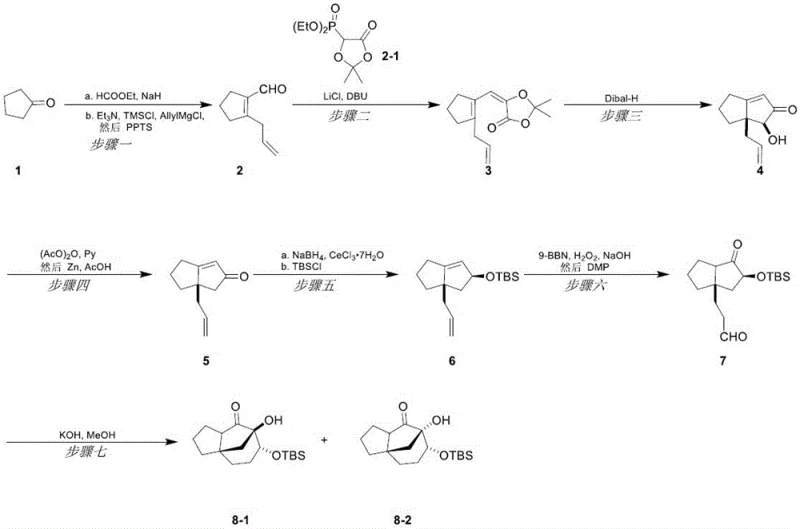
Scalable Synthesis of Cedrol Tricyclic Skeleton for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex natural product skeletons, particularly for sesquiterpenes like cedrol which exhibit significant biological activity. Patent CN116284105A discloses a groundbreaking preparation method for the tricyclic skeleton of sesquiterpene cedrol, addressing the critical need for cost reduction in pharmaceutical intermediates manufacturing. This innovative approach utilizes cyclopentanone, a cheap and readily available starting material, to construct the complex bridged ring system through a series of highly selective organic transformations. The core of this technology lies in the strategic application of a reductive oxy-Nazarov electrocyclization combined with an intramolecular Aldol reaction, which dramatically shortens the synthetic route compared to historical precedents. By eliminating the reliance on expensive noble metal catalysts and reducing the total number of steps, this method offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for high-purity OLED material and pharmaceutical precursors alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of cedrol and its derivatives has been plagued by inefficiencies that hinder large-scale commercial adoption. Early methodologies, such as the 23-step sequence reported by the Stork group, are far too lengthy and labor-intensive for modern industrial requirements, leading to cumulative yield losses that make the final product prohibitively expensive. Furthermore, many traditional routes rely heavily on transition metal catalysis, including gold or rhodium complexes, which not only inflate raw material costs but also introduce significant challenges in post-reaction purification. The removal of trace metal residues to meet stringent pharmaceutical purity specifications often requires additional processing steps, such as specialized filtration or chelation, which further extends the production lead time for high-purity pharmaceutical intermediates. Additionally, some conventional methods utilize harsh reaction conditions or unstable intermediates that pose safety hazards during scale-up, creating bottlenecks for supply chain heads who prioritize operational continuity and risk mitigation in their manufacturing facilities.
The Novel Approach
In stark contrast, the method disclosed in the patent data introduces a streamlined strategy that fundamentally reimagines the construction of the cedrol tricyclic core. By leveraging a reductive oxy-Nazarov electrocyclization, the process efficiently builds the critical [5.5] fused ring system in a single operational step, bypassing the need for multiple ring-closing reactions that typically degrade overall yield. The use of cyclopentanone as the foundational building block ensures that the raw material supply is stable and cost-effective, directly addressing the concerns of a reliable agrochemical intermediate supplier or pharma partner looking to minimize input costs. The reaction conditions are notably mild, avoiding extreme temperatures or pressures, which simplifies the engineering requirements for reactor setups and enhances safety profiles. This novel approach not only improves the chemical efficiency but also aligns perfectly with green chemistry principles by reducing waste generation and energy consumption, thereby offering substantial cost savings in the long-term manufacturing lifecycle.
Mechanistic Insights into Reductive Oxy-Nazarov Electrocyclization
The chemical elegance of this synthesis lies in the precise orchestration of the reductive oxy-Nazarov electrocyclization, which serves as the pivotal step for skeleton construction. In this mechanism, a divinyl ketone precursor, generated from the initial aldol and Grignard additions, undergoes a 4-pi electrocyclization under reductive conditions facilitated by diisobutylaluminum hydride. This specific reduction potential allows for the selective formation of the cyclopentenone intermediate without over-reducing sensitive functional groups, demonstrating exceptional chemoselectivity. The subsequent intramolecular Aldol reaction, triggered by a base such as potassium hydroxide in methanol, closes the final ring to establish the rigid tricyclic framework characteristic of cedrol. This cascade of reactions is designed to maximize stereocontrol, ensuring that the resulting diastereomers possess the correct spatial arrangement required for downstream biological activity. For technical teams, understanding this mechanism is crucial for troubleshooting and optimizing reaction parameters to maintain consistent quality across different batch sizes.
Impurity control is another critical aspect where this methodology excels, particularly regarding the management of side reactions during the reduction and oxidation phases. The protocol incorporates specific protection strategies, such as the use of tert-butyldimethylchlorosilane to mask hydroxyl groups during the hydroboration-oxidation sequence. This prevents unwanted intramolecular reactions or polymerization that could otherwise lead to complex impurity profiles difficult to separate. Furthermore, the choice of zinc powder for reductive deoxygenation in acidic media provides a clean conversion that minimizes the formation of heavy metal waste, simplifying the workup procedure. By carefully controlling the stoichiometry of reagents like 9-BBN and Dess-Martin periodinane, the process ensures that over-oxidation or incomplete conversion is kept to a minimum. This rigorous attention to impurity profiles guarantees that the final tricyclic skeleton meets the stringent purity specifications demanded by regulatory bodies for pharmaceutical applications.
How to Synthesize Cedrol Tricyclic Skeleton Efficiently
Implementing this synthesis route requires a disciplined approach to reaction monitoring and intermediate isolation to ensure optimal yields at every stage. The process begins with the functionalization of cyclopentanone, followed by the critical cyclization events that define the molecular architecture. Detailed standard operating procedures for each transformation, including specific solvent grades and temperature ramping rates, are essential for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent data to assist process chemists in replicating this high-efficiency route.
- Initiate the synthesis by reacting cyclopentanone with ethyl formate and allylmagnesium chloride to form the key aldehyde intermediate.
- Perform a Horner-Wadsworth-Emmons reaction followed by a critical reductive oxy-Nazarov electrocyclization to build the [5.5] ring system.
- Complete the tricyclic skeleton formation through acetylation, reduction, protection, oxidation, and a final intramolecular Aldol cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible strategic advantages that go beyond simple chemical yield. The elimination of noble metal catalysts removes a major cost driver and supply risk, as the prices of metals like gold or palladium can be volatile and their sourcing subject to geopolitical constraints. By relying on abundant base metals and organic reagents, the manufacturing process becomes more resilient to market fluctuations, ensuring a stable cost structure for long-term contracts. Additionally, the simplified purification workflow reduces the consumption of solvents and chromatography media, which are significant contributors to the overall cost of goods sold in fine chemical manufacturing. This efficiency allows for a more competitive pricing model without compromising on the quality of the final intermediate.
- Cost Reduction in Manufacturing: The removal of expensive noble metal catalysts from the synthetic route directly lowers the raw material expenditure per kilogram of product. Furthermore, the reduced number of synthetic steps minimizes labor hours and utility consumption, such as heating and cooling loads, leading to substantial cost savings in the overall production budget. The use of inexpensive starting materials like cyclopentanone further enhances the economic viability of the process, making it an attractive option for cost-sensitive projects. This economic efficiency is achieved without sacrificing yield, as the high selectivity of the key reactions ensures that material throughput remains high.
- Enhanced Supply Chain Reliability: Sourcing reagents for this process is straightforward, as the required chemicals are commodity items available from multiple global vendors. This diversity in supply sources mitigates the risk of single-supplier dependency, which is a common vulnerability in complex pharmaceutical supply chains. The robustness of the reaction conditions means that production is less likely to be interrupted by equipment failures or safety incidents, ensuring consistent delivery schedules. For supply chain heads, this reliability is paramount for maintaining just-in-time inventory levels and meeting the demanding timelines of downstream drug development programs.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction types that are well-understood in large-scale reactor environments. The absence of hazardous high-pressure steps or pyrophoric reagents simplifies the safety validation process for new manufacturing lines. Moreover, the reduced generation of heavy metal waste aligns with increasingly strict environmental regulations, lowering the costs associated with waste disposal and treatment. This environmental compatibility enhances the corporate sustainability profile of the manufacturing partner, a key factor for multinational corporations evaluating their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cedrol synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity for technical decision-makers evaluating this route for their own production needs. Understanding these details is essential for assessing the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the primary advantages of this cedrol synthesis route over traditional methods?
A: Unlike conventional methods that rely on long synthetic routes and expensive noble metal catalysts, this patent-disclosed method utilizes cheap cyclopentanone and avoids noble metals entirely. This significantly reduces raw material costs and simplifies post-processing by eliminating heavy metal removal steps, making it highly suitable for industrial scale-up.
Q: How does this method ensure high purity for pharmaceutical applications?
A: The process employs specific protection and deprotection strategies, such as using tert-butyldimethylchlorosilane, alongside controlled reduction steps like Luche reduction. These measures effectively minimize side reactions and impurity formation, resulting in a tricyclic skeleton with high stereochemical integrity and purity suitable for downstream drug synthesis.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the method is explicitly designed for industrial feasibility. It uses mild reaction conditions, readily available reagents like zinc powder and common solvents, and avoids hazardous high-pressure or high-temperature steps. This ensures safety and consistency when transitioning from laboratory grams to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cedrol Supplier
The technical potential of this metal-free synthesis route is immense, offering a pathway to high-value sesquiterpene intermediates that was previously inaccessible due to cost and complexity constraints. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this patent technology to life. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of cedrol skeleton produced meets the exacting standards of the global pharmaceutical industry. We understand the critical nature of supply continuity and are committed to delivering consistent quality through our robust quality management systems.
We invite you to discuss how this innovative synthesis route can optimize your current supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments that will demonstrate the viability of this process for your next project. Let us partner with you to transform this scientific breakthrough into a commercial reality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
