Scaling Chiral Synthesis: Advanced Biocatalytic Production of (R)-4-Chloro-3-Hydroxy-Butyrate Esters
Scaling Chiral Synthesis: Advanced Biocatalytic Production of (R)-4-Chloro-3-Hydroxy-Butyrate Esters
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methods for producing chiral building blocks, particularly those serving as precursors for high-value therapeutics like L-carnitine. A significant breakthrough in this domain is documented in Chinese Patent CN111172124A, which details a novel carbonyl reductase mutant derived from Saccharomyces cerevisiae. This innovation addresses the critical bottleneck of low substrate tolerance found in wild-type enzymes, offering a pathway to drastically improve manufacturing efficiency. By engineering specific amino acid substitutions, notably the replacement of phenylalanine with methionine at position 85 (F85M), researchers have unlocked the potential for high-concentration biocatalysis. This report analyzes the technical merits of this patented technology, providing strategic insights for R&D directors, procurement managers, and supply chain leaders looking to secure a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biocatalytic reduction of chloroacetoacetate to produce (R)-4-chloro-3-hydroxy-butyrate has been hindered by severe substrate inhibition when using wild-type carbonyl reductases such as YOL151W. Literature indicates that conventional processes are restricted to extremely low substrate concentrations, typically below 10 g/L or less than 1% by mass. This limitation imposes a massive burden on manufacturing infrastructure, requiring enormous reactor volumes to process meaningful quantities of material, which in turn leads to excessive water consumption and energy usage for heating and cooling. Furthermore, the low volumetric productivity necessitates complex and costly downstream processing steps to isolate the product from dilute aqueous streams. For procurement and supply chain teams, these inefficiencies translate into higher operational expenditures and extended lead times, making the traditional wild-type enzymatic route economically unviable for large-scale commercial applications where cost reduction in pharmaceutical intermediate manufacturing is paramount.
The Novel Approach
The patented technology introduces a paradigm shift by utilizing a site-directed mutant of the YOL151W enzyme that exhibits remarkably enhanced tolerance to high substrate loads. Unlike the wild-type counterpart, this engineered biocatalyst operates efficiently at substrate concentrations ranging from 10% to 30% (w/v), representing a thirty-fold increase in processing density. This dramatic improvement allows for the use of significantly smaller reactors to achieve the same output, thereby reducing capital expenditure and footprint. The process employs a biphasic system involving a water-immiscible organic solvent, such as toluene, which helps solubilize the hydrophobic substrate and product while protecting the enzyme from potential organic phase toxicity. By overcoming the substrate inhibition barrier, this novel approach transforms the synthesis of (R)-4-chloro-3-hydroxy-butyrate from a niche, low-yield laboratory procedure into a robust, industrially viable manufacturing process capable of meeting global demand.
Mechanistic Insights into YOL151W-Catalyzed Asymmetric Reduction
The core of this technological advancement lies in the precise protein engineering of the carbonyl reductase active site. The primary mutation involves substituting the phenylalanine residue at position 85 with methionine (F85M), which alters the steric and electronic environment of the substrate binding pocket. This modification facilitates better accommodation of the chloroacetoacetate molecule, reducing steric hindrance and enhancing the catalytic turnover rate. Additionally, the patent describes further optimization through mutations at positions 128, 132, and 162, which can synergistically improve enzyme performance. The reaction mechanism relies on the stereoselective transfer of a hydride ion from the cofactor NADPH to the prochiral ketone group of the substrate. To make this process economically feasible, the system incorporates a glucose dehydrogenase (GDH) coupled cycle that regenerates the expensive NADPH cofactor in situ using inexpensive glucose as the sacrificial electron donor. This cofactor recycling loop ensures that only catalytic amounts of NADP+ are required, significantly lowering the cost of goods.
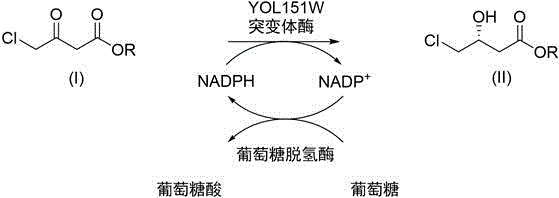
Furthermore, the control of impurities and stereoselectivity is intrinsic to the enzyme's chiral recognition capability. The mutant enzyme maintains excellent enantioselectivity, consistently producing the (R)-enantiomer with an enantiomeric excess (ee) of greater than 97%, as confirmed by chiral HPLC analysis of various ester derivatives. The use of a biphasic reaction system also aids in impurity control by extracting the product into the organic phase as it forms, which can prevent product inhibition and minimize side reactions such as non-enzymatic hydrolysis or racemization. The reaction conditions are mild, typically conducted at temperatures between 20°C and 50°C and a neutral pH range of 6.5 to 7.5, which preserves enzyme stability and minimizes the formation of thermal degradation byproducts. This high level of stereocontrol is critical for downstream synthesis of active pharmaceutical ingredients, where chirality directly impacts biological activity and regulatory compliance.
How to Synthesize (R)-4-Chloro-3-Hydroxy-Butyrate Efficiently
The implementation of this biocatalytic route requires a structured approach to strain construction and process optimization to ensure reproducibility and high yield. The patent outlines a comprehensive workflow starting from gene cloning to the final isolation of the chiral ester. It emphasizes the importance of co-expressing the mutant reductase and the glucose dehydrogenase in a robust host like E. coli BL21(DE3) to create a self-sufficient whole-cell biocatalyst. The following guide summarizes the critical operational parameters derived from the patent data, serving as a foundational reference for process development teams aiming to replicate or scale this technology.
- Construct recombinant E. coli BL21(DE3) co-expressing the YOL151W mutant (e.g., F85M) and glucose dehydrogenase (GDH) for cofactor regeneration.
- Cultivate the engineered strain in LB medium with antibiotics, induce expression with IPTG at 18°C, and harvest whole cells as the biocatalyst.
- Perform asymmetric reduction in a biphasic system (buffer/toluene) with high substrate loading (10-30% w/v) and glucose, maintaining pH 6.5-7.5.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for sourcing and logistics, the transition to this high-concentration biocatalytic process offers substantial strategic benefits beyond mere technical novelty. The ability to run reactions at 10-30% substrate concentration fundamentally changes the economics of production by drastically reducing the volume of water and solvents required per kilogram of product. This reduction in bulk translates directly into lower costs for waste treatment, energy consumption for heating and cooling, and storage requirements. Moreover, the elimination of transition metal catalysts, which are often used in chemical asymmetric hydrogenation, removes the need for expensive and time-consuming heavy metal scavenging steps. This simplification of the downstream process not only accelerates the manufacturing timeline but also ensures a cleaner product profile that meets stringent regulatory standards for residual metals in pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic increase in volumetric productivity. By processing thirty times more substrate per liter of reaction volume compared to wild-type methods, manufacturers can achieve the same output with a fraction of the infrastructure investment. The use of whole-cell biocatalysts eliminates the need for costly enzyme purification steps, as the cells themselves serve as the immobilized catalyst. Additionally, the in situ regeneration of the NADPH cofactor using glucose removes the dependency on stoichiometric amounts of expensive reducing agents. These factors combine to create a leaner, more cost-efficient manufacturing model that significantly lowers the cost of goods sold without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Securing a stable supply of chiral intermediates is critical for preventing bottlenecks in drug development and commercialization. This biocatalytic route utilizes readily available raw materials such as chloroacetoacetate and glucose, which are commodity chemicals with robust global supply chains. The engineered E. coli strains are stable and can be stored for long periods, ensuring consistent batch-to-batch performance. The mild reaction conditions reduce the risk of equipment failure or safety incidents associated with high-pressure or high-temperature chemical processes. Consequently, partners adopting this technology can expect shorter lead times for high-purity pharmaceutical intermediates and a more resilient supply chain capable of scaling rapidly to meet market surges.
- Scalability and Environmental Compliance: Sustainability is increasingly a key criterion for vendor selection in the pharmaceutical industry. This enzymatic process operates under green chemistry principles, utilizing aqueous buffers and recyclable organic solvents like toluene. The absence of toxic heavy metals and the generation of benign byproducts (gluconic acid from glucose oxidation) simplify wastewater treatment and reduce the environmental footprint of the facility. The process is inherently scalable; the biphasic system performs well in large stirred-tank reactors, and the whole-cell catalyst can be easily separated by centrifugation. This ease of scale-up from gram to ton scale ensures that the technology remains viable and compliant with environmental regulations as production volumes increase to meet commercial demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. They are derived from the specific experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is essential for evaluating the suitability of this route for your specific project requirements.
Q: What is the primary advantage of the YOL151W mutant over the wild-type enzyme?
A: The YOL151W mutant (specifically F85M) exhibits significantly improved catalytic activity towards chloroacetoacetate, allowing for substrate concentrations up to 30% (w/v), compared to less than 1% for the wild-type enzyme.
Q: What enantiomeric excess (ee) can be achieved with this biocatalytic process?
A: The process consistently achieves high stereoselectivity, with reported ee values exceeding 97% for various ester derivatives including ethyl, methyl, and tert-butyl esters.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of whole-cell biocatalysts in a biphasic system simplifies downstream processing and eliminates the need for expensive cofactor addition, making it highly scalable and cost-effective for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-4-Chloro-3-Hydroxy-Butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in modernizing the supply of critical chiral building blocks. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the YOL151W mutant system can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments, including chiral HPLC and GC-MS, to guarantee that every batch of (R)-4-chloro-3-hydroxy-butyrate meets the highest quality standards required by global regulatory agencies. Our commitment to technical excellence ensures that our clients receive materials that facilitate smooth downstream synthesis and regulatory filings.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cutting-edge technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-efficiency biocatalytic route. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. Let us help you optimize your manufacturing process, reduce costs, and secure a sustainable supply of high-quality intermediates for your most valuable products.
