Advanced FeCl3-Catalyzed Synthesis of Ganaxolone for Commercial Pharmaceutical Manufacturing
Advanced FeCl3-Catalyzed Synthesis of Ganaxolone for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways for central nervous system (CNS) therapeutics, particularly for high-demand candidates like ganaxolone. Patent CN102549007A introduces a transformative approach to synthesizing 3α-hydroxy, 3β-methyl-5α-pregnan-20-one, commonly known as ganaxolone, by overcoming historical barriers in stereoselective methylation. This technology enables the direct conversion of 5α-pregnane-3,20-dione into the target API intermediate using specialized organometallic reagents, bypassing the need for cumbersome protection-deprotection sequences. For R&D directors and procurement specialists, this represents a significant leap forward in process efficiency, offering a route that minimizes unit operations while maximizing chemical yield and purity. The ability to produce high-purity ganaxolone through a single chemical step addresses the critical need for cost-effective manufacturing processes required for therapies demanding high daily dosages.
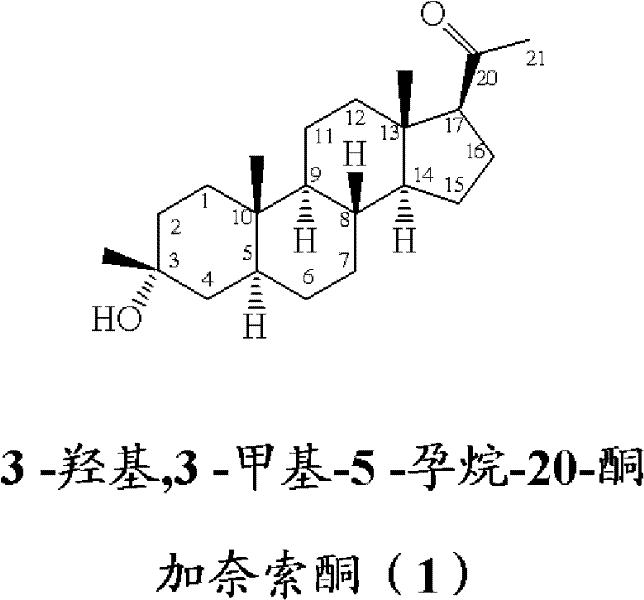
The structural complexity of neuroactive steroids necessitates precise control over stereochemistry at the C3 and C20 positions. As illustrated in the molecular architecture, the 3β-methyl configuration is critical for biological activity, yet achieving this selectively in the presence of a competing C20 ketone has long been a synthetic bottleneck. Traditional methods often struggled to differentiate between these two carbonyl environments, leading to heterogeneous product streams that required extensive downstream processing. The innovation detailed in this patent leverages the unique electronic and steric properties of iron-based organometallic complexes to discriminate between the C3 and C20 sites effectively. This specificity not only streamlines the synthesis but also ensures that the final product meets the rigorous impurity specifications demanded by global regulatory bodies for clinical and commercial use.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing ganaxolone were plagued by inherent inefficiencies that drove up manufacturing costs and extended lead times significantly. The standard approach typically involved the protection of the C20 carbonyl group, often as a ketal, prior to methylation at the C3 position, followed by a subsequent deprotection step to reveal the ketone. This multi-step sequence inherently reduces overall yield due to material losses at each isolation stage and increases the consumption of solvents and reagents. Furthermore, alternative routes utilizing Corey reagents for epoxidation followed by reduction were found to be equally problematic, often generating difficult-to-remove byproducts such as 17-hydroxy-ganaxolone. These legacy processes resulted in complex product mixtures where undesired isomers possessed physical properties similar to the target molecule, making purification via crystallization or chromatography both technically challenging and economically burdensome for large-scale operations.
The Novel Approach
In stark contrast to these convoluted pathways, the novel method described in the patent achieves the transformation in a single chemical step with remarkable precision. By reacting 5α-pregnane-3,20-dione directly with an organometallic methylating agent generated in situ from ferric chloride and a Grignard reagent, the process eliminates the need for any protecting groups. This direct methylation strategy capitalizes on the formation of specific iron-ate complexes, such as trimethylferrate species, which exhibit superior regioselectivity for the C3 carbonyl over the C20 carbonyl. The result is a dramatic simplification of the workflow, where the starting material is converted to the desired 3β-methyl isomer with high fidelity. This approach not only reduces the number of reactor vessels required but also minimizes the generation of hazardous waste associated with protection and deprotection chemistries, aligning perfectly with modern green chemistry principles and cost-reduction mandates.
Mechanistic Insights into FeCl3-Catalyzed Regioselective Methylation
The success of this synthesis hinges on the nuanced behavior of the organometallic reagent system, specifically the interaction between iron halides and methylating agents like methylmagnesium chloride. When anhydrous ferric chloride is treated with excess Grignard reagent at low temperatures, typically ranging from -40°C to -20°C, it generates highly reactive nucleophilic species often described as ate complexes. These complexes, potentially existing as equilibria of Me3FeLi or Me3FeMgCl species, possess a steric bulk and electronic character that favors attack on the less hindered or more electronically accessible C3 ketone. The low temperature is critical not only for the stability of these transient iron species but also for suppressing the thermodynamic drive towards the C20 adduct, which is a common side reaction in uncatalyzed systems. This kinetic control ensures that the methylation occurs almost exclusively at the desired position, preventing the formation of the regioisomeric impurities that plagued earlier attempts at direct functionalization.
Furthermore, the stereoselectivity of the reaction is governed by the trajectory of the nucleophilic attack on the steroid backbone. The bulky iron-methyl complex approaches the C3 carbonyl from the equatorial direction, driven by the conformational constraints of the A-ring in the 5α-pregnane system. This equatorial attack leads predominantly to the formation of the axial hydroxyl group and the equatorial methyl group, corresponding to the desired 3α-hydroxy, 3β-methyl configuration. The patent data indicates that by fine-tuning the stoichiometry of the iron salt and the Grignard reagent, along with the addition of lithium salts to modify the aggregation state of the reagent, the formation of the unwanted 3α-methyl epimer can be suppressed to negligible levels. This level of control over the stereochemical outcome is paramount for pharmaceutical applications, where even minor deviations in isomeric purity can impact the safety and efficacy profile of the final drug product.
How to Synthesize Ganaxolone Efficiently
Implementing this synthesis requires careful attention to reagent preparation and thermal management to ensure consistent quality. The process begins with the generation of the active methylating species in an inert solvent like tetrahydrofuran, followed by the controlled addition of the diketone substrate. Maintaining the reaction temperature within the specified cryogenic range is essential to preserve the selectivity of the iron complex. Following the reaction completion, a straightforward acidic workup quenches the organometallic species and liberates the product. The crude material can then be purified through a simple recrystallization or slurring process, often using ethyl acetate, to remove trace impurities and residual starting materials. For detailed operational parameters and specific stoichiometric ratios validated at scale, please refer to the standardized protocol below.
- Preparation of the organometallic methylating reagent by reacting anhydrous ferric chloride with methylmagnesium chloride in THF at low temperatures (-35°C to -40°C) to form active ate complexes.
- Regioselective addition of 5α-pregnane-3,20-dione to the pre-formed iron-methyl complex, maintaining strict temperature control between -25°C and -20°C to ensure C3 selectivity.
- Quenching the reaction with acid, followed by a simplified purification process involving hot ethyl acetate slurring to achieve >99% HPLC purity without chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. By collapsing a multi-step synthesis into a single transformation, the process drastically reduces the consumption of raw materials, solvents, and energy, leading to significant cost savings in manufacturing. The elimination of protection and deprotection steps removes the need for specialized reagents and the associated waste disposal costs, simplifying the environmental compliance burden. Moreover, the use of commodity chemicals like ferric chloride and methylmagnesium chloride ensures a stable and reliable supply of inputs, mitigating the risk of shortages associated with exotic or custom-synthesized catalysts. This robustness allows for more predictable production scheduling and shorter lead times, enabling suppliers to respond more agilely to market demands for this critical CNS therapeutic intermediate.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the reduction in unit operations, which translates directly to lower operational expenditures. By avoiding the isolation of intermediates and the use of protecting groups, the process minimizes solvent usage and waste generation, resulting in a leaner manufacturing footprint. The high yield and selectivity reduce the need for expensive chromatographic purification, allowing for cost-effective isolation via crystallization. This efficiency makes the production of ganaxolone viable for high-volume applications where cost-per-gram is a decisive factor in project feasibility.
- Enhanced Supply Chain Reliability: The reliance on widely available, off-the-shelf reagents enhances the security of the supply chain against disruptions. Unlike processes dependent on proprietary catalysts or scarce chiral auxiliaries, this method utilizes iron salts and Grignard reagents that are produced globally in massive quantities. This accessibility ensures that production can be scaled rapidly without waiting for long-lead-time materials. Additionally, the simplified workflow reduces the number of potential failure points in the manufacturing line, increasing the overall reliability of delivery schedules for downstream API producers.
- Scalability and Environmental Compliance: The reaction conditions are amenable to large-scale batch processing, with thermal profiles that can be managed using standard industrial cooling systems. The absence of heavy metal catalysts or toxic protecting group byproducts simplifies the effluent treatment process, facilitating compliance with increasingly stringent environmental regulations. The ability to purify the product through simple slurring techniques rather than complex separations further supports scalability, as it reduces the equipment footprint and processing time required to achieve pharmaceutical-grade purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding the nuances of impurity control and reaction kinetics is essential for successful technology transfer. The answers provided are derived directly from the experimental data and mechanistic insights contained within the patent documentation, ensuring accuracy and relevance for technical decision-makers evaluating this process for adoption.
Q: Why was direct methylation of 5α-pregnane-3,20-dione previously considered unfeasible?
A: Historically, direct methylation resulted in complex mixtures due to non-selective attack on both C3 and C20 carbonyls, producing difficult-to-separate epimers and regioisomers that compromised pharmaceutical purity standards.
Q: How does the iron-catalyzed method improve impurity profiles compared to traditional routes?
A: By utilizing specific iron-ate complexes, the process achieves high stereoselectivity for the 3β-methyl isomer and regioselectivity for the C3 position, significantly reducing the formation of C20-adducts and 3α-epimers.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method utilizes readily available reagents like FeCl3 and MeMgCl in common ether solvents, avoiding expensive protecting group chemistry, which facilitates straightforward scale-up from kilogram to multi-ton batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ganaxolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for high-value pharmaceutical intermediates like ganaxolone. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which are equipped to detect and quantify trace impurities at ppm levels. Our capability to handle complex organometallic chemistry under cryogenic conditions allows us to leverage patents like CN102549007A to offer clients a competitive edge in terms of both cost and quality.
We invite you to collaborate with us to optimize your supply chain for neuroactive steroids. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your development timelines and commercial goals efficiently.
