatography, making the process more viable for commercial production.
How to Synthesize Chiral Bridged Ring Monoesters Efficiently
Implementing this enzymatic route requires careful attention to reaction parameters to maximize yield and optical purity. The process begins with the preparation of the diester precursor, followed by the critical biocatalytic step where the enzyme selectively cleaves one ester bond. The detailed standardized synthesis steps below outline the specific conditions, reagent ratios, and workup procedures necessary to replicate the high-performance results described in the patent documentation. Following these guidelines ensures that the resulting monoester meets the stringent quality requirements needed for subsequent sulfonation and coupling reactions.
- Prepare the diester precursor (Compound 02) by reacting the corresponding diol (Compound 01) with acid chlorides or anhydrides in the presence of a base like triethylamine and a catalyst like DMAP.
- Conduct the enzymatic hydrolysis reaction by suspending Compound 02 in a buffered solution (pH 3.0-12.0) with a specific lipase or esterase (e.g., E. coli esterase or Novozymes 435) at temperatures between 0°C and 80°C.
- Isolate the resulting chiral monoester (Compound 03) via extraction and purification, achieving an enantiomeric excess (ee) value exceeding 40%, often reaching over 90% depending on the enzyme selected.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The primary value driver is the substantial improvement in material efficiency. By circumventing the theoretical 50% yield limit inherent in traditional racemic resolution, this method effectively doubles the potential output from the same amount of starting raw materials. This dramatic increase in atom economy translates directly into significant cost reductions in pharmaceutical intermediate manufacturing, as less feedstock is required to produce the same quantity of active chiral building blocks. Additionally, the elimination of complex resolution steps reduces the number of unit operations, lowering both capital expenditure on equipment and operational expenditures related to energy and labor.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for expensive chiral resolving agents and the associated loss of half the batch mass. Since the enzyme acts as a highly specific catalyst, it avoids the use of stoichiometric amounts of chiral auxiliaries or heavy metal catalysts often found in asymmetric chemical synthesis. This reduction in reagent costs, combined with the higher overall yield, leads to a drastically simplified cost structure. Furthermore, the mild reaction conditions reduce energy consumption for heating or cooling, contributing to lower utility costs per kilogram of product. The ability to recycle the unreacted diester or convert it further adds another layer of economic efficiency, ensuring that raw material investments are maximized throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Reliance on enzymatic processes enhances supply chain resilience by utilizing robust and commercially available biocatalysts. Unlike some specialized chiral ligands that may have long lead times or single-source risks, the lipases and esterases specified in the patent are widely produced by major enzyme suppliers. The process uses common organic solvents and buffer salts, which are readily accessible in the global chemical market, reducing the risk of supply disruptions. Moreover, the high selectivity of the reaction minimizes the generation of hard-to-remove impurities, which often cause delays in quality control and batch release. This predictability in production timelines ensures a more stable and continuous supply of critical intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this green chemistry approach aligns perfectly with modern sustainability goals. The use of biocatalysts operates under mild conditions, reducing the carbon footprint associated with high-temperature or high-pressure reactions. The aqueous-organic solvent systems are generally easier to treat and recycle compared to the harsh acidic or basic waste streams generated by traditional resolution methods. The high purity of the product reduces the need for extensive chromatographic purification, which is a major source of solvent waste in the pharmaceutical industry. Consequently, scaling this process from pilot plant to multi-ton commercial production is straightforward, with fewer environmental compliance hurdles and lower waste disposal costs, making it an attractive option for long-term manufacturing contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic bridged ring synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this technology into their existing pipelines.
Q: What is the primary advantage of the enzymatic method over traditional resolution?
A: Traditional methods involve synthesizing a racemic mixture and then resolving it, which theoretically limits the maximum yield to 50%. The enzymatic kinetic resolution described in CN111194304B allows for the direct production of the desired chiral monoester from a prochiral or racemic diester precursor with high optical purity, significantly improving material efficiency.
Q: Which enzymes are suitable for this transformation?
A: The patent specifies a wide range of biocatalysts including recombinant Escherichia coli esterase, Candida rugosa lipase, Pseudomonas fluorescens lipase, and Lipase Novozymes 435. The choice of enzyme dictates the stereochemical outcome, allowing access to either enantiomer of the target compound.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction conditions are mild, typically operating between 0°C and 80°C in common organic solvents or buffer systems. The use of robust enzymes and standard workup procedures like extraction and crystallization makes the process highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bridged Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are fully equipped to implement the advanced enzymatic kinetic resolution techniques described in CN111194304B, leveraging our state-of-the-art fermentation and chemical synthesis facilities to deliver bridged ring compounds with stringent purity specifications. Our rigorous QC labs utilize advanced analytical methods to verify enantiomeric excess and impurity profiles, guaranteeing that every batch meets the exacting standards required by global regulatory authorities.
We invite you to collaborate with us to optimize your supply chain for these vital pharmaceutical building blocks. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in enzymatic synthesis can drive value and reliability for your organization.
