Revolutionizing Chiral Alcohol Synthesis with Engineered Carbonyl Reductase Mutants for Industrial Scale
Revolutionizing Chiral Alcohol Synthesis with Engineered Carbonyl Reductase Mutants for Industrial Scale
The pharmaceutical industry is currently witnessing a paradigm shift towards biocatalysis for the synthesis of complex chiral intermediates, driven by the urgent need for greener and more efficient manufacturing processes. A groundbreaking development in this field is detailed in patent CN113801859A, which discloses a series of novel carbonyl reductase mutants specifically engineered to overcome the limitations of traditional chemical synthesis and wild-type enzymes. This technology focuses on the asymmetric reduction of ketone substrates to produce high-purity chiral alcohols, which are critical precursors for antiviral drugs such as Tenofovir Alafenamide. By leveraging directed evolution techniques, the inventors have created enzyme variants that demonstrate exceptional catalytic efficiency and stereoselectivity, addressing the longstanding challenges of low yield and poor enantiomeric excess that have plagued previous methods. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this patent represents a significant leap forward in process intensification and cost optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
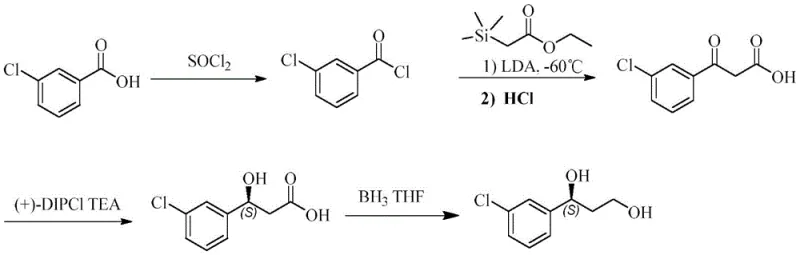
Historically, the synthesis of key chiral diol intermediates, such as those used in liver-targeting prodrugs, has relied heavily on traditional chemical routes that are fraught with inefficiencies and environmental hazards. As illustrated in the prior art, the conventional chemical synthesis often involves multi-step sequences utilizing hazardous reagents like thionyl chloride and strong bases such as LDA at cryogenic temperatures. Furthermore, the critical asymmetric reduction step typically requires expensive chiral catalysts, such as (+)-DIPCL, which not only drives up the raw material costs but also introduces complex operational difficulties regarding catalyst recovery and recycling. The resulting chiral purity from these chemical methods often hovers around 98%, necessitating additional and costly purification steps like recrystallization to meet the stringent registration requirements for pharmaceutical ingredients. These factors collectively result in a high production cost and a significant environmental footprint, making the conventional chemical route increasingly unsustainable for large-scale industrial applications.
The Novel Approach
In stark contrast to the cumbersome chemical pathways, the novel biocatalytic approach presented in the patent utilizes engineered carbonyl reductase mutants to achieve direct and highly selective reduction of the ketone substrate. This method operates under mild aqueous conditions, typically at temperatures between 25-35°C and neutral pH, eliminating the need for extreme temperatures and hazardous organic solvents. The core innovation lies in the specific amino acid mutations introduced into the enzyme structure, such as V45I, G141V, and I195L, which reshape the active site to better accommodate the substrate. This structural optimization allows the enzyme to tolerate substrate concentrations exceeding 100g/L, a metric that is crucial for economic viability in bulk manufacturing. By replacing toxic chemical reagents with a renewable biocatalyst, this approach not only simplifies the downstream processing to a simple extraction but also aligns perfectly with the global trend towards green chemistry and sustainable manufacturing practices in the fine chemical sector.

Mechanistic Insights into Carbonyl Reductase Mutant Catalysis
The exceptional performance of the disclosed carbonyl reductase mutants can be attributed to precise modifications in the protein sequence that enhance both substrate binding affinity and catalytic turnover. The patent highlights specific mutations, including G141A/V and I195L, which are strategically located near the enzyme's active pocket. These substitutions likely reduce steric hindrance and optimize the hydrophobic interactions between the enzyme and the aromatic ring of the substrate, thereby facilitating a more efficient hydride transfer from the cofactor NAD(P)H to the carbonyl group. The result is a dramatic improvement in stereoselectivity, consistently achieving ee values greater than 99.5% for the desired (S)-configuration. This level of precision is critical for pharmaceutical applications where the presence of the wrong enantiomer can lead to toxicity or reduced efficacy. Furthermore, the mutants exhibit robust stability in the presence of co-solvents like isopropanol, which is often used as a co-substrate for cofactor regeneration, ensuring that the enzyme remains active throughout the prolonged reaction times required for high conversion rates.
From an impurity control perspective, the high specificity of the enzymatic reaction inherently minimizes the formation of by-products that are common in chemical reductions, such as over-reduced species or racemized products. The biological system operates with a lock-and-key mechanism that discriminates sharply between the pro-chiral faces of the ketone, effectively suppressing the formation of the (R)-enantiomer. This intrinsic selectivity means that the crude product obtained after the reaction already possesses high optical purity, significantly reducing the burden on downstream purification units. For quality control teams, this translates to a cleaner impurity profile and a more predictable manufacturing process, reducing the risk of batch failures due to out-of-specification chiral purity. The ability to maintain high conversion rates (>99%) even at elevated substrate loadings further ensures that residual starting material is minimized, simplifying the final isolation of the target chiral alcohol intermediate.
How to Synthesize (S)-1-(3-Chlorophenyl)-1,3-propanediol Efficiently
The practical implementation of this technology involves a straightforward biotransformation process that can be easily integrated into existing fermentation and recovery infrastructure. The process begins with the preparation of the reaction mixture, where the ketone substrate (Formula IV) is suspended in a phosphate buffer system along with a co-substrate like glucose or isopropanol to drive the cofactor regeneration cycle. The engineered carbonyl reductase, either in the form of wet cells, crude enzyme powder, or immobilized enzyme, is then added to initiate the reduction. The reaction is typically conducted at a controlled temperature of 30-35°C with mechanical stirring to ensure adequate mass transfer, especially given the high substrate concentrations employed. Monitoring the reaction progress via HPLC allows for precise determination of the endpoint, usually achieved within 10 to 24 hours depending on the specific mutant and loading used. Once the conversion is complete, the product is extracted using organic solvents like ethyl acetate, followed by standard drying and concentration steps to yield the pure chiral alcohol.
- Prepare the reaction system by mixing the ketone substrate (Formula IV) with a phosphate buffer solution and a co-substrate such as glucose or isopropanol.
- Add the engineered carbonyl reductase mutant (e.g., WO08) along with a coenzyme regeneration system (like GDH) and the necessary cofactor (NAD+ or NADP+).
- Maintain the reaction at 25-35°C and pH 7.0-7.5 until conversion exceeds 95%, then extract the product using ethyl acetate.

Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology offers compelling economic and logistical benefits that directly impact the bottom line. The most significant advantage is the drastic reduction in raw material costs associated with the elimination of expensive chiral chemical catalysts and hazardous reagents. By switching to a biocatalytic route, manufacturers can avoid the volatility in pricing associated with precious metal catalysts and specialized chiral ligands, leading to a more stable and predictable cost structure. Additionally, the simplified work-up procedure, which often requires only extraction and concentration rather than complex chromatographic separations or multiple recrystallizations, significantly reduces solvent consumption and waste disposal costs. This streamlining of the process not only lowers the variable cost per kilogram but also shortens the overall production cycle time, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The transition from chemical to enzymatic synthesis removes the dependency on costly chiral auxiliaries and harsh reagents, resulting in substantial cost savings. The high catalytic efficiency of the mutants means that less enzyme is required per unit of product, further driving down the cost of goods sold. Moreover, the ability to run reactions at high substrate concentrations improves the volumetric productivity of the reactors, allowing for more product to be manufactured in the same footprint without capital expenditure on new equipment.
- Enhanced Supply Chain Reliability: Biocatalytic processes are generally more robust and easier to scale than complex chemical syntheses involving sensitive reagents. The use of recombinant E. coli strains for enzyme production ensures a consistent and renewable supply of the biocatalyst, mitigating the risk of supply disruptions common with specialty chemical suppliers. The mild reaction conditions also reduce the wear and tear on production equipment, leading to lower maintenance costs and higher asset availability, which is crucial for maintaining continuous supply to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful operation at substrate loads of over 100g/L, which is a key indicator of industrial feasibility. From an environmental standpoint, the reduction in hazardous waste and solvent usage simplifies regulatory compliance and reduces the carbon footprint of the manufacturing site. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental regulations but also enhances the brand value of the final pharmaceutical product among eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonyl reductase technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs.
Q: What are the key advantages of the carbonyl reductase mutants described in CN113801859A over wild-type enzymes?
A: The mutants exhibit significantly higher enzyme activity and stereoselectivity (ee > 99.5%), allowing for substrate concentrations exceeding 100g/L, which is critical for industrial viability compared to the insufficient activity of wild-type strains.
Q: How does this enzymatic route improve environmental compliance compared to chemical synthesis?
A: The biocatalytic method eliminates the need for hazardous chemical reducing agents and expensive chiral catalysts like (+)-DIPCL, drastically reducing toxic waste generation and simplifying the post-reaction purification process.
Q: Can this process be scaled for commercial production of Tenofovir Alafenamide intermediates?
A: Yes, the high tolerance to organic solvents and ability to operate at high substrate loading makes this method highly suitable for large-scale commercial manufacturing of key antiviral drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-1-(3-Chlorophenyl)-1,3-propanediol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic technologies described in CN113801859A and are fully equipped to leverage them for our clients' benefit. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities include rigorous QC labs and stringent purity specifications that guarantee the delivery of high-quality intermediates meeting the exacting standards of the global pharmaceutical industry. We are committed to providing a reliable supply of complex chiral building blocks, utilizing advanced biocatalytic methods to ensure consistency, purity, and cost-effectiveness for our partners.
We invite you to collaborate with us to explore how this innovative enzymatic route can optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in biocatalysis can add value to your next project.
