Advanced Synthesis of N-Difluoromethyl Azaindole Compounds for Pharmaceutical Applications
Advanced Synthesis of N-Difluoromethyl Azaindole Compounds for Pharmaceutical Applications
The introduction of fluorine atoms into organic molecules is a cornerstone strategy in modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of drug candidates. Specifically, the difluoromethyl group (-CF2H) serves as a unique lipophilic hydrogen bond donor, offering distinct advantages over trifluoromethyl groups in modulating protein-ligand interactions. Patent CN112279849A discloses a groundbreaking synthetic methodology for producing N-difluoromethyl azaindole compounds, a class of heterocycles increasingly vital for developing kinase inhibitors and other bioactive agents. This innovation addresses critical bottlenecks in existing manufacturing processes by utilizing inexpensive, commercially available reagents under exceptionally mild conditions. The general structural scope of these valuable intermediates is illustrated below, highlighting the versatility of the substitution patterns achievable through this robust protocol.
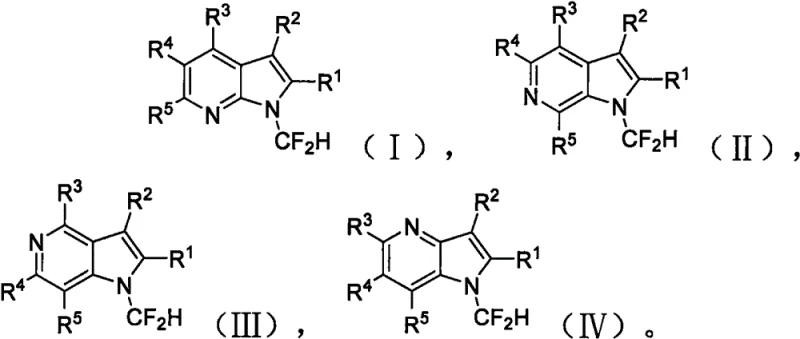
Furthermore, the patent details a comprehensive library of specific derivatives, demonstrating the method's adaptability to various azaindole isomers including 4-, 5-, 6-, and 7-azaindoles. The ability to introduce the difluoromethyl moiety directly onto the nitrogen atom without protecting group manipulation represents a significant leap forward in process efficiency. By enabling the rapid assembly of diverse chemical space around the azaindole core, this technology empowers research teams to accelerate lead optimization campaigns. The specific examples provided in the disclosure confirm that even sterically hindered or electronically deactivated substrates can be successfully transformed, ensuring a reliable supply of high-purity pharmaceutical intermediates for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the N-difluoromethylation of nitrogen-containing heterocycles has been plagued by significant economic and operational challenges that hinder large-scale adoption. Early methodologies, such as those reported by the Hu Jinbo group in 2007, relied heavily on chlorodifluoromethylbenzenesulfone as the difluorocarbene source. As depicted in the reaction scheme below, while effective, this reagent is prohibitively expensive and poses supply chain vulnerabilities for industrial manufacturers seeking cost-effective solutions.
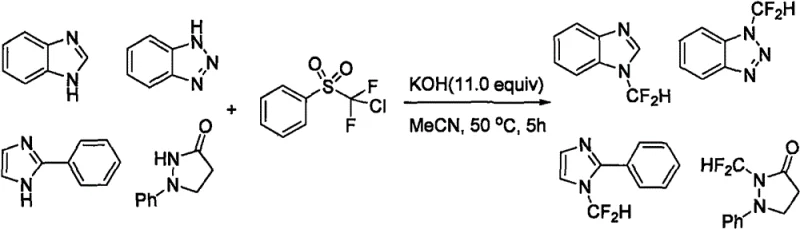
Subsequent advancements attempted to mitigate some issues but introduced new drawbacks; for instance, the Shouggchang group utilized triphenylphosphonium difluoroacetate inner salts, which, despite improved reactivity, remained cost-prohibitive for bulk production. Other approaches, such as the Prakash method employing TMSCF3, necessitated extreme reaction temperatures reaching 170°C, creating severe safety hazards and energy inefficiencies incompatible with green chemistry principles. Additionally, many traditional routes require transition metal catalysts, introducing the risk of heavy metal contamination that demands rigorous and costly purification steps to meet stringent pharmaceutical quality standards. These cumulative factors have historically limited the accessibility of N-difluoromethyl azaindoles, creating a bottleneck for the commercialization of promising fluorinated drug candidates.
The Novel Approach
The methodology described in CN112279849A fundamentally disrupts this status quo by leveraging ethyl bromodifluoroacetate (BrCF2CO2Et) as a superior difluorocarbene precursor. This reagent is not only significantly cheaper and more readily available on the global market but also exhibits high reactivity under ambient conditions. The process operates effectively at room temperature, typically between 20°C and 25°C, eliminating the need for energy-intensive heating or cryogenic cooling systems. By employing simple inorganic or organic bases like potassium tert-butoxide, the reaction proceeds smoothly without the necessity for exotic ligands or sensitive metal catalysts. This metal-free paradigm shift ensures that the resulting products are free from trace metal impurities, simplifying the downstream purification workflow and enhancing the overall safety profile of the manufacturing process. Consequently, this approach offers a scalable, economically viable pathway for producing high-value fluorinated intermediates.
Mechanistic Insights into Base-Mediated N-Difluoromethylation
The core of this synthetic breakthrough lies in the efficient generation of difluorocarbene species under mild basic conditions. Upon treatment with a strong base such as potassium tert-butoxide, ethyl bromodifluoroacetate undergoes rapid deprotonation followed by alpha-elimination to release the reactive difluorocarbene intermediate in situ. This electrophilic species is immediately captured by the nucleophilic nitrogen atom of the azaindole ring system, forming the desired N-CF2H bond with high regioselectivity. The absence of competing side reactions, such as O-difluoromethylation or ring decomposition, underscores the chemoselectivity of this transformation. Furthermore, the reaction kinetics are favorable enough to reach completion within 12 to 24 hours, balancing throughput with operational simplicity. This mechanistic elegance allows for the preservation of sensitive functional groups elsewhere on the molecule, which is critical for late-stage functionalization in complex drug synthesis.
From an impurity control perspective, the simplicity of the reaction matrix facilitates straightforward purification strategies. Since the byproducts primarily consist of inorganic salts and ethanol derivatives, they can be easily removed via aqueous workup and standard column chromatography. The lack of metal catalysts removes the need for specialized scavenging resins or activated carbon treatments, which often result in product loss. Moreover, the broad substrate scope indicates that electronic effects from substituents on the pyridine or pyrrole rings do not significantly inhibit the nucleophilic attack, suggesting a robust and forgiving reaction profile. This reliability is paramount for maintaining consistent batch-to-batch quality in a GMP environment, ensuring that the final API intermediates meet the rigorous purity specifications required by regulatory agencies worldwide.
How to Synthesize N-Difluoromethyl Azaindole Efficiently
To implement this synthesis effectively, operators should adhere to the optimized parameters outlined in the patent examples, which prioritize safety and yield. The process begins with the careful selection of the solvent, with acetonitrile identified as the optimal medium due to its ability to solubilize both the organic substrate and the inorganic base while maintaining reaction stability. Detailed standard operating procedures regarding stoichiometry, addition rates, and quenching protocols are essential for maximizing the 90% yields observed in benchmark examples. For a comprehensive guide on the step-by-step execution of this protocol, please refer to the standardized synthesis instructions provided below.
- Dissolve the azaindole substrate, ethyl bromodifluoroacetate (BrCF2CO2Et), and a base such as potassium tert-butoxide in an organic solvent like acetonitrile.
- Stir the reaction mixture at room temperature for 12 to 24 hours under a nitrogen atmosphere to ensure complete conversion.
- Quench the reaction with water, extract with ethyl acetate, dry the organic phase, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs, driven by the substitution of exotic, high-priced fluorinating agents with commodity chemicals like ethyl bromodifluoroacetate. This shift significantly lowers the cost of goods sold (COGS) for the final intermediate, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients. Additionally, the elimination of expensive transition metal catalysts removes a major cost center associated with both reagent purchase and waste disposal, further enhancing the economic viability of the process on a multi-ton scale.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free, room-temperature protocol is profound, as it removes the dependency on volatile precious metal markets and reduces energy consumption. By avoiding high-temperature reactors and complex catalyst recovery systems, capital expenditure (CAPEX) requirements for production facilities are minimized. The simplified workup procedure also reduces labor hours and solvent usage, contributing to substantial operational savings. These cumulative efficiencies allow for a more aggressive pricing strategy while maintaining healthy profit margins, making the supply of these critical intermediates more resilient to market fluctuations.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved because the key reagents, such as ethyl bromodifluoroacetate and common alkali bases, are produced by multiple global suppliers with established logistics networks. Unlike proprietary reagents that may face single-source bottlenecks, these commodity chemicals ensure continuous availability even during periods of global supply chain disruption. The mild reaction conditions also reduce the risk of batch failures due to thermal runaway or equipment malfunction, leading to higher on-time delivery rates. This reliability is crucial for pharmaceutical partners who depend on just-in-time delivery models to maintain their own clinical trial and commercial production schedules without interruption.
- Scalability and Environmental Compliance: The environmental footprint of this process is significantly smaller than traditional methods, aligning with the increasing demand for sustainable manufacturing practices. The absence of heavy metals simplifies wastewater treatment and reduces the generation of hazardous solid waste, lowering disposal costs and regulatory compliance burdens. The room-temperature operation inherently reduces the carbon footprint associated with heating and cooling, supporting corporate sustainability goals. Furthermore, the robustness of the reaction allows for seamless scale-up from kilogram to multi-ton batches without the need for extensive re-optimization, ensuring that supply can rapidly expand to meet surging market demand for fluorinated APIs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-difluoromethylation technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on performance metrics and operational requirements. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. For further specific data points or custom feasibility studies, direct engagement with our technical sales team is recommended.
Q: What are the advantages of this N-difluoromethylation method over conventional sulfone-based routes?
A: Conventional methods often rely on expensive reagents like chlorodifluoromethylbenzenesulfone or require harsh conditions such as high temperatures (170°C). This novel protocol utilizes ethyl bromodifluoroacetate, which is significantly cheaper and more readily available, and proceeds under mild room temperature conditions, drastically reducing energy consumption and safety risks.
Q: Does this synthesis method tolerate diverse functional groups on the azaindole scaffold?
A: Yes, the method demonstrates excellent functional group tolerance. It is compatible with a wide range of substituents including electron-withdrawing groups like nitro, cyano, and halogens, as well as electron-donating groups like methyl and esters, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Q: Is metal catalysis required for this difluoromethylation process?
A: No, this process is metal-free. It relies on a base-mediated mechanism using alkali bases such as potassium tert-butoxide. This eliminates the need for expensive transition metal catalysts and avoids the complex purification steps associated with removing heavy metal residues, ensuring higher purity for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Difluoromethyl Azaindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to delivering products that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the advanced synthesis methods described in CN112279849A, we offer a superior value proposition that combines technical excellence with commercial reliability.
We invite you to collaborate with us to optimize your supply chain for N-difluoromethyl azaindole derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our process improvements can lower your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey from discovery to commercial success with our premium chemical solutions.
