Mastering Regioselectivity: A Commercial Breakthrough in 2-Hydroxyquinoxaline Manufacturing
In the highly competitive landscape of pharmaceutical intermediate manufacturing, the ability to control molecular architecture with precision is the defining factor between a viable commercial process and a laboratory curiosity. Patent CN110790713A introduces a transformative regioselective one-step method for the synthesis of 2-hydroxyquinoxalines, addressing a longstanding bottleneck in the production of these vital heterocyclic scaffolds. Quinoxaline moieties are ubiquitous in biologically active drug-like molecules, particularly in the treatment of abnormal cell growth and cancer, yet their synthesis has historically been plagued by the formation of intractable regioisomeric mixtures. This new technology leverages a counter-intuitive approach involving low-temperature condensation and reagent excess to achieve regioselectivity exceeding 99%, thereby offering a reliable pharmaceutical intermediate supplier pathway that bypasses the need for costly purification steps. By shifting the paradigm from mixture generation to single-isomer dominance, this innovation provides a robust foundation for cost reduction in API manufacturing and ensures a more stable supply chain for downstream drug developers.
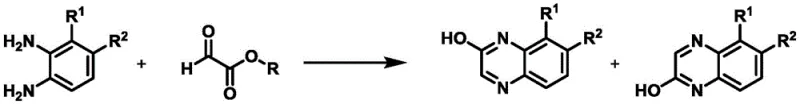
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the condensation of 3,4-disubstituted-1,2-phenylenediamine derivatives with glyoxylates or glyoxylic acids has been a source of significant frustration for process chemists due to its inherent lack of selectivity. As documented in prior art such as WO 2006/134378 A1 and WO 2010/054229 A1, standard reaction conditions typically yield a non-selective mixture of 2-hydroxy-7,8-disubstituted quinoxalines and their 2-hydroxy-5,6-disubstituted regioisomers. This issue is not merely academic; the separation of these structural analogs is often extremely difficult, frequently requiring preparative chromatography, which is a generally undesirable step in large-scale process sequences due to high solvent consumption and low throughput. For instance, previous attempts described in the literature resulted in product mixtures containing up to 30% of the unwanted regioisomer, necessitating complex purification protocols that erode profit margins and extend lead times significantly.
The Novel Approach
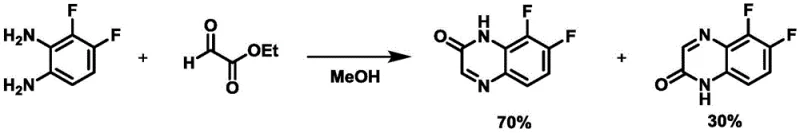
The methodology disclosed in CN110790713A represents a decisive break from these conventional limitations by introducing a kinetically controlled environment that favors the formation of a single regioisomer. The core of this innovation lies in the specific manipulation of reaction parameters: maintaining the reaction matrix at a low temperature range of approximately -20°C to 5°C and utilizing a substantial excess of the glyoxylic acid component. By slowly adding the 1,2-phenylenediamine solution to the excess glyoxylic acid under these cryogenic conditions, the process effectively suppresses the formation of the thermodynamic byproduct. This strategic inversion of addition order and thermal management allows for the direct isolation of the desired 2-hydroxyquinoxaline with purities often exceeding 98%, transforming a previously messy transformation into a clean, high-yielding operation suitable for industrial application.
Mechanistic Insights into Low-Temperature Regioselective Cyclization
The success of this regioselective synthesis can be attributed to the delicate interplay between kinetic control and steric hindrance management during the cyclization event. At elevated temperatures, the reaction tends to equilibrate towards a mixture of isomers driven by thermodynamic stability, where the energy difference between the 5,6- and 7,8-substituted products is insufficient to drive exclusive formation of one species. However, by operating at temperatures near 0°C or below, the system is locked into a kinetic regime where the activation energy barrier for the formation of the desired isomer is preferentially overcome. Furthermore, the use of at least 3 to 5 molar equivalents of glyoxylic acid ensures that the diamine is immediately surrounded by a high concentration of the electrophile, minimizing side reactions and promoting the specific orientation required for the 7,8-closure over the 5,6-alternative. This mechanistic understanding is crucial for R&D teams aiming to replicate these results, as even slight deviations in temperature or stoichiometry can revert the system to the non-selective behavior observed in older patents.
From an impurity control perspective, this mechanism offers profound advantages by preventing the formation of the regioisomer at the source rather than removing it post-reaction. In traditional syntheses, the regioisomer acts as a persistent impurity that co-elutes during crystallization, forcing manufacturers to resort to silica gel chromatography or recrystallization loops that sacrifice yield. The new method's ability to achieve regioselectivity of greater than 95%, and in many cases over 99%, means that the crude product is essentially the final product after a simple filtration and wash. This drastic simplification of the impurity profile not only enhances the safety margin for pharmaceutical applications by reducing unknown related substances but also streamlines the analytical workload required for batch release, allowing quality control laboratories to focus on critical parameters rather than chasing trace isomers.
How to Synthesize 2-Hydroxyquinoxalines Efficiently
The practical implementation of this regioselective protocol is designed for ease of operation, requiring standard laboratory equipment capable of precise temperature control and slow addition rates. The process begins with the preparation of a solution containing the glyoxylic acid source, such as glyoxylic acid monohydrate or 2,2-dialkoxyacetic acid, in a polar solvent like methanol or ethanol. Once the reaction vessel is cooled to the target range of -20°C to 5°C, the substituted 1,2-phenylenediamine is introduced dropwise to ensure that the local concentration of the amine never exceeds the capacity of the excess acid to react selectively. Detailed standardized synthesis steps, including specific workup procedures and purification metrics for various substrates, are outlined in the technical guide below to assist process engineers in rapid technology transfer.
- Prepare a solution of glyoxylic acid, glyoxylic acid monohydrate, or 2,2-dialkoxyacetic acid in a suitable solvent such as methanol or ethanol.
- Cool the reaction mixture to a low temperature range between -20°C and 5°C, preferably around 0°C, to ensure kinetic control.
- Slowly add the 1,2-phenylenediamine derivative dropwise to the excess glyoxylic acid solution while maintaining strict temperature control to achieve high regioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this regioselective synthesis method translates directly into enhanced operational efficiency and reduced risk exposure. The elimination of preparative chromatography, which is explicitly cited as a bottleneck in prior art, removes a major cost center associated with solvent recovery, stationary phase disposal, and extended processing time. This streamlined workflow allows for a significant reduction in the overall cost of goods sold (COGS) without compromising on the stringent purity specifications required for oncology intermediates. Furthermore, the reliance on commodity chemicals such as glyoxylic acid and substituted anilines ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with exotic catalysts or specialized reagents.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the removal of the purification bottleneck. By achieving high regioselectivity directly in the reactor, manufacturers avoid the expensive and time-consuming step of separating isomers via chromatography. This leads to substantial cost savings in terms of solvent usage, labor hours, and equipment occupancy time. Additionally, the high yields reported, such as the 85% isolated yield in Example 1, maximize the output per unit of raw material input, further driving down the unit cost and improving the overall margin profile for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this low-temperature condensation reaction contributes to a more predictable supply chain. Because the process does not rely on sensitive transition metal catalysts that might be subject to supply shortages or price spikes, production continuity is better assured. The simplicity of the workup, which often involves mere filtration and washing as demonstrated in the patent examples, reduces the likelihood of batch failures due to operational complexity. This reliability is critical for maintaining just-in-time inventory levels and ensuring that downstream API synthesis schedules are not disrupted by intermediate shortages.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with green chemistry principles. The reduction in solvent volume associated with avoiding column chromatography significantly lowers the facility's environmental footprint and waste disposal costs. Moreover, the reaction conditions are readily scalable from gram-scale laboratory experiments to multi-ton commercial production using standard jacketed reactors. The ability to handle the exotherm through controlled addition at low temperatures is a well-understood engineering challenge, making the commercial scale-up of complex quinoxaline derivatives a manageable and low-risk endeavor for established chemical manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this regioselective synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is regioselectivity critical in quinoxaline synthesis?
A: Conventional methods often produce inseparable mixtures of 5,6- and 7,8-disubstituted isomers. High regioselectivity eliminates the need for expensive preparative chromatography, drastically reducing production costs and lead times.
Q: What are the optimal reaction conditions for this process?
A: The patent specifies a low-temperature range of -20°C to 5°C and the use of at least 3 to 5 molar equivalents of glyoxylic acid relative to the diamine to drive the reaction towards the desired isomer.
Q: Can this method be scaled for industrial production?
A: Yes, the process utilizes simple filtration for product isolation rather than complex chromatography, making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxyquinoxaline Supplier
As the demand for high-quality oncology intermediates continues to rise, partnering with a manufacturer who possesses deep technical expertise in regioselective synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced process knowledge to deliver compounds with exceptional purity and consistency. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We operate with stringent purity specifications and maintain rigorous QC labs equipped to verify regioisomeric ratios, guaranteeing that every batch meets the exacting standards required for clinical and commercial drug substance manufacturing.
We invite you to discuss how our capabilities can support your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, cost-effective supply of these critical building blocks for your next generation of therapies.
