Scalable Manufacturing of Stavudine Intermediates via Novel Zinc Reductive Elimination from 5-Methyluridine
Scalable Manufacturing of Stavudine Intermediates via Novel Zinc Reductive Elimination from 5-Methyluridine
The pharmaceutical industry continuously seeks robust and economically viable pathways for the large-scale production of antiretroviral agents, particularly for the treatment of HIV/AIDS. Patent CN1193021A presents a groundbreaking methodology for the synthesis of 2',3'-didehydro-3'-deoxythymidine, commonly known as stavudine or d4T, utilizing 5-methyluridine as the primary starting material. This innovation addresses critical bottlenecks in existing manufacturing processes by replacing expensive thymidine with a more accessible nucleoside precursor while introducing a highly efficient zinc-mediated reductive elimination step. The technical significance of this patent lies not only in the cost savings associated with raw materials but also in the strategic selection of reaction conditions that minimize side reactions such as glycosidic bond cleavage. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and scalable production capabilities for essential antiviral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of d4T has relied heavily on thymidine as the starting material, which imposes severe economic constraints due to the high cost and limited availability of this natural nucleoside. Alternative approaches attempting to utilize 5-methyluridine have faced significant technical hurdles, including the Corey-Winter thermal decomposition which requires hazardous reagents and complex thiocarbonyl derivatives, or the Eastwood reaction which often results in inefficient cleavage of the nuclear bond. A pervasive issue in these legacy methods is the formation of thymine as a stubborn byproduct, caused by the instability of the glycosidic bond under harsh reaction conditions, making purification extremely difficult and yield-limiting. Furthermore, many prior art methods necessitate selective pre-protection of the 5'-hydroxyl group, adding unnecessary synthetic steps, increasing solvent consumption, and prolonging the overall production cycle time. These inefficiencies collectively drive up the cost of goods sold and complicate the regulatory landscape regarding impurity profiles and residual solvents.
The Novel Approach
The methodology disclosed in the patent introduces a streamlined sequence that begins with the triple mesylation of 5-methyluridine followed by a base-catalyzed cyclization to form a stable 2,2'-anhydro intermediate. This approach cleverly bypasses the need for selective 5'-protection in the early stages by leveraging the differential reactivity of the hydroxyl groups during the mesylation and subsequent substitution steps. The core innovation is the generation of a novel cis-2'-halo-3'-sulfonate derivative, specifically the bromo-mesylate species, which serves as an ideal substrate for zinc-mediated reductive elimination. This specific intermediate configuration allows for the formation of the critical 2',3'-double bond with exceptional stereocontrol and minimal degradation of the nucleobase. By optimizing the reaction sequence to merge protection, functionalization, and elimination into a cohesive flow, the process drastically reduces the number of isolation steps and enhances the overall throughput suitable for industrial scale-up.
Mechanistic Insights into Zinc-Mediated Reductive Elimination
The heart of this synthetic strategy is the zinc reductive elimination step, which transforms the halogenated sugar moiety into the unsaturated d4T framework with high fidelity. The mechanism involves the activation of the zinc metal surface, often facilitated by acetic acid or copper coupling, to effectuate the simultaneous removal of the 2'-bromo and 3'-mesylate groups. Unlike hydride-based reductions which can be indiscriminate, the zinc system specifically targets the vicinal leaving groups to generate the alkene without reducing the uracil base or affecting the 5'-ester protecting group. The use of polar aprotic solvents mixed with alcohols creates an environment that stabilizes the transition state while ensuring the solubility of the intermediate species. This mechanistic precision is crucial for maintaining the integrity of the glycosidic bond, thereby preventing the release of free thymine which plagues other synthetic routes.
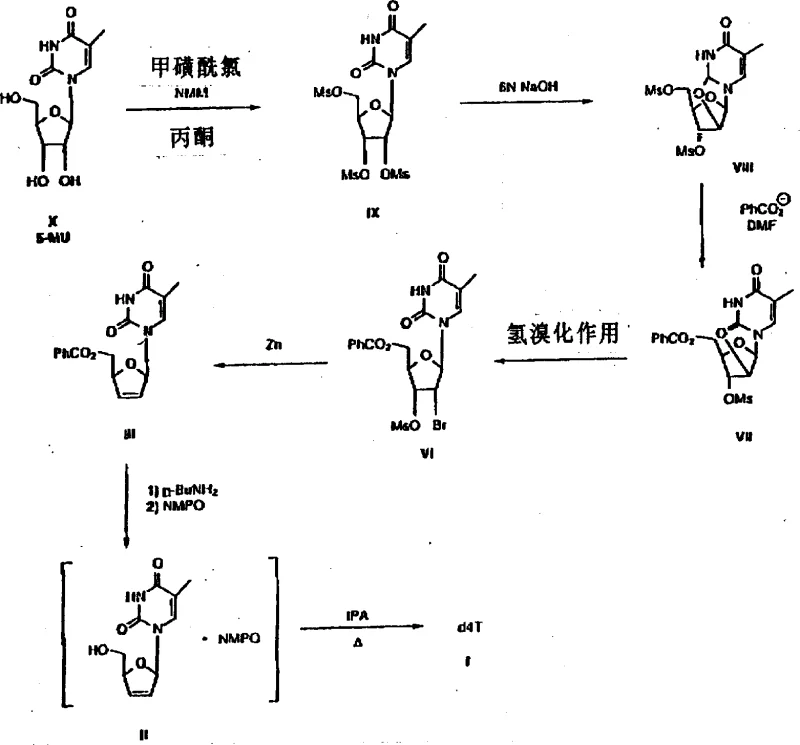
Following the formation of the double bond, the process employs a unique deprotection and purification strategy involving n-butylamine and N-methylpyrrolidone (NMPO). The removal of the 5'-benzoyl group using n-butylamine proceeds under conditions that avoid the formation of inorganic salts which are notoriously difficult to separate from water-soluble nucleosides. Instead of traditional aqueous workups that require ion-exchange resins, the product is converted into a crystalline NMPO solvate. This solvate formation acts as a powerful purification tool, effectively excluding organic impurities and unreacted starting materials from the crystal lattice. The subsequent desolvation in isopropanol yields the final active pharmaceutical ingredient with high purity, demonstrating a sophisticated understanding of solid-state chemistry to solve liquid-phase separation challenges.
How to Synthesize Stavudine (d4T) Efficiently
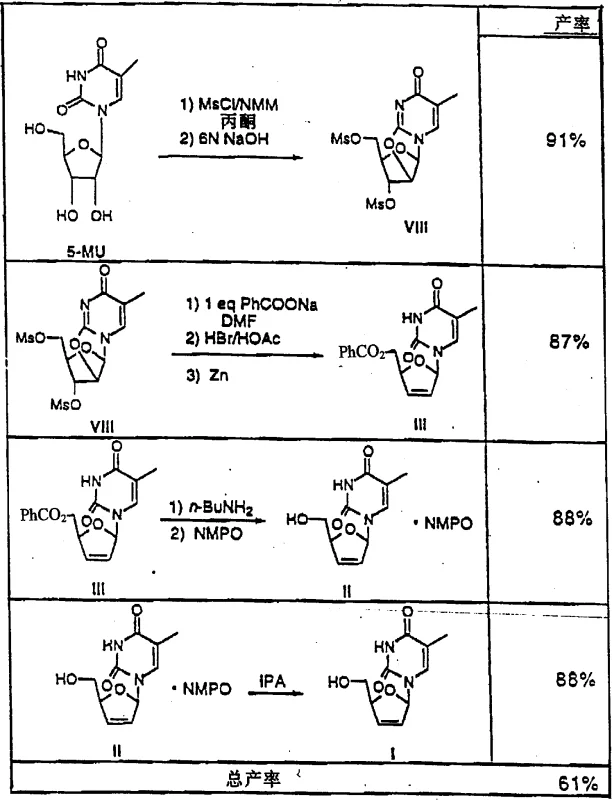
The synthesis of d4T according to this patent involves a carefully orchestrated series of transformations starting from the inexpensive 5-methyluridine. The initial phase requires the activation of all three hydroxyl groups via mesylation using methanesulfonyl chloride in the presence of an organic base such as N-methylmorpholine, followed by cyclization with sodium hydroxide to yield the anhydro sugar. Subsequent steps involve the regioselective opening of the anhydro ring with benzoate and bromination to install the leaving groups necessary for elimination. The detailed standardized synthetic steps for executing this high-yield pathway are outlined in the guide below, ensuring reproducibility and adherence to the patented parameters for temperature and stoichiometry.
- Mesylation of 5-methyluridine followed by cyclization to form the 2,2'-anhydro derivative using organic bases like N-methylmorpholine.
- Substitution of the 5'-mesyl group with benzoate, followed by hydrobromination to generate the cis-2'-bromo-3'-mesylate intermediate.
- Reductive elimination using zinc powder to form the 2',3'-double bond, followed by deprotection and purification via NMPO solvate formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from thymidine to 5-methyluridine as the feedstock represents a fundamental improvement in cost structure and supply security. 5-Methyluridine is a commodity chemical with a robust global supply chain, unlike thymidine which is subject to greater price volatility and sourcing constraints. The elimination of expensive reagents such as tributyltin hydride or complex phosphites further reduces the direct material costs associated with the synthesis. Additionally, the process avoids the use of chlorinated solvents like dichloromethane in the final isolation steps, replacing them with greener alternatives like isopropanol and ethyl acetate, which simplifies waste disposal and lowers environmental compliance costs. These factors combine to create a manufacturing profile that is not only cheaper but also more resilient to market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by consolidating multiple reaction steps and eliminating the need for chromatographic purifications through the innovative NMPO solvate crystallization technique. By avoiding the use of precious metal catalysts or stoichiometric amounts of toxic tin reagents, the expense of heavy metal scavenging and disposal is completely removed from the cost equation. The high yields reported in the patent examples indicate that less starting material is wasted per kilogram of final product, directly improving the atom economy and reducing the cost per unit of the active pharmaceutical ingredient. Furthermore, the ability to perform reactions at moderate temperatures without cryogenic cooling reduces energy consumption and infrastructure requirements in the production facility.
- Enhanced Supply Chain Reliability: Utilizing 5-methyluridine ensures a stable and diversified supply base, mitigating the risk of production stoppages due to raw material shortages. The simplified workflow, which merges protection and functionalization steps, shortens the overall lead time from raw material intake to finished intermediate. The robustness of the zinc reduction step means that the process is less sensitive to minor variations in reaction parameters, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers. This reliability is critical for pharmaceutical manufacturers who must maintain continuous production lines to meet global demand for antiretroviral therapies without interruption.
- Scalability and Environmental Compliance: The methodology is explicitly designed for large-scale production, evidenced by the use of common industrial solvents like acetone, DMF, and ethyl acetate which are easily handled in multi-ton reactors. The avoidance of strong acidic resins and the minimization of aqueous waste streams simplify the effluent treatment process, aligning with increasingly stringent environmental regulations. The crystallization of the NMPO solvate provides a scalable purification method that does not rely on labor-intensive column chromatography, facilitating a smooth transition from pilot plant to commercial manufacturing scales. This scalability ensures that the supply chain can rapidly expand capacity to meet surges in demand without compromising on product quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for d4T. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the technology for potential licensees and manufacturing partners. Understanding these nuances is essential for R&D teams evaluating process transfer and for procurement officers assessing the long-term viability of this supply source.
Q: Why is 5-methyluridine preferred over thymidine for d4T production?
A: 5-Methyluridine is significantly more economical and readily available compared to thymidine. The patented process overcomes previous limitations by avoiding expensive starting materials and complex protection/deprotection sequences associated with thymidine routes.
Q: How does the zinc reductive elimination step improve product purity?
A: Traditional methods often suffer from glycosidic bond cleavage leading to thymine byproducts which are difficult to remove. The specific zinc reductive elimination conditions described in the patent minimize this cleavage, resulting in higher yields and easier purification of the final nucleoside.
Q: What is the role of the NMPO solvate in the purification process?
A: The formation of the d4T·N-methylpyrrolidone (NMPO) solvate allows for effective crystallization and separation of the product from reaction impurities and salts. This step avoids the use of strong acidic resins and chlorinated solvents typically required in other deprotection methods, enhancing environmental compliance and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stavudine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient nucleoside synthesis in the global fight against viral diseases. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the zinc reductive elimination described in CN1193021A are executed with precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of d4T intermediate meets the highest international pharmacopoeia standards. We are committed to delivering high-purity pharmaceutical intermediates that empower our clients to bring life-saving medications to market faster and more cost-effectively.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to serve as your trusted and reliable stavudine supplier for both clinical and commercial stage requirements.
