Scalable Synthesis of Imidazopyridine Compounds Using Earth-Abundant Iron Catalysts
Scalable Synthesis of Imidazopyridine Compounds Using Earth-Abundant Iron Catalysts
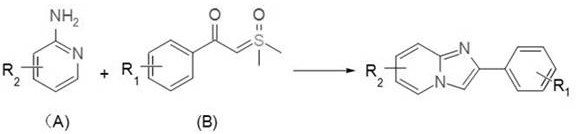
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways to access privileged scaffolds like imidazo[1,2-a]pyridines, which are core structures in blockbuster drugs such as Zolpidem and Nicorandil. Patent CN112538087B introduces a groundbreaking synthetic methodology that leverages iron phthalocyanine (FeIIPc) as a photocatalyst to drive the cyclization of aminopyridines and sulfur ylides. This innovation represents a significant paradigm shift from traditional noble metal catalysis, offering a green, efficient, and economically viable route for the production of high-purity pharmaceutical intermediates. By utilizing visible light irradiation in conjunction with thermal energy, this photo-thermal concerted catalytic system achieves remarkable yields under mild conditions, addressing critical pain points regarding catalyst cost and environmental impact in modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the imidazo[1,2-a]pyridine skeleton has relied heavily on transition metal catalysts based on ruthenium, rhodium, iridium, and palladium. While these noble metal systems have demonstrated efficacy in C-H activation and cyclization reactions, they suffer from severe economic and logistical drawbacks that hinder large-scale commercial adoption. The exorbitant cost of precursors like dichlorobis(p-cymene)ruthenium(II) or iridium dimers creates a substantial burden on the bill of materials, particularly for high-volume API manufacturing. Furthermore, the toxicity profiles of these heavy metals necessitate complex and expensive downstream purification processes to meet stringent regulatory limits for residual metals in drug substances. Many existing protocols also require harsh reaction conditions, including high temperatures exceeding 120°C and the use of stoichiometric oxidants or strict inert atmospheres, which complicates process safety and increases energy consumption.
The Novel Approach
The methodology disclosed in the patent data offers a transformative solution by replacing expensive noble metals with iron phthalocyanine, an earth-abundant and environmentally benign catalyst. This new approach utilizes a synergistic combination of blue light irradiation (465 nm) and moderate heating (80°C) to activate the sulfur ylide and aminopyridine substrates efficiently. Unlike previous methods that often struggle with substrate scope or require specialized ligands, this iron-catalyzed system demonstrates exceptional versatility, accommodating a wide range of functional groups including halogens, ethers, and trifluoromethyl groups without compromising yield. The ability to run the reaction under air atmosphere further simplifies the operational protocol, removing the need for costly glovebox techniques or extensive degassing procedures, thereby streamlining the workflow for process chemists.
Mechanistic Insights into FePc-Catalyzed Photo-thermal Cyclization
The success of this synthetic route lies in the unique photophysical properties of iron phthalocyanine, which acts as a potent photocatalyst capable of absorbing visible light to generate excited states that facilitate bond formation. Mechanistic studies suggest a photo-thermal concerted catalysis pathway where the FeIIPc catalyst, in the presence of a zinc salt additive like Zn(OTf)2, coordinates with the sulfur ylide to form a reactive metal-carbene intermediate. This highly electrophilic species then undergoes intermolecular insertion with the o-aminopyridine, followed by tautomerization and protonation to generate an imine intermediate. The final cyclization step closes the ring to form the stable imidazo[1,2-a]pyridine core. UV-Vis absorption analysis confirms that the reaction system exhibits strong absorption in the blue light region, allowing for efficient energy transfer that lowers the activation energy barrier compared to purely thermal processes.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions often associated with radical pathways or uncontrolled carbene insertions. The specific coordination environment provided by the macrocyclic phthalocyanine ligand ensures high regioselectivity, preventing the formation of isomeric byproducts that are difficult to separate. Experimental data indicates that the system is highly tolerant to steric hindrance and electronic variations; for instance, para-chloro substituted sulfur ylides afford products with yields up to 90%, while electron-deficient substrates bearing trifluoromethyl groups still react efficiently to give 73% yield. This robustness ensures a clean reaction profile, reducing the burden on purification teams and enhancing the overall mass balance of the manufacturing process.

How to Synthesize Imidazopyridine Efficiently
The practical implementation of this technology involves a straightforward procedure that can be easily adapted for both laboratory discovery and pilot plant operations. The process begins with the preparation of the sulfur ylide precursor, followed by the key cyclization step in a photochemical reactor equipped with blue LED arrays. The simplicity of the reagent setup—requiring only the catalyst, additive, and solvent—makes it highly attractive for rapid scale-up. For detailed standard operating procedures and specific molar ratios optimized for different substrate classes, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining o-aminopyridine, sulfur ylide, FeIIPc catalyst (15 mol%), and Zn(OTf)2 additive in DCE solvent within a quartz tube.
- Set the photochemical reactor to 10W blue light (465 nm) and heat the system to 80°C while maintaining water circulation.
- After 24 hours of irradiation, cool the mixture, extract with ethyl acetate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this iron-catalyzed protocol offers compelling strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic reduction in raw material costs associated with replacing precious metal catalysts with commodity-grade iron complexes. This shift not only lowers the direct cost of goods sold but also mitigates the supply risk associated with the volatile market prices of ruthenium and iridium, which are often subject to geopolitical constraints and mining bottlenecks. Additionally, the elimination of toxic heavy metals simplifies waste management protocols and reduces the environmental compliance burden, aligning with corporate sustainability goals and green chemistry initiatives.
- Cost Reduction in Manufacturing: The substitution of noble metals with iron phthalocyanine results in significant cost savings by removing the need for expensive catalyst recovery systems and reducing the overall catalyst loading cost. Since iron is one of the most abundant elements on earth, the supply chain for the catalyst is stable and immune to the price fluctuations that plague the precious metal market. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product manufactured.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials and a robust catalytic system that functions under air, the manufacturing process becomes less susceptible to disruptions caused by specialized equipment failures or inert gas supply issues. The broad substrate scope means that a single platform technology can be used to produce a diverse library of intermediates, allowing for greater flexibility in production scheduling and inventory management. This reliability ensures consistent delivery timelines for downstream API synthesis, preventing bottlenecks in the drug development pipeline.
- Scalability and Environmental Compliance: The photo-thermal nature of the reaction allows for scalable continuous flow processing, which offers superior heat and mass transfer compared to batch reactors. The use of benign solvents and the absence of stoichiometric oxidants minimize the generation of hazardous waste streams, simplifying effluent treatment and reducing disposal costs. This environmental compatibility facilitates easier regulatory approval for new drug filings, as the impurity profile is cleaner and the residual metal levels are inherently lower than those from noble metal catalysis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis platform. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and licensees.
Q: What are the advantages of using Iron Phthalocyanine over Ruthenium or Iridium catalysts?
A: Iron Phthalocyanine is an earth-abundant, non-toxic transition metal catalyst that significantly reduces raw material costs compared to precious metals like Ruthenium or Iridium. It also eliminates the need for rigorous heavy metal removal steps often required for pharmaceutical grade APIs.
Q: Does this photo-thermal method tolerate diverse functional groups?
A: Yes, the method exhibits broad substrate universality. It successfully tolerates electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens (Cl, Br, I) and trifluoromethyl, with yields reaching up to 91% for specific chloro-substituted derivatives.
Q: Is the reaction sensitive to oxygen or moisture?
A: The process is robust and can be conducted under air atmosphere, simplifying the operational requirements compared to traditional methods that often necessitate strict inert gas protection like nitrogen or argon.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iron-catalyzed photo-thermal technology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-purity imidazopyridine derivatives that support your drug development timelines while optimizing your cost structure.
We invite you to collaborate with our technical procurement team to explore how this novel synthetic route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this earth-abundant catalyst system for your specific project needs. Contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, and let us help you engineer a more sustainable and profitable future for your pharmaceutical portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
