Advanced Large-Scale Preparation of d4T for Global Pharmaceutical Supply Chains
Advanced Large-Scale Preparation of d4T for Global Pharmaceutical Supply Chains
The global demand for antiretroviral therapies continues to drive the need for efficient, scalable, and safe manufacturing processes for key nucleoside analogues. Patent CN1043646C introduces a groundbreaking methodology for the large-scale preparation of 2',3'-didehydro-3'-deoxythymidine, commonly known as d4T or Stavudine. This intellectual property represents a significant leap forward in process chemistry, specifically addressing the bottlenecks that have historically plagued the industrial synthesis of this critical pharmaceutical intermediate. By shifting away from hazardous reagents and complex purification protocols, this technology offers a streamlined pathway that aligns perfectly with modern Good Manufacturing Practice (GMP) standards. For R&D directors and process engineers, the core innovation lies in the strategic use of an N-Methylpyrrolidone (NMP) solvate for purification, a technique that dramatically simplifies downstream processing while ensuring exceptional product quality.
Furthermore, the economic implications of this patented route cannot be overstated for procurement specialists managing tight budgets. Traditional methods often rely on expensive chromatographic separations or toxic solvents that incur high disposal costs and regulatory burdens. In contrast, the disclosed method utilizes readily available starting materials and common organic solvents, creating a robust framework for cost reduction in API manufacturing. The ability to produce high-purity d4T without the need for column chromatography or dangerous reagents like hexamethylphosphoramide (HMPA) positions this technology as a superior choice for long-term supply contracts. As we delve deeper into the technical specifics, it becomes clear that this process is not merely a laboratory curiosity but a fully engineered solution designed for the rigors of commercial chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of d4T has been fraught with challenges that make large-scale operation economically and environmentally untenable. One of the earliest and most cited approaches, often referred to as Route 1, relies on the dehydration of nucleoside intermediates using potassium tert-butoxide in dimethyl sulfoxide (DMSO). 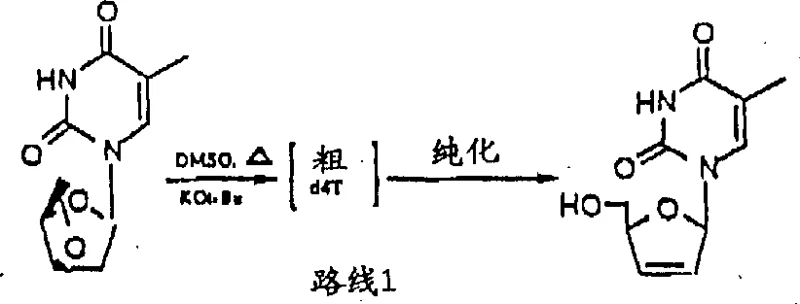 As illustrated in the reaction scheme above, this method requires highly basic conditions and elevated temperatures that frequently lead to product decomposition. From a process engineering perspective, the removal of DMSO is a notorious bottleneck; it has a high boiling point and tends to degrade the product during distillation, necessitating complex workup procedures that lower overall yield. Moreover, the sensitivity of the reagents to moisture adds a layer of operational complexity that increases the risk of batch failure in a manufacturing setting.
As illustrated in the reaction scheme above, this method requires highly basic conditions and elevated temperatures that frequently lead to product decomposition. From a process engineering perspective, the removal of DMSO is a notorious bottleneck; it has a high boiling point and tends to degrade the product during distillation, necessitating complex workup procedures that lower overall yield. Moreover, the sensitivity of the reagents to moisture adds a layer of operational complexity that increases the risk of batch failure in a manufacturing setting.
Other historical attempts, such as the selenium-mediated elimination shown in Route 4, introduce even greater liabilities regarding safety and waste management. The use of phenyl selenyl derivatives generates toxic selenium waste, which requires specialized handling and disposal protocols that are prohibitively expensive for high-volume production. Similarly, methods utilizing HMPA as a solvent pose severe health risks due to its carcinogenic nature, making it increasingly difficult to justify its use in a regulated pharmaceutical environment. These conventional pathways often result in oily residues that are difficult to crystallize, forcing manufacturers to rely on preparative chromatography—a technique that simply does not translate well to multi-ton scales due to solvent consumption and throughput limitations.
The Novel Approach
The methodology disclosed in CN1043646C fundamentally reimagines the synthetic trajectory by introducing a stable anhydro intermediate and a unique solvate-based purification strategy. Instead of struggling with the removal of high-boiling polar aprotic solvents, this novel approach leverages the formation of a 3',5'-anhydro thymidine intermediate, which is surprisingly stable under the subsequent elimination conditions. The process replaces hazardous reagents with safer alternatives, such as using aqueous sodium hydroxide for the cyclization step and potassium hydroxide in isopropanol for the final elimination. This shift not only mitigates safety risks but also simplifies the reaction workup, as inorganic salts can be easily filtered off. The true genius of this approach, however, lies in the final purification step where the crude product is converted into an N-Methylpyrrolidone (NMP) solvate.
This solvate formation acts as a highly effective crystallization driver, allowing the product to precipitate out of the solution while leaving impurities behind in the mother liquor. This eliminates the need for chromatographic purification entirely, a massive advantage for scaling up. The solid solvate can then be thermally degraded in alcohol to release the pure free base of d4T. This sequence ensures that the final product meets stringent purity specifications without the logistical nightmare of solvent swaps or column loading. For supply chain managers, this translates to a more predictable production timeline and a significant reduction in the volume of hazardous waste generated per kilogram of product, aligning with green chemistry principles and reducing environmental compliance costs.
Mechanistic Insights into Base-Catalyzed Elimination and Solvate Crystallization
To fully appreciate the robustness of this synthesis, one must understand the mechanistic nuances that allow it to succeed where others fail. The core transformation involves the conversion of a 3',5'-anhydro intermediate into the unsaturated d4T structure via a base-catalyzed elimination reaction.  Unlike previous methods where the nucleoside base or sugar moiety might degrade under harsh alkaline conditions, this patent reveals that the d4T product exhibits remarkable stability when heated in an alcoholic potassium hydroxide solution. This stability is counter-intuitive given the sensitivity of glycosidic bonds in basic media, suggesting that the specific electronic environment of the anhydro ring facilitates a clean E2-type elimination without compromising the integrity of the nucleobase. The use of isopropanol as the solvent is critical here, as it provides the necessary solubility for the intermediate while acting as a proton source that helps moderate the basicity of the hydroxide ions.
Unlike previous methods where the nucleoside base or sugar moiety might degrade under harsh alkaline conditions, this patent reveals that the d4T product exhibits remarkable stability when heated in an alcoholic potassium hydroxide solution. This stability is counter-intuitive given the sensitivity of glycosidic bonds in basic media, suggesting that the specific electronic environment of the anhydro ring facilitates a clean E2-type elimination without compromising the integrity of the nucleobase. The use of isopropanol as the solvent is critical here, as it provides the necessary solubility for the intermediate while acting as a proton source that helps moderate the basicity of the hydroxide ions.
Following the elimination, the purification mechanism relies on the specific intermolecular interactions between the d4T molecule and N-Methylpyrrolidone (NMP). The formation of the d4T-NMP solvate is a thermodynamically favored process under the described conditions, likely driven by hydrogen bonding between the hydroxyl groups of the sugar and the carbonyl oxygen of the lactam ring in NMP. This interaction lowers the solubility of the product in the presence of non-polar or moderately polar anti-solvents like acetone or ethyl acetate, causing it to crash out of solution as a defined crystalline solid. This crystallization is highly selective; impurities that do not form similar stable solvates remain dissolved in the filtrate. Subsequent heating of this solvate in alcohol breaks these intermolecular bonds, releasing the pure d4T and allowing the NMP to be recovered or removed, thus closing the loop on a highly efficient purification cycle that ensures consistent quality batch after batch.
How to Synthesize d4T Efficiently
The practical execution of this synthesis is broken down into distinct, manageable unit operations that are well-suited for standard reactor setups found in most fine chemical facilities. The process begins with the activation of thymidine through mesylation, followed by cyclization to the anhydro species, and concludes with the elimination and solvate isolation steps. Each stage has been optimized to minimize side reactions and maximize throughput. For process chemists looking to implement this technology, the key lies in strict control of pH and temperature during the hydrolysis and elimination phases to prevent the formation of degradation byproducts. The following guide outlines the standardized workflow derived from the patent examples, providing a clear roadmap from raw materials to the final high-purity active pharmaceutical ingredient.
- Perform mesylation of thymidine using methanesulfonyl chloride and an organic base like N-methylmorpholine in acetone to form the bis-mesylate intermediate.
- Convert the bis-mesylate to the 3',5'-anhydro thymidine intermediate using aqueous sodium hydroxide under controlled pH and temperature conditions.
- Execute the elimination reaction using potassium hydroxide in isopropanol, followed by isolation of the N-Methylpyrrolidone (NMP) solvate for high-purity final product recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented process offers tangible benefits that extend far beyond simple yield improvements. The primary value proposition is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By eliminating the need for chromatography, the facility can process significantly larger batches in the same amount of time, effectively increasing capacity without capital investment in new equipment. Furthermore, the substitution of toxic solvents like HMPA and DMSO with greener alternatives like acetone and isopropanol reduces the burden on waste treatment facilities and lowers the cost of regulatory compliance. This makes the supply chain more resilient against environmental regulations that often disrupt the availability of restricted chemicals.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive and time-consuming purification steps. Traditional routes often require silica gel chromatography, which consumes vast quantities of solvents and stationary phase, driving up the cost per kilogram exponentially. By replacing this with a crystallization-based purification via the NMP solvate, the process achieves substantial cost savings. Additionally, the use of inexpensive inorganic bases like potassium hydroxide and sodium hydroxide, instead of costly organic bases or metal catalysts, further drives down the raw material bill. The high yields reported in the patent examples mean that less starting material is wasted, optimizing the atom economy and ensuring that every dollar spent on thymidine translates efficiently into final product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialty reagents that have limited suppliers or long lead times. This method mitigates that risk by utilizing commodity chemicals that are readily available in the global market. Acetone, isopropanol, methanesulfonyl chloride, and potassium hydroxide are produced at massive scales worldwide, ensuring that production is never halted due to a shortage of a niche reagent. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, reducing the likelihood of batch failures that can disrupt delivery schedules. This reliability is crucial for maintaining the steady flow of materials needed for downstream drug formulation.
- Scalability and Environmental Compliance: Scaling a chemical process from the bench to the plant floor often reveals hidden dangers, particularly regarding heat management and exothermic reactions. This patented route is explicitly designed for scale-up, with reaction conditions that are manageable in large vessels. The avoidance of highly exothermic steps and the use of non-flammable or low-flammability solvents where possible enhances plant safety. From an environmental standpoint, the reduction in hazardous waste generation is significant. By avoiding selenium waste and minimizing DMSO discharge, the facility can meet stricter environmental standards with less effort. This 'green' profile not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain, a factor that is increasingly important for major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
Understanding the technical details of a new manufacturing process is essential for making informed sourcing decisions. The following questions address common concerns regarding the implementation and benefits of this specific d4T synthesis route. These answers are derived directly from the technical disclosures within the patent documentation, ensuring accuracy and relevance for technical evaluators. Whether you are concerned about impurity profiles, scalability limits, or regulatory acceptance, these insights provide a solid foundation for assessing the viability of this technology for your specific supply chain needs.
Q: Why is the N-Methylpyrrolidone (NMP) solvate step critical in this process?
A: The formation of the d4T-NMP solvate serves as a powerful purification mechanism. It allows for the selective crystallization of the product while leaving difficult-to-remove impurities in the mother liquor, significantly enhancing purity without requiring chromatography.
Q: How does this method improve safety compared to traditional routes?
A: This method eliminates the use of highly toxic and carcinogenic reagents such as HMPA (hexamethylphosphoramide) and organoselenium compounds found in prior art. Additionally, it avoids the use of large volumes of DMSO, which is difficult to remove and poses thermal hazards on a large scale.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the patent explicitly details improvements for scale-up, including the use of common solvents like acetone and isopropanol, and robust reaction conditions that tolerate heating in alkaline alcohol solutions, making it ideal for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable d4T Supplier
The technological advancements detailed in patent CN1043646C represent a significant opportunity for pharmaceutical companies seeking a more efficient and sustainable source of antiretroviral intermediates. At NINGBO INNO PHARMCHEM, we have closely analyzed this methodology and integrated its core principles into our own manufacturing capabilities to ensure we deliver the highest quality products to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full commercialization is seamless and risk-free. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify that every batch of d4T meets or exceeds international pharmacopoeia standards.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis route can benefit your specific projects. By leveraging our expertise in nucleoside chemistry, we can offer a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to our supply chain can reduce your overall cost of goods sold. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, high-quality, and cost-effective supply of d4T that supports your mission to bring life-saving medications to patients worldwide.
