Revolutionizing Pharmaceutical Intermediates Scalable Metal-Free Synthesis of N-Difluoromethyl Azaindole Compounds for Global Supply Chains
The patent CN112279849A introduces a groundbreaking synthetic methodology for N-difluoromethyl azaindole compounds that addresses critical limitations in pharmaceutical intermediate manufacturing through innovative metal-free chemistry. This technical advancement represents a significant leap forward in fluorinated heterocycle synthesis by leveraging readily available reagents under exceptionally mild conditions at room temperature. The process demonstrates remarkable functional group tolerance across diverse substrates including substituted azaindoles with halogens, nitro groups, and carbonyl functionalities while achieving high yields without transition metal catalysts. This innovation directly responds to industry demands for sustainable manufacturing routes that reduce environmental impact while maintaining stringent purity requirements essential for pharmaceutical applications. The methodology's commercial viability is underscored by its straightforward implementation using standard laboratory equipment and common solvents like acetonitrile without requiring specialized infrastructure or hazardous materials handling protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to N-difluoromethylation have been severely constrained by multiple critical limitations that hinder industrial adoption including harsh reaction conditions requiring elevated temperatures up to 90°C or even higher in some cases such as Prakash's method operating at 170°C which significantly increases energy consumption and safety risks. These conventional processes frequently rely on expensive difluoromethylation reagents like chlorodifluoromethylbenzenesulfone or TMSCF3 that drive up raw material costs while necessitating complex purification steps to remove transition metal catalysts that contaminate final products. The narrow substrate scope observed in prior art methods such as those developed by Hujinbo in 2007 or Shouggchang in 2013 restricts applicability to specific molecular frameworks while poor functional group tolerance leads to low yields when handling sensitive substituents commonly found in pharmaceutical intermediates. Furthermore these established techniques often require specialized equipment capable of withstanding high temperatures or pressures creating substantial barriers to scale-up and increasing capital expenditure for manufacturing facilities seeking commercial implementation.
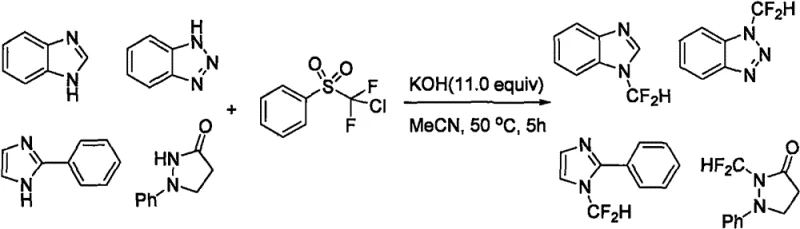
The Novel Approach
The patented methodology overcomes these historical challenges through an elegant room temperature process that employs ethyl difluorobromoacetate as a cost-effective difluorocarbene source combined with potassium tert-butoxide as base in acetonitrile solvent without any metal catalysts. This innovation achieves complete reaction conversion within just six to twelve hours under ambient conditions eliminating energy-intensive heating requirements while maintaining excellent yields between sixty-five percent and ninety percent across diverse substrates as demonstrated in multiple examples within the patent documentation. The process exhibits exceptional functional group compatibility allowing successful modification of azaindoles bearing halogens nitro groups cyano substituents and various carbonyl functionalities without side reactions or decomposition issues that plagued previous approaches. Crucially this method avoids expensive reagents and transition metals entirely removing both cost barriers and contamination risks while simplifying downstream purification through straightforward column chromatography with standard solvent systems.
Mechanistic Insights into Metal-Free Difluoromethylation
The reaction mechanism proceeds through a base-mediated deprotonation pathway where potassium tert-butoxide generates the azaindole anion that subsequently attacks the electrophilic carbon of ethyl difluorobromoacetate forming a key intermediate that undergoes rapid elimination to yield the N-difluoromethylated product. This metal-free process avoids radical pathways or transition metal coordination complexes entirely relying instead on simple nucleophilic substitution chemistry that operates efficiently at room temperature due to the favorable electrophilicity of the difluorocarbene precursor. The absence of metal catalysts eliminates potential coordination effects that could lead to undesired side products while maintaining precise control over regioselectivity at the nitrogen position of the azaindole ring system. This mechanistic simplicity directly contributes to the exceptional functional group tolerance observed across diverse substrates including those with electron-withdrawing or electron-donating substituents that would typically interfere with metal-catalyzed processes.
Impurity control is achieved through careful optimization of reaction parameters including precise stoichiometry with a three-fold excess of ethyl difluorobromoacetate relative to azaindole substrate ensuring complete conversion while minimizing unreacted starting materials. The room temperature conditions prevent thermal degradation pathways that could generate decomposition products while the use of acetonitrile as solvent provides optimal polarity for intermediate stabilization without promoting side reactions. Column chromatography purification using petroleum ether and ethyl acetate eluent systems effectively separates minor impurities resulting from incomplete reactions or trace byproducts yielding final products with purity levels suitable for pharmaceutical intermediate applications without requiring additional recrystallization steps. This controlled impurity profile directly addresses regulatory concerns regarding residual metals or organic impurities that could compromise drug safety profiles during subsequent manufacturing stages.
How to Synthesize N-Difluoromethyl Azaindole Efficiently
This section outlines the standardized procedure for manufacturing N-difluoromethyl azaindole compounds based on patent CN112279849A which represents a significant advancement over conventional methods through its mild reaction conditions and exceptional substrate scope. The process eliminates hazardous high temperatures while maintaining excellent yield consistency across diverse molecular frameworks making it ideal for pharmaceutical intermediate production where purity and reproducibility are paramount concerns. Detailed operational parameters have been optimized through extensive experimentation as documented in the patent examples ensuring robust performance under commercial manufacturing conditions. The following standardized synthesis steps provide a clear pathway for R&D teams to implement this technology effectively within their existing production infrastructure.
- Combine azaindole compound, difluoromethyl reagent such as ethyl difluorobromoacetate at a molar ratio of 1: 3, potassium tert-butoxide as base at equimolar ratio to reagent, and acetonitrile solvent in a nitrogen-purged reactor.
- Stir the reaction mixture at room temperature for precisely 6 to 12 hours to ensure complete conversion without thermal degradation or side reactions.
- Purify the crude product using column chromatography with petroleum ether and ethyl acetate as eluents to achieve high-purity N-difluoromethyl azaindole compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic benefits specifically addressing critical pain points faced by procurement and supply chain decision-makers in pharmaceutical manufacturing organizations seeking reliable sources for complex intermediates. The elimination of transition metal catalysts removes significant cost drivers associated with catalyst procurement handling and subsequent removal processes while enabling direct scale-up from laboratory to commercial production without infrastructure modifications. These advantages translate into tangible operational improvements across multiple dimensions including cost structure reliability and environmental compliance making this approach particularly valuable for organizations managing global supply chains with stringent quality requirements.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes both procurement costs and complex purification steps required to eliminate metal residues from final products significantly reducing overall production expenses while maintaining high purity standards essential for pharmaceutical applications without requiring additional quality control measures.
- Enhanced Supply Chain Reliability: Utilization of readily available commercial reagents such as ethyl difluorobromoacetate and potassium tert-butoxide ensures consistent raw material availability while room temperature processing eliminates temperature control dependencies that could disrupt production schedules during seasonal variations or equipment maintenance periods.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production maintains consistent yields without requiring specialized equipment modifications while generating minimal hazardous waste streams due to mild reaction conditions and standard solvent systems facilitating regulatory compliance with environmental standards across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address specific technical and commercial concerns raised by industry professionals regarding implementation of this patented synthesis methodology based on detailed analysis of both background limitations and demonstrated advantages within patent CN112279849A documentation.
Q: How does this method overcome high temperature requirements in conventional difluoromethylation?
A: The novel process operates at room temperature by utilizing ethyl difluorobromoacetate with potassium tert-butoxide in acetonitrile, eliminating energy-intensive heating steps required by prior methods that needed temperatures up to 90°C or higher.
Q: What are the advantages of metal-free synthesis for pharmaceutical intermediate production?
A: Metal-free conditions prevent heavy metal contamination in final products, significantly reducing purification costs and ensuring compliance with stringent pharmaceutical quality standards without requiring additional metal removal steps.
Q: How does room temperature processing improve scalability and cost efficiency?
A: Mild reaction conditions enable direct scale-up from laboratory to commercial production without specialized equipment modifications, substantially lowering capital expenditure while maintaining consistent yields across batch sizes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Difluoromethyl Azaindole Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs that ensure consistent quality across all batch sizes. This patented methodology aligns perfectly with our core competencies in complex fluorinated heterocycle synthesis where we have successfully implemented similar metal-free processes for global pharmaceutical clients requiring high-purity intermediates meeting exacting regulatory standards. Our technical team stands ready to support seamless integration of this innovative route into your manufacturing workflow while providing comprehensive documentation packages that facilitate regulatory submissions across major markets worldwide.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today who can provide immediate access to specific COA data and route feasibility assessments demonstrating how this technology can optimize your supply chain operations while ensuring uninterrupted access to critical intermediates.
