Advanced Lewis Acid Catalysis for Scalable Tezacaftor Intermediate Production
Advanced Lewis Acid Catalysis for Scalable Tezacaftor Intermediate Production
The pharmaceutical industry is constantly seeking more efficient and cost-effective pathways for synthesizing complex drug intermediates, particularly for life-saving medications like Tezacaftor used in the treatment of cystic fibrosis. A groundbreaking development detailed in patent CN111187197B introduces a superior synthetic methodology that fundamentally alters the production landscape for key Tezacaftor intermediates. This innovation replaces traditional, costly palladium-catalyzed cyclization steps with a highly efficient Lewis acid-mediated process, specifically utilizing zinc salts. For R&D directors and procurement specialists, this shift represents a critical opportunity to optimize supply chains, reduce heavy metal contamination risks, and lower overall manufacturing expenditures while maintaining exceptional product quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the indole-based core structure essential for Tezacaftor has relied heavily on transition metal catalysis, specifically employing palladium reagents. As illustrated in prior art such as patent CN103038214A, conventional routes typically involve a multi-step sequence where palladium catalysts are indispensable for forming the critical carbon-carbon bonds. 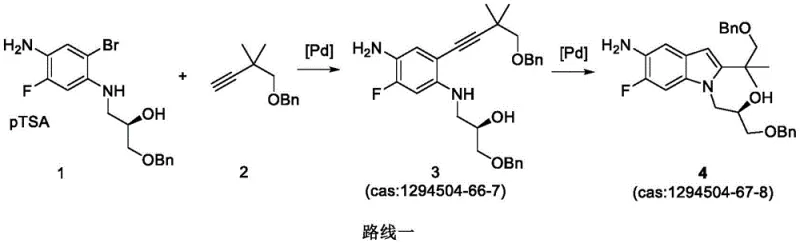 These legacy methods suffer from significant drawbacks that hinder industrial scalability. The primary concern is the exorbitant cost associated with palladium reagents, which are not only expensive to purchase but also require complex recovery systems to prevent loss. Furthermore, the presence of palladium residues in the final intermediate poses a severe risk for downstream API manufacturing, necessitating rigorous and often yield-reducing purification steps to meet strict elemental impurity guidelines set by health authorities. Additionally, these older processes often report lower reaction yields, compounding the economic inefficiency and making them less attractive for high-volume commercial production.
These legacy methods suffer from significant drawbacks that hinder industrial scalability. The primary concern is the exorbitant cost associated with palladium reagents, which are not only expensive to purchase but also require complex recovery systems to prevent loss. Furthermore, the presence of palladium residues in the final intermediate poses a severe risk for downstream API manufacturing, necessitating rigorous and often yield-reducing purification steps to meet strict elemental impurity guidelines set by health authorities. Additionally, these older processes often report lower reaction yields, compounding the economic inefficiency and making them less attractive for high-volume commercial production.
The Novel Approach
In stark contrast to the palladium-dependent legacy routes, the technology disclosed in CN111187197B presents a streamlined and economically superior alternative. The core innovation lies in the utilization of a Lewis acid catalyst system, preferably comprising zinc bromide (ZnBr2), potentially augmented with inorganic bases like sodium hydroxide and polar aprotic solvents such as NMP. 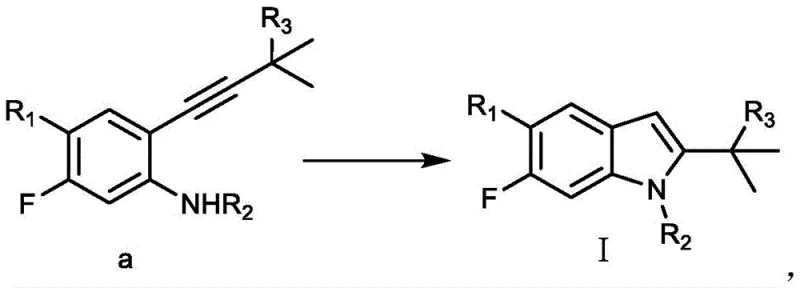 This novel approach facilitates the cyclization of the alkynyl-aniline precursor directly into the desired indole structure with remarkable efficiency. By eliminating the need for precious metal catalysts in the cyclization step, the process drastically reduces raw material costs and simplifies the workup procedure. The reaction conditions are robust, typically proceeding under reflux in solvents like chlorobenzene, and deliver outstanding results with total yields exceeding 90% and product purity surpassing 99%. This transition from noble metal catalysis to base metal Lewis acid catalysis marks a significant technological leap, offering a sustainable and cost-competitive solution for the manufacture of high-value pharmaceutical intermediates.
This novel approach facilitates the cyclization of the alkynyl-aniline precursor directly into the desired indole structure with remarkable efficiency. By eliminating the need for precious metal catalysts in the cyclization step, the process drastically reduces raw material costs and simplifies the workup procedure. The reaction conditions are robust, typically proceeding under reflux in solvents like chlorobenzene, and deliver outstanding results with total yields exceeding 90% and product purity surpassing 99%. This transition from noble metal catalysis to base metal Lewis acid catalysis marks a significant technological leap, offering a sustainable and cost-competitive solution for the manufacture of high-value pharmaceutical intermediates.
Mechanistic Insights into Zinc-Mediated Cyclization
The mechanistic elegance of this new process centers on the activation of the alkyne moiety by the zinc species. In the presence of a Lewis acid like ZnBr2, the electron density of the triple bond in the alkynyl-aniline precursor is perturbed, making it more susceptible to nucleophilic attack by the adjacent amine nitrogen. This intramolecular hydroamination or cyclization event is the pivotal step that constructs the indole ring system. Unlike palladium cycles which involve oxidative addition and reductive elimination steps that can be sensitive to ligand environments and oxygen levels, the zinc-mediated pathway is operationally simpler and more tolerant. The inclusion of additives like NMP likely serves to stabilize the zinc species or solvate the transition state, enhancing the reaction kinetics. Furthermore, the optional addition of a mild base such as NaOH assists in deprotonating the amine or managing the proton balance during the cyclization, ensuring the reaction proceeds to completion without stalling at intermediate stages. This mechanistic simplicity translates directly to process reliability, a key metric for process chemists evaluating technology transfer.
From an impurity control perspective, the switch to zinc chemistry offers profound advantages. Palladium-catalyzed reactions are notorious for generating metal-associated impurities and requiring scavengers that can introduce their own contaminants. By utilizing zinc, a metal with much higher permissible daily exposure limits and lower toxicity profiles compared to palladium, the risk profile of the intermediate is significantly lowered. The patent data indicates that the resulting product achieves a purity of greater than 99%, suggesting that side reactions such as polymerization of the alkyne or over-alkylation are effectively suppressed under these specific Lewis acid conditions. 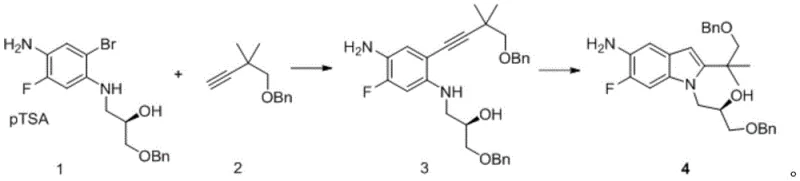 The high purity is further evidenced by the straightforward workup described in the examples, where simple aqueous washes and filtration suffice to isolate the product, avoiding the need for complex chromatographic separations that often plague palladium-mediated syntheses. This clean reaction profile ensures that the impurity spectrum is manageable and predictable, facilitating easier regulatory filing and approval for the final drug substance.
The high purity is further evidenced by the straightforward workup described in the examples, where simple aqueous washes and filtration suffice to isolate the product, avoiding the need for complex chromatographic separations that often plague palladium-mediated syntheses. This clean reaction profile ensures that the impurity spectrum is manageable and predictable, facilitating easier regulatory filing and approval for the final drug substance.
How to Synthesize Tezacaftor Intermediate Efficiently
The synthesis of the target Tezacaftor intermediate via this patented route involves a logical two-stage sequence that balances classic coupling chemistry with the innovative cyclization step. Initially, the alkynyl-aniline precursor (Compound 3 in the patent examples) is prepared via a Sonogashira coupling reaction between a bromo-aniline derivative and a protected alkyne. While this initial step may still utilize palladium, the critical value-add of this technology is the subsequent transformation. The detailed standardized synthetic steps for the cyclization and isolation are outlined below, providing a clear roadmap for laboratory validation and scale-up.
- Perform Sonogashira coupling between the bromo-aniline derivative and the protected alkyne using palladium acetate and copper iodide in acetonitrile under reflux to generate the alkynyl-aniline precursor.
- Subject the crude alkynyl-aniline precursor to cyclization conditions using zinc bromide (ZnBr2) as the Lewis acid catalyst, optionally with sodium hydroxide and NMP in chlorobenzene solvent.
- Reflux the reaction mixture for approximately 24 hours, followed by workup involving filtration, washing with ammonium chloride and brine, and purification to achieve over 90% yield and 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-catalyzed synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of palladium from the critical cyclization step directly addresses two major pain points in pharmaceutical supply chains: cost volatility and supply security. Precious metal prices are subject to significant market fluctuations, and reliance on them introduces financial uncertainty into long-term production contracts. By shifting to abundant and inexpensive zinc salts, manufacturers can lock in more stable and predictable cost structures. Furthermore, the removal of heavy metal residues simplifies the quality control workflow, reducing the turnaround time for batch release and minimizing the risk of batch rejection due to out-of-specification metal content. This operational efficiency translates into a more resilient supply chain capable of meeting the demanding delivery schedules of global pharmaceutical partners.
- Cost Reduction in Manufacturing: The economic impact of replacing palladium reagents with zinc bromide cannot be overstated. Palladium is a precious metal with a high unit cost, and its usage often requires stoichiometric or near-stoichiometric amounts in difficult transformations, alongside expensive ligands. In contrast, zinc salts are commodity chemicals available at a fraction of the cost. This substitution leads to a drastic reduction in direct material costs for the cyclization step. Additionally, the simplified purification process reduces the consumption of solvents and stationary phases used in chromatography, further driving down the cost of goods sold (COGS). The high yield reported (>90%) ensures that raw material utilization is maximized, minimizing waste and enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: Dependence on specialized palladium catalysts can create bottlenecks, especially during periods of high global demand or geopolitical instability affecting metal supply. Zinc, being a base metal with a robust global supply infrastructure, offers superior availability and sourcing flexibility. This ensures that production schedules are not held hostage by catalyst shortages. Moreover, the robustness of the reaction conditions—using common solvents like chlorobenzene and standard heating methods—means that the process can be easily replicated across different manufacturing sites without requiring specialized equipment or highly exotic reagents. This geographic flexibility strengthens the supply chain against regional disruptions and ensures continuous availability of this critical intermediate for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, particularly regarding heat management and waste disposal. The high yield and selectivity of this Lewis acid method mean that fewer by-products are generated, simplifying waste stream management. The absence of palladium eliminates the need for complex metal recovery units or the disposal of hazardous metal-laden waste, aligning the process better with green chemistry principles and environmental regulations. The use of recyclable solvents and the potential for telescoping steps further enhance the process mass intensity (PMI) metrics. This environmental friendliness not only reduces disposal costs but also positions the manufacturer favorably in an increasingly eco-conscious regulatory landscape, facilitating smoother audits and approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines or for procurement teams negotiating supply agreements based on these improved specifications.
Q: Why is the switch from Palladium to Zinc catalysts significant for Tezacaftor intermediate production?
A: Traditional methods rely on expensive palladium reagents which increase production costs and pose risks of heavy metal residue in the final API. The novel zinc-catalyzed method eliminates these costly metals, significantly lowering raw material expenses and simplifying the purification process to meet stringent pharmaceutical purity standards.
Q: What purity levels can be achieved with this Lewis acid cyclization method?
A: The patented process demonstrates exceptional quality control, consistently achieving product purity levels exceeding 99%. This high purity is critical for downstream API synthesis, reducing the burden on subsequent purification steps and ensuring compliance with rigorous regulatory specifications for cystic fibrosis medications.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the method is specifically designed for industrial scalability. By utilizing robust and inexpensive reagents like zinc bromide and common solvents such as chlorobenzene, combined with a high total yield of over 90%, the process offers a economically viable and operationally stable pathway for commercial-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tezacaftor Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Lewis acid-catalyzed synthesis route for Tezacaftor intermediates described in CN111187197B. As a leading CDMO partner, we possess the technical expertise and infrastructure to seamlessly adopt and optimize this advanced methodology for our clients. Our facilities are equipped to handle diverse synthetic pathways, ranging from early-stage process development to full-scale commercial manufacturing. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is smooth and efficient. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and high-yielding technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you secure a reliable, high-quality supply of Tezacaftor intermediates that drives down your costs and accelerates your time to market.
