Revolutionizing Nintedanib Intermediate Production: A Safe, Scalable, and High-Purity Synthetic Route
Revolutionizing Nintedanib Intermediate Production: A Safe, Scalable, and High-Purity Synthetic Route
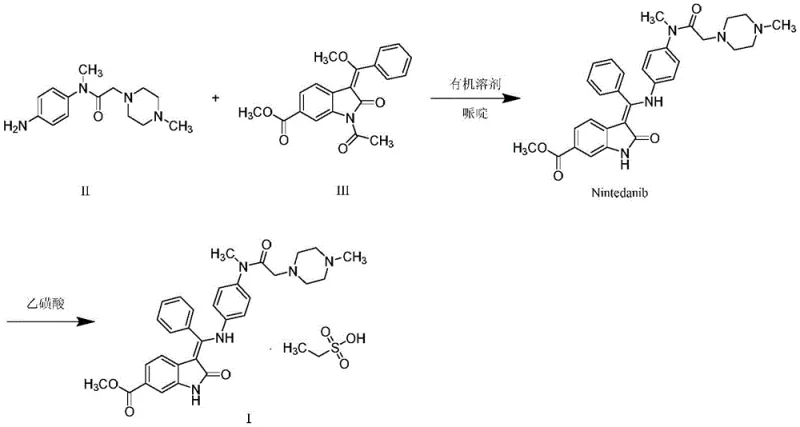
The global demand for idiopathic pulmonary fibrosis (IPF) treatments has surged, placing Nintedanib (marketed as OFEV®) at the forefront of respiratory medicine. However, the manufacturing of this complex tyrosine kinase inhibitor relies heavily on the efficient production of its key building blocks. Patent CN111777576A introduces a transformative methodology for synthesizing the critical intermediate, N-(4-aminophenyl)-N-methyl-2-(4-methylpiperazin-1-yl)acetamide (Compound II). This patent addresses long-standing bottlenecks in the supply chain by replacing hazardous high-pressure hydrogenation and unstable chemical reductions with mild, ambient-condition protocols. For pharmaceutical manufacturers, this represents a pivotal shift towards safer, more robust, and cost-effective production capabilities that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of the nitro precursor (Compound IV) to the amine (Compound II) has been plagued by significant operational hazards and inefficiencies. Traditional literature, including patents like WO2012068441, predominantly relies on palladium-carbon catalyzed hydrogenation under high pressure. This approach necessitates expensive autoclave reactors and rigorous safety protocols to prevent catastrophic explosions caused by hydrogen leakage or air ingress. Furthermore, alternative chemical reduction methods utilizing sodium dithionite (insurance powder) present severe environmental and safety liabilities; this reagent is highly flammable upon contact with moisture and generates toxic sulfur dioxide and hydrogen sulfide gases, complicating waste treatment and posing acute health risks to plant personnel. These legacy methods create substantial barriers to entry for generic manufacturers and inflate the cost of goods sold due to specialized equipment requirements and hazardous waste disposal fees.
The Novel Approach
The technology disclosed in CN111777576A fundamentally reengineers this reduction step by introducing three distinct, mild pathways that operate at atmospheric pressure and moderate temperatures. The primary innovation involves the use of trichlorosilane coupled with organic bases, or alternatively, transfer hydrogenation systems using ammonium formate or hydrazine hydrate with iron catalysts. These methods eliminate the need for gaseous hydrogen and high-pressure vessels entirely, allowing the reaction to proceed safely in standard glass-lined or stainless steel reactors. By shifting the reaction conditions to a range of 0°C to 60°C, the process not only enhances operator safety but also significantly improves the impurity profile, yielding Compound II with purity levels consistently above 99.6%. This strategic pivot from hazardous legacy chemistry to controlled, mild reduction creates a far more reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Trichlorosilane-Mediated Nitro Reduction
The core of the first novel technical solution lies in the activation of the nitro group via silicon-hydrogen bond cleavage in the presence of a tertiary amine base. In this mechanism, trichlorosilane (HSiCl3) acts as a potent hydride donor, facilitated by the coordination of the organic base, such as triethylamine or N,N-diisopropylethylamine. The base serves a dual purpose: it scavenges the hydrochloric acid generated during the reaction to prevent the protonation of the sensitive piperazine nitrogen, and it activates the silane species for nucleophilic attack on the electron-deficient nitro group. This synergistic interaction allows for the stepwise reduction of the nitro functionality through nitroso and hydroxylamine intermediates directly to the primary amine without over-reduction or cleavage of other sensitive functional groups within the molecule. The reaction kinetics are highly favorable at room temperature, ensuring rapid conversion while minimizing thermal stress on the product.
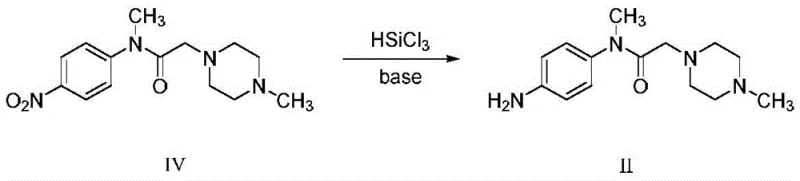
Impurity control is rigorously maintained through the specific choice of solvents and crystallization protocols outlined in the patent. The use of chlorinated solvents like dichloromethane or chloroform provides excellent solubility for the starting material while allowing for precise control over the exotherm during silane addition. Post-reaction, the introduction of a reversed-phase solvent, such as methyl tert-butyl ether or n-hexane, induces selective crystallization of the target amine salt or free base, effectively excluding unreacted starting materials and silicon-containing byproducts. This crystallization-driven purification is superior to traditional column chromatography or extensive aqueous workups, as it locks the molecular lattice into a high-purity form early in the process. Consequently, the final isolated solid exhibits an HPLC purity of approximately 99.66%, demonstrating that the mechanistic pathway inherently suppresses the formation of azo-coupling byproducts or dehalogenated impurities often seen in metal-catalyzed reductions.
How to Synthesize Nintedanib Intermediate Efficiently
Implementing this advanced synthesis requires precise adherence to stoichiometric ratios and temperature controls to maximize yield and safety. The process begins with the dissolution of the nitro compound in a dry chlorinated solvent, followed by the controlled addition of the silane reducing agent and base. Detailed standard operating procedures regarding reagent addition rates, quenching protocols, and crystallization parameters are essential for reproducibility. For a comprehensive breakdown of the exact experimental steps, reagent quantities, and workup procedures validated in the patent examples, please refer to the technical guide below.
- Dissolve the nitro precursor (Compound IV) in a suitable organic solvent such as dichloromethane or chloroform under inert atmosphere.
- Add trichlorosilane and an organic base like triethylamine slowly while maintaining the temperature between 0°C and 50°C to initiate reduction.
- Upon completion, precipitate the product by adding a reversed-phase solvent like methyl tert-butyl ether, followed by filtration and vacuum drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into enhanced operational resilience and reduced total cost of ownership. By eliminating the dependency on high-pressure hydrogenation infrastructure, manufacturers can utilize existing standard reactor trains, thereby avoiding massive capital expenditure on specialized autoclaves and associated safety systems. The removal of hazardous reagents like sodium dithionite also simplifies regulatory compliance and waste management logistics, removing the need for costly scrubbing systems designed to handle sulfur emissions. This streamlining of the manufacturing process ensures a more consistent supply of high-quality intermediates, mitigating the risk of production stoppages due to safety audits or equipment maintenance.
- Cost Reduction in Manufacturing: The transition to ambient pressure conditions significantly lowers energy consumption and equipment depreciation costs. Without the need for high-pressure containment, the facility can operate with lower insurance premiums and reduced maintenance schedules for reactor vessels. Furthermore, the high selectivity of the trichlorosilane or transfer hydrogenation methods reduces the loss of valuable starting materials to side reactions, improving the overall mass balance and lowering the effective cost per kilogram of the active intermediate. The ability to use common, commercially available reagents instead of specialized catalysts or gases further stabilizes raw material pricing against market volatility.
- Enhanced Supply Chain Reliability: Reliance on gaseous hydrogen often introduces logistical vulnerabilities, requiring specialized transport and storage facilities that can be subject to regulatory delays. By switching to liquid reagents like trichlorosilane or ammonium formate, the supply chain becomes more robust and less susceptible to external disruptions. The simplified process flow also shortens the batch cycle time, allowing for increased throughput and faster response to market demand fluctuations. This agility is crucial for maintaining continuous production schedules for life-saving medications like Nintedanib, ensuring that downstream API synthesis is never halted by intermediate shortages.
- Scalability and Environmental Compliance: The mild reaction conditions facilitate seamless scale-up from pilot plant to multi-ton commercial production without the engineering challenges associated with heat and mass transfer in high-pressure systems. The reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, reducing the carbon footprint of the manufacturing process. This eco-friendly profile not only future-proofs the production site against tightening emission standards but also enhances the brand reputation of the pharmaceutical company as a responsible manufacturer committed to sustainable chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is vital for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: What are the safety advantages of the new trichlorosilane reduction method over traditional hydrogenation?
A: The new method operates at atmospheric pressure and moderate temperatures (0-50°C), eliminating the need for high-pressure autoclaves and flammable hydrogen gas, thereby drastically reducing explosion risks and infrastructure costs.
Q: How does this process improve the purity profile of the Nintedanib intermediate?
A: By avoiding harsh conditions and unstable reagents like sodium dithionite, the novel routes minimize side reactions and degradation, consistently achieving HPLC purity levels exceeding 99.6%.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the process uses common reagents and standard equipment without specialized high-pressure vessels, making it highly scalable and economically viable for multi-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nintedanib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the complexity of kinase inhibitor synthesis demands a partner with deep technical expertise and unwavering commitment to quality. Our R&D team has extensively analyzed the pathways described in CN111777576A and possesses the capability to optimize these routes for maximum efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications required for global regulatory filings. Our rigorous QC labs are equipped to detect and quantify trace impurities, guaranteeing that the Nintedanib intermediates we supply provide a flawless foundation for your final API synthesis.
We invite you to collaborate with us to leverage these advanced manufacturing technologies for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can drive value and security into your Nintedanib production program.
