Advanced Green Synthesis of Sotorasib Intermediates for Commercial Scale Production
Advanced Green Synthesis of Sotorasib Intermediates for Commercial Scale Production
The pharmaceutical industry is currently witnessing a paradigm shift in the treatment of KRAS-mutated cancers, driven largely by the development of small molecule inhibitors like AMG510 (Sotorasib). As demand for this critical oncology therapy surges, the efficiency and sustainability of its supply chain have become paramount concerns for global stakeholders. Patent CN114907259A introduces a groundbreaking synthetic methodology for key Sotorasib intermediates that addresses historical bottlenecks in yield, cost, and environmental safety. This technical insight report analyzes the proprietary advancements detailed in the patent, specifically focusing on the streamlining of the nicotinamide precursor synthesis and the safer construction of the urea linkage. By transitioning from multi-step halogenation sequences to direct hydrolysis and replacing hazardous gaseous reagents with solid alternatives, this technology offers a robust pathway for reliable API intermediate supplier networks seeking to optimize their manufacturing portfolios.
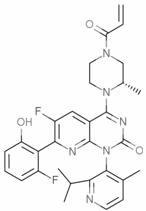
The structural complexity of AMG510 necessitates precise control over impurity profiles, particularly regarding halogenated by-products and heavy metal residues. The patent outlines a comprehensive strategy that not only enhances the chemical purity of the final intermediate—reported at 99.8% in exemplary runs—but also fundamentally alters the economic landscape of its production. For R&D directors and procurement managers alike, understanding these mechanistic shifts is crucial for evaluating long-term supply security. The integration of these green chemistry principles ensures that the production of high-purity pharmaceutical intermediates can be scaled without compromising on safety or regulatory compliance, positioning manufacturers who adopt these methods as leaders in the competitive oncology market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical 2,6-dichloro-5-fluoronicotinamide building block has been plagued by inefficiency and environmental hazards. Traditional routes often necessitate a cumbersome three-step reaction sequence to install the amide functionality, which inherently accumulates waste and lowers overall throughput. Furthermore, the conventional preparation of the pyridine amine fragment typically relies on 2-bromo starting materials. While bromine is an effective leaving group, it is significantly more expensive than chlorine and generates toxic bromination by-products that require complex and costly waste treatment protocols. Additionally, the final urea coupling step in legacy processes frequently employs oxalyl chloride, a reagent known to release corrosive hydrogen chloride gas and toxic carbon monoxide during activation. These factors collectively create a high-barrier entry for cost reduction in pharmaceutical manufacturing, posing significant risks to both operational safety and profit margins.
The Novel Approach
The innovative methodology presented in the patent radically simplifies this landscape by introducing a one-step sulfuric acid hydrolysis technique. This approach converts 2,6-dichloro-5-fluoronicotinonitrile directly into the desired amide, bypassing two intermediate steps and achieving exceptional yields of up to 96%. Simultaneously, the synthesis of the amine fragment is optimized by utilizing cheaper 2-chloro compounds as starting materials, facilitated by a specialized zinc-mediated coupling system that maintains high reactivity without the environmental penalty of bromine. Perhaps most critically, the urea formation is executed using bis(trichloromethyl) carbonate, commonly known as solid phosgene. This solid reagent is easier to handle, does not damage equipment through gas corrosion, and eliminates the immediate release of toxic gases, thereby drastically improving the safety profile of the entire operation.
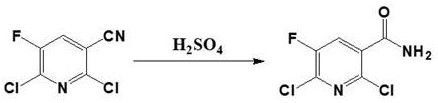
Mechanistic Insights into Acid-Catalyzed Hydrolysis and Urea Coupling
The core of this technological advancement lies in the rigorous optimization of reaction conditions to favor thermodynamic stability and kinetic efficiency. In the hydrolysis step, concentrated sulfuric acid acts as both a solvent and a catalyst, protonating the nitrile nitrogen to facilitate nucleophilic attack by water molecules upon quenching. This mechanism avoids the formation of unstable imidate intermediates that often plague milder hydrolysis conditions, ensuring a clean conversion to the primary amide. The subsequent purification via recrystallization from ethyl acetate further refines the product, removing trace acidic residues and unreacted nitrile to achieve the reported 99.7% purity. This level of control is essential for preventing downstream side reactions during the final coupling, where even minor impurities could lead to difficult-to-remove urea derivatives.
Regarding the final assembly of the Sotorasib intermediate, the mechanism involves the in situ generation of an isocyanate or activated carbamoyl chloride species from the amide using solid phosgene. Triethylamine serves as a proton scavenger, neutralizing the HCl generated during the activation and maintaining the basicity required for the nucleophilic attack by the pyridine amine. The use of a dichloromethane and chloroform solvent system provides the necessary solubility for both polar and non-polar intermediates, ensuring a homogeneous reaction environment. This precise control over the reaction milieu minimizes the formation of symmetric urea by-products, a common issue in intermolecular couplings. The result is a highly selective transformation that delivers the target molecule with a yield of 92.4%, demonstrating the robustness of this catalytic and stoichiometric balance.
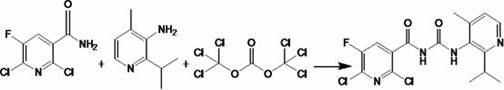
How to Synthesize Sotorasib Intermediate Efficiently
The implementation of this synthetic route requires careful attention to temperature control and reagent addition rates to maximize safety and yield. The process begins with the exothermic hydrolysis reaction, followed by the preparation of the organozinc reagent under inert atmosphere to prevent oxidation. Finally, the coupling reaction must be maintained at low temperatures initially to control the evolution of gases before warming to complete the transformation. Detailed standard operating procedures regarding specific molar ratios, stirring speeds, and work-up protocols are essential for reproducibility at scale. For a comprehensive guide on executing these steps with precision, please refer to the standardized synthesis instructions provided below.
- Perform one-step sulfuric acid hydrolysis of 2,6-dichloro-5-fluoronicotinonitrile at 50°C to obtain the amide precursor with high purity.
- Synthesize 2-isopropyl-4-methylpyridin-3-amine via zinc-mediated coupling using 2-chloro-3-amino-4-methylpyridine and a Grignard-derived zinc reagent.
- Couple the amine and amide precursors using bis(trichloromethyl) carbonate (solid phosgene) in dichloromethane/chloroform with triethylamine base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into tangible operational benefits and risk mitigation. The shift from a three-step to a one-step synthesis for the nicotinamide precursor significantly reduces the consumption of solvents, energy, and labor hours, leading to substantial cost savings in the overall manufacturing budget. By eliminating the need for expensive brominated starting materials and replacing them with readily available chloro-analogs, the raw material cost base is lowered while simultaneously simplifying the sourcing strategy. This reduction in material complexity enhances supply chain resilience, as chloro-compounds are generally more abundant and less subject to volatile market pricing than their brominated counterparts.
- Cost Reduction in Manufacturing: The consolidation of reaction steps and the substitution of high-cost reagents with economical alternatives drive down the cost of goods sold (COGS). Eliminating the oxalyl chloride step removes the need for specialized gas scrubbing equipment and reduces maintenance costs associated with acid corrosion. Furthermore, the high yields reported in the patent examples mean less raw material is wasted per kilogram of finished product, optimizing the atom economy of the process. These cumulative efficiencies allow for a more competitive pricing structure without sacrificing margin, making the final API more accessible to the market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like sulfuric acid and chloro-derivatives rather than specialized brominated intermediates reduces dependency on niche suppliers. The simplified process flow also shortens the production cycle time, allowing manufacturers to respond more agilely to fluctuations in market demand. With fewer unit operations and purification stages, the potential for batch failures due to equipment malfunction or human error is minimized. This reliability ensures a consistent flow of high-quality intermediates, securing the production schedule for the final drug substance and preventing costly delays in the pharmaceutical pipeline.
- Scalability and Environmental Compliance: The replacement of toxic gaseous reagents with solid phosgene significantly eases the regulatory burden associated with emissions and worker safety. This makes the process more scalable, as the safety constraints that often limit batch sizes in traditional methods are relaxed. The reduction in hazardous waste streams, particularly bromine-containing effluents, simplifies wastewater treatment and aligns the manufacturing process with increasingly stringent global environmental standards. This commitment to green chemistry not only protects the environment but also future-proofs the supply chain against tightening regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, safety protocols, and material substitutions. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: How does the new hydrolysis method improve efficiency compared to traditional routes?
A: The patented method simplifies the synthesis of 2,6-dichloro-5-fluoronicotinamide from a three-step process into a single-step sulfuric acid hydrolysis, drastically reducing reaction time and waste generation while achieving yields up to 96%.
Q: What safety advantages does using solid phosgene offer over oxalyl chloride?
A: Replacing oxalyl chloride with non-toxic solid phosgene eliminates the generation of hazardous hydrogen chloride and carbon monoxide gases during the urea coupling step, significantly enhancing operational safety and reducing equipment corrosion risks.
Q: Why is the switch from bromo- to chloro-starting materials beneficial for supply chains?
A: Utilizing 2-chloro compounds instead of expensive 2-bromo analogs lowers raw material costs and reduces the environmental burden associated with bromination by-products, ensuring a more sustainable and cost-effective supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sotorasib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the rigorous demands of the modern pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and utilizing our rigorous QC labs to verify that every batch of Sotorasib intermediate meets the highest global standards. Our infrastructure is designed to handle complex chemistries safely, leveraging the very green chemistry principles outlined in recent patents to deliver superior value.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your partner in delivering high-quality oncology intermediates with speed, safety, and sustainability.
