Advanced Enzymatic Synthesis of Chiral Bridged Ring Compounds for Commercial Pharmaceutical Production
Advanced Enzymatic Synthesis of Chiral Bridged Ring Compounds for Commercial Pharmaceutical Production
The pharmaceutical industry constantly seeks robust methodologies for constructing complex chiral scaffolds, particularly bridged ring systems that serve as critical cores for bioactive molecules. Patent CN111194304A introduces a groundbreaking preparation method for these bridged ring compounds, leveraging biocatalytic precision to overcome longstanding synthetic hurdles. This technology represents a paradigm shift from traditional chemical resolution, offering a pathway to high optical purity intermediates with enantiomeric excess (ee) values exceeding 40%, and in optimized embodiments, surpassing 90%. For R&D directors and procurement specialists, this innovation signals a move towards more sustainable, cost-effective, and scalable manufacturing processes for high-value drug intermediates. By integrating enzymatic selectivity early in the synthetic route, manufacturers can bypass inefficient racemic separations, thereby securing a more reliable supply chain for complex pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of single-configuration bridged ring drugs involved arduous multi-step sequences that were inherently inefficient and costly. The conventional approach typically required the bis-sulfonation of a starting material containing the bridge ring segment, followed by a series of transformations to generate a racemic intermediate. This racemic mixture then necessitated a resolution step to isolate the desired enantiomer, a process theoretically capped at a maximum yield of 50%. 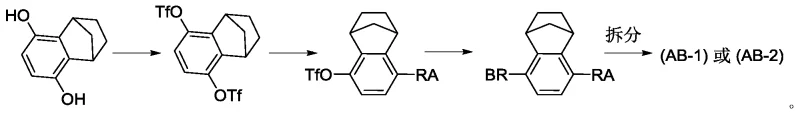 Furthermore, the presence of bis-sulfonate disubstituted byproducts was unavoidable during the ligation of molecular fragments, complicating purification and driving up waste disposal costs. These inefficiencies not only inflated the cost of goods sold (COGS) but also introduced significant supply chain risks due to the complexity of operations and the potential for batch-to-batch variability in optical purity.
Furthermore, the presence of bis-sulfonate disubstituted byproducts was unavoidable during the ligation of molecular fragments, complicating purification and driving up waste disposal costs. These inefficiencies not only inflated the cost of goods sold (COGS) but also introduced significant supply chain risks due to the complexity of operations and the potential for batch-to-batch variability in optical purity.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a strategic enzymatic desymmetrization or kinetic resolution of diester precursors to directly access chiral monoesters. Instead of resolving a fully formed racemic drug candidate late in the synthesis, this approach installs chirality at an earlier stage using highly selective biocatalysts. 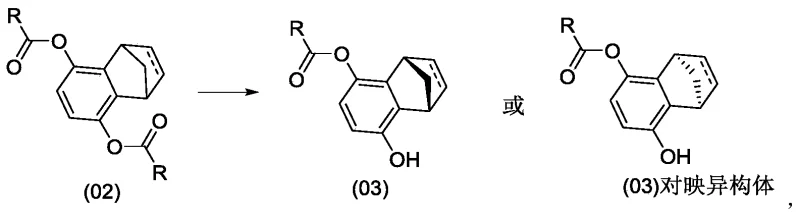 The process involves reacting a diester compound under the action of specific enzymes such as lipases or esterases to obtain a monoester compound with high optical purity. This monoester can then undergo sulfonation and subsequent coupling reactions to yield the final single-configuration drug or intermediate. By shifting the stereocontrol to the enzymatic step, the process eliminates the 50% yield ceiling of traditional resolution and avoids the formation of difficult-to-remove bis-sulfonated impurities, streamlining the entire production workflow.
The process involves reacting a diester compound under the action of specific enzymes such as lipases or esterases to obtain a monoester compound with high optical purity. This monoester can then undergo sulfonation and subsequent coupling reactions to yield the final single-configuration drug or intermediate. By shifting the stereocontrol to the enzymatic step, the process eliminates the 50% yield ceiling of traditional resolution and avoids the formation of difficult-to-remove bis-sulfonated impurities, streamlining the entire production workflow.
Mechanistic Insights into Enzyme-Catalyzed Stereoselective Hydrolysis
The core of this technological advancement lies in the precise mechanistic interaction between the enzyme active site and the prochiral or racemic diester substrate. The patent elucidates that the configuration of the product is heavily influenced by the specific class of enzyme employed. For instance, utilizing recombinant Escherichia coli esterase or Pseudomonas fluorescens lipase yields one specific enantiomer of the monoester, whereas switching to lipase Novozym 435 or Aspergillus fungal protease can invert the stereoselectivity to produce the opposite enantiomer. This tunability is crucial for R&D teams targeting specific biological activities that depend on absolute stereochemistry. The reaction proceeds under mild aqueous-organic biphasic conditions, where the enzyme selectively hydrolyzes one ester bond over the other, driven by steric and electronic complementarity within the enzyme pocket. This biological precision ensures that the resulting monoester possesses an ee value that can be rigorously controlled, often exceeding 70% or even 90% depending on the enzyme-substrate pairing and reaction parameters such as pH and temperature.
Furthermore, the mechanism allows for exceptional control over impurity profiles, a key concern for regulatory compliance. In traditional chemical hydrolysis, non-selective cleavage often leads to a mixture of diols, monoesters, and unreacted diesters that are challenging to separate. However, the enzymatic pathway described here demonstrates high chemoselectivity, primarily generating the target monoester while leaving the second ester group intact or hydrolyzing it at a negligible rate. The subsequent sulfonation step, converting the chiral monoester into a sulfonate intermediate, proceeds cleanly because the starting material is already enriched in a single configuration. 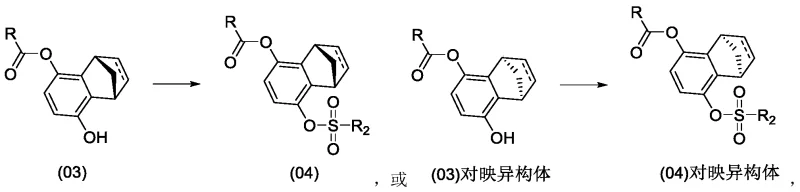 This minimizes the formation of di-sulfonated byproducts that plagued previous methods. The ability to fine-tune reaction conditions, such as maintaining a pH between 5.0 and 11.0 and temperatures from 0°C to 50°C, further suppresses side reactions, ensuring a high-purity output that simplifies downstream processing and crystallization steps.
This minimizes the formation of di-sulfonated byproducts that plagued previous methods. The ability to fine-tune reaction conditions, such as maintaining a pH between 5.0 and 11.0 and temperatures from 0°C to 50°C, further suppresses side reactions, ensuring a high-purity output that simplifies downstream processing and crystallization steps.
How to Synthesize Chiral Bridged Ring Monoesters Efficiently
Implementing this synthesis route requires careful attention to enzyme selection and reaction engineering to maximize both yield and optical purity. The process begins with the preparation of the diester substrate, typically achieved by reacting the corresponding diol with acyl chlorides or anhydrides in the presence of a base like triethylamine and a catalyst such as DMAP. Once the diester is secured, the critical enzymatic step is executed in a buffered system, potentially containing co-solvents like ethyl acetate or tetrahydrofuran to enhance substrate solubility. The choice of buffer, ranging from phosphate to glycine systems, and the precise control of pH are vital for maintaining enzyme activity and stability throughout the reaction course. Following the biocatalytic transformation, the crude monoester is isolated and can be directly subjected to sulfonation using reagents like methanesulfonyl chloride or p-toluenesulfonyl chloride.
- Prepare the diester substrate by reacting the bridged ring diol with acyl chlorides or anhydrides in the presence of a base and catalyst.
- Perform enzymatic hydrolysis using specific lipases or esterases in a buffered aqueous-organic solvent system to obtain the chiral monoester.
- Subject the resulting chiral monoester to sulfonation using sulfonyl chlorides or anhydrides to generate the active coupling intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic advantages regarding cost, reliability, and scalability. The shift from chemical resolution to enzymatic kinetic resolution fundamentally alters the economic model of producing these complex intermediates. By avoiding the inherent 50% material loss associated with resolving racemates, the overall material throughput is effectively doubled for the same amount of starting input. Moreover, the elimination of harsh chemical reagents and the reduction in purification steps lead to significant operational expenditure savings. The use of commercially available enzymes and standard solvents ensures that the supply chain is not dependent on exotic or proprietary catalysts that might face availability bottlenecks. This robustness enhances supply continuity, allowing manufacturers to plan long-term production schedules with greater confidence and reduced risk of disruption.
- Cost Reduction in Manufacturing: The enzymatic process drastically simplifies the synthetic route by removing the need for early-stage bis-sulfonation and late-stage resolution, which are both resource-intensive operations. The mild reaction conditions reduce energy consumption compared to high-temperature chemical processes, and the high selectivity minimizes waste generation, lowering disposal costs. Additionally, the ability to recycle or reuse enzyme preparations in certain immobilized formats can further drive down the variable costs per kilogram of product. This comprehensive reduction in process complexity and material waste results in a substantially lower cost of goods, making the final pharmaceutical product more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Reliance on biocatalysis leverages a mature and stable supply chain for enzymes, which are produced at industrial scales for various sectors. Unlike specialized transition metal catalysts that may be subject to geopolitical supply constraints or price volatility, lipases and esterases are readily accessible from multiple global vendors. The process tolerance to a wide range of solvents and buffers means that manufacturing facilities can utilize existing infrastructure without requiring expensive retrofits for high-pressure or cryogenic capabilities. This flexibility ensures that production can be maintained even if specific raw material sources face temporary shortages, as alternative solvents or buffer systems can be substituted without revalidating the entire process, thereby securing a steady flow of high-purity intermediates.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated effectively from gram-scale laboratory experiments to multi-kilogram pilot runs. The aqueous nature of the enzymatic step aligns perfectly with green chemistry principles, reducing the reliance on volatile organic compounds (VOCs) and hazardous reagents. This environmental compatibility simplifies regulatory approvals and permits, as the process generates less hazardous waste and effluent. For large-scale commercial production, this means faster time-to-market for new drugs utilizing this scaffold and a reduced environmental footprint. The ability to scale up while maintaining high enantiomeric excess ensures that quality remains consistent regardless of batch size, a critical factor for meeting the stringent demands of international pharmaceutical regulators.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patented process is essential for stakeholders evaluating its integration into their manufacturing portfolios. The following questions address common inquiries regarding the practical application, scope, and benefits of this enzymatic synthesis method. These answers are derived directly from the technical disclosures within the patent documentation, providing a clear picture of what can be achieved regarding purity, yield, and operational flexibility.
Q: What is the primary advantage of the enzymatic method over traditional resolution?
A: The enzymatic method avoids the theoretical 50% yield loss associated with traditional racemic resolution and eliminates the need for harsh bis-sulfonation steps early in the synthesis, significantly improving overall process efficiency and cost-effectiveness.
Q: Which enzymes are suitable for this transformation?
A: The patent specifies a wide range of biocatalysts including recombinant Escherichia coli esterase, Candida rugosa lipase, Pseudomonas fluorescens lipase, and Novozym 435, allowing for flexibility in optimizing stereoselectivity for specific substrates.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes mild reaction conditions (0°C to 80°C) and commercially available enzymes in standard solvent systems, making it highly amenable to commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bridged Ring Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis methods described in patent CN111194304A for producing high-value bridged ring compounds. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle biocatalytic processes with precision, maintaining stringent purity specifications and utilizing rigorous QC labs to verify enantiomeric excess and chemical identity at every stage. We are committed to delivering reliable pharmaceutical intermediate supplies that meet the highest global standards, leveraging our technical expertise to optimize yields and minimize costs for our partners.
We invite you to collaborate with us to unlock the full commercial potential of your bridged ring drug candidates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this enzymatic route can improve your margins. Please contact us to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us help you secure a sustainable and efficient supply chain for your next-generation therapeutics.
