Advanced Nickel-Catalyzed Synthesis of Sotorasib Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and cost-effective pathways for the synthesis of complex oncology therapeutics, particularly for KRAS G12C inhibitors like Sotorasib. Patent CN113603636A introduces a groundbreaking preparation method for the key intermediate 2-isopropyl-3-amino-4-methylpyridine, addressing critical bottlenecks in existing manufacturing protocols. This novel approach replaces expensive palladium-catalyzed systems with a more economical nickel or iron-catalyzed cross-coupling strategy, fundamentally altering the cost structure of the supply chain. By leveraging readily available industrial feedstocks such as 3-cyano-4-methyl-2-pyridone, the process eliminates the need for difficult purification steps associated with positional isomers. For R&D directors and procurement managers alike, this patent represents a significant opportunity to optimize the production of high-purity pharmaceutical intermediates while ensuring long-term supply stability.
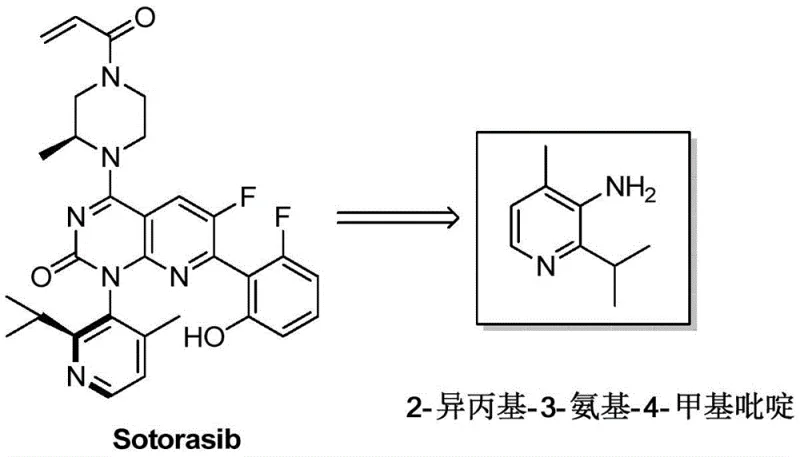
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating 2-isopropyl-3-amino-4-methylpyridine have historically relied on Negishi coupling reactions, which present substantial challenges for large-scale commercial manufacturing. These legacy methods typically utilize 2-bromo-3-amino-4-methylpyridine as a starting material, requiring a palladium catalyst and isopropyl zinc bromide to install the necessary alkyl group. The reliance on palladium introduces severe cost pressures due to the high price of the metal and the stringent requirement for anaerobic reaction conditions to prevent catalyst deactivation. Furthermore, alternative routes involving direct nitration of the pyridine ring often suffer from poor regioselectivity, leading to the formation of unavoidable positional isomers that require complex and yield-reducing rectification purification processes. These factors combined result in a fragile supply chain with high operational expenditures and significant technical risks regarding batch-to-batch consistency.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113603636A offers a streamlined and economically superior pathway that circumvents the drawbacks of precious metal catalysis. The new process initiates with the halogenation of 3-cyano-4-methyl-2-pyridone, a substrate where the substitution pattern is already fixed, thereby inherently preventing the formation of regioisomers. The core innovation lies in the subsequent cross-coupling step, which employs an isopropyl Grignard reagent in the presence of a nickel or iron catalyst rather than expensive palladium and organozinc species. This shift not only drastically reduces raw material costs but also simplifies the operational requirements by removing the need for extreme anaerobic environments. The resulting synthetic route is characterized by high atom economy and operational simplicity, making it ideally suited for the reliable manufacturing of complex pyridine derivatives at a commercial scale.
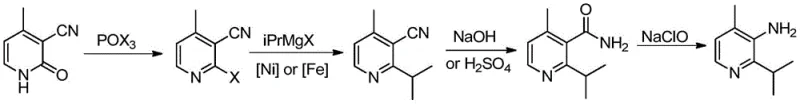
Mechanistic Insights into Nickel-Catalyzed Cross-Coupling and Hofmann Rearrangement
The heart of this innovative synthesis lies in the transition metal-catalyzed cross-coupling reaction, which efficiently installs the isopropyl moiety onto the pyridine core. Mechanistically, the nickel catalyst, such as 1,2-bis(diphenylphosphinoethane)nickel chloride, undergoes oxidative addition with the 2-halo-3-cyano-4-methylpyridine intermediate to form a reactive organonickel species. This is followed by transmetallation with the isopropyl magnesium halide Grignard reagent, transferring the alkyl group to the metal center. The final reductive elimination step releases the desired 2-isopropyl-3-cyano-4-methylpyridine product and regenerates the active catalyst. This catalytic cycle is highly efficient and tolerant of the functional groups present on the pyridine ring, ensuring high conversion rates without the need for exotic ligands or extreme temperatures that are often required in palladium chemistry.
Following the coupling, the process employs a classic yet highly effective Hofmann rearrangement to convert the nitrile-derived amide into the primary amine. The hydrolysis of the nitrile group first yields 2-isopropyl-4-methylnicotinamide, which is then subjected to basic conditions with sodium hypochlorite. This reaction proceeds through the formation of an N-chloroamide intermediate, which rearranges to an isocyanate before hydrolyzing to the final amine. From an impurity control perspective, this sequence is advantageous because the initial halogenation and coupling steps establish the carbon skeleton with high fidelity, minimizing the risk of structural impurities. The mild conditions of the Hofmann rearrangement, typically conducted at 0-10°C, further suppress side reactions, ensuring that the final API intermediate meets the stringent purity specifications required for oncology drug manufacturing.
How to Synthesize 2-isopropyl-3-amino-4-methylpyridine Efficiently
The synthesis of this critical oncology intermediate is achieved through a logical four-step sequence that prioritizes safety, yield, and scalability. The process begins with the activation of the pyridone ring using phosphorus oxychloride, followed by the pivotal nickel-catalyzed alkylation. Subsequent hydrolysis and rearrangement steps finalize the functional group installation. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures derived from the patent examples, are outlined in the guide below for technical reference.
- React 3-cyano-4-methyl-2-pyridone with phosphorus oxychloride to form the 2-halo intermediate.
- Perform nickel-catalyzed cross-coupling with isopropyl Grignard reagent to install the isopropyl group.
- Hydrolyze the nitrile group to an amide followed by Hofmann rearrangement to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into enhanced operational resilience and significant cost optimization. By eliminating the dependency on palladium catalysts and organozinc reagents, manufacturers can insulate their production costs from the volatility of precious metal markets. The use of commodity chemicals like Grignard reagents and nickel salts ensures a stable and diverse supplier base, reducing the risk of single-source bottlenecks. Furthermore, the simplified purification requirements resulting from improved regioselectivity mean that fewer processing units and less solvent are needed, which streamlines the overall manufacturing footprint and reduces waste disposal costs.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with abundant nickel or iron catalysts represents a fundamental shift in the cost basis of the intermediate. Since the Grignard reagents used are significantly cheaper and easier to handle than organozinc compounds, the direct material costs are substantially lowered. Additionally, the avoidance of complex rectification columns for isomer separation reduces energy consumption and capital expenditure on specialized distillation equipment, leading to a leaner and more profitable production model.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as 3-cyano-4-methyl-2-pyridone and phosphorus oxychloride, are widely available industrial chemicals with established global supply chains. This contrasts sharply with the specialized and often scarce reagents needed for Negishi coupling. By relying on commoditized inputs, the supply chain becomes more robust against disruptions, ensuring consistent delivery schedules for downstream API manufacturers who depend on timely intermediate supplies for their clinical and commercial batches.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are温和 and operate within standard temperature ranges that are easily manageable in large-scale reactors. The removal of heavy metal palladium from the process simplifies the environmental compliance landscape, as there is no need for rigorous and costly heavy metal scavenging steps to meet residual metal limits in the final drug substance. This facilitates a smoother regulatory approval process and aligns with modern green chemistry principles by reducing the overall toxic load of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this technology for commercial production.
Q: Why is the Nickel-catalyzed route preferred over Palladium for this intermediate?
A: The Nickel-catalyzed route utilizes significantly cheaper catalysts and Grignard reagents compared to the expensive Palladium and organozinc reagents required in conventional Negishi coupling, drastically reducing raw material costs.
Q: How does this method improve regioselectivity?
A: By starting with 3-cyano-4-methyl-2-pyridone, the substitution pattern is pre-determined, avoiding the formation of positional isomers often encountered during direct nitration of the pyridine ring in older methods.
Q: What are the critical reaction conditions for the Hofmann rearrangement step?
A: The rearrangement requires precise temperature control between 0-10°C using sodium hypochlorite and sodium hydroxide to ensure high yield and prevent side reactions during the conversion of the amide to the amine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-isopropyl-3-amino-4-methylpyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the race to deliver life-saving oncology treatments to patients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and robust. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-isopropyl-3-amino-4-methylpyridine meets the highest quality standards required by global regulatory agencies.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology for their Sotorasib supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and security for your critical drug development programs.
