Advanced Base-Free Acylation for Commercial Scale-Up of Complex Agrochemical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance high purity with economic efficiency, and patent CN102015649A presents a compelling solution for the manufacturing of aryl carboxamides. This intellectual property details a novel method for preparing compounds of formula (I), which serve as critical intermediates for high-value crop protection agents such as Boscalid and Bixafen. The core innovation lies in a base-free acylation strategy that operates under reduced pressure, effectively overcoming the limitations of traditional high-dilution methods. By utilizing specific reaction conditions involving aromatic solvents and precise metering of reactants, this process achieves superior yields while minimizing the formation of problematic byproducts. As a reliable agrochemical intermediate supplier, understanding the nuances of such patented methodologies is essential for delivering consistent quality to global markets. The structural versatility of formula (I) allows for a wide range of substituents on the phenyl, pyridyl, or pyrazolyl rings, making this technology highly adaptable for diverse chemical portfolios.
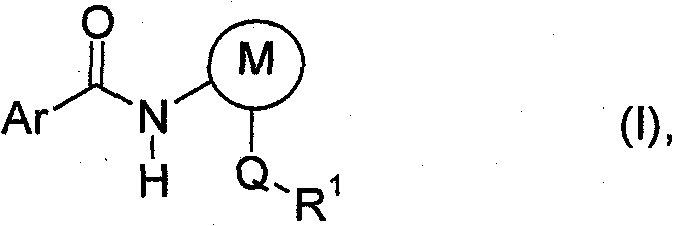
The significance of this patent extends beyond mere academic interest; it addresses real-world manufacturing bottlenecks associated with scaling up amide bond formations. Traditional approaches often struggle with heat management and byproduct accumulation, but the disclosed method utilizes a controlled pressure environment to drive the reaction to completion. This ensures that the resulting high-purity aryl carboxamides meet the stringent specifications required for downstream formulation into final active ingredients. For procurement managers and supply chain heads, the adoption of such efficient synthetic routes translates directly into enhanced supply security and potential cost optimization. The ability to produce these complex molecules without the need for expensive auxiliary bases or extensive purification steps represents a substantial advancement in process chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques for synthesizing aryl carboxamides, such as those described in JP-A 2001/172276, typically rely on high-dilution conditions to manage the exothermic nature of the reaction and prevent side reactions. However, these methods are fundamentally flawed when applied to technical scale manufacturing due to the excessive volumes of solvents required, which drastically increases operational costs and environmental waste. Furthermore, attempting to run these reactions at higher concentrations often leads to severe mixing problems and the formation of agglomerates, which significantly reduces the productive rate of the valuable product. The reliance on auxiliary bases in many conventional protocols introduces additional complexity, as the resulting salt byproducts must be separated and disposed of, adding further steps to the isolation process. These inefficiencies create a bottleneck for manufacturers aiming to reduce lead time for high-purity agrochemical intermediates, as the downstream processing becomes cumbersome and time-consuming. Consequently, there is a critical need for a more concentrated and efficient operating method that maintains high yields without compromising on safety or purity.
The Novel Approach
The method disclosed in CN102015649A revolutionizes this landscape by enabling the reaction to proceed in the absence of an auxiliary base under carefully controlled reduced pressure. By establishing a pressure of 0 to 700 mbar, the process facilitates the continuous removal of hydrogen chloride gas, which drives the equilibrium towards the desired product and prevents the accumulation of acidic byproducts that could degrade the reactants. The strategic addition sequence, where the acid chloride is added first followed by the metered introduction of the aryl amine, ensures that the reaction mixture remains homogeneous and manageable even at higher concentrations. This approach effectively eliminates the agglomeration issues seen in prior art, allowing for a much more spissated operating method that is suitable for large-scale production. The use of aromatic hydrocarbons like toluene or xylene as solvents further enhances the solubility of the reactants and products, facilitating easier isolation through crystallization. This novel approach not only improves the overall yield but also streamlines the workflow, making it an ideal candidate for cost reduction in agrochemical manufacturing.
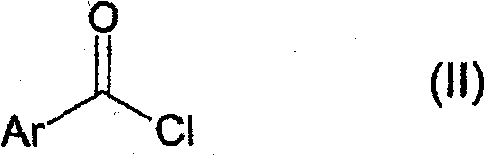
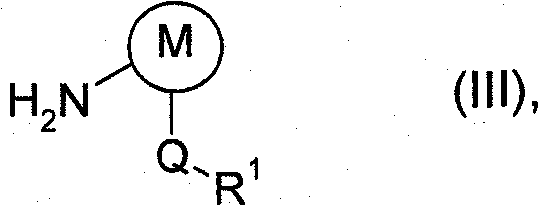
Mechanistic Insights into Base-Free Acylation under Reduced Pressure
The mechanistic elegance of this process lies in its ability to leverage thermodynamic principles to overcome kinetic barriers without the aid of stoichiometric bases. In a typical acylation reaction between an acid chloride and an amine, hydrogen chloride is generated as a byproduct, which can protonate the unreacted amine and render it nucleophilically inactive. By operating under reduced pressure (preferably 200-600 mbar), the system continuously strips away the HCl gas as it forms, preventing the protonation of the amine and maintaining a high concentration of the active nucleophile throughout the reaction. This dynamic removal of byproducts shifts the reaction equilibrium strongly towards the formation of the amide bond, resulting in high conversion rates even with near-stoichiometric ratios of reactants. The temperature range of 20-120°C, preferably 70-100°C, provides sufficient thermal energy to overcome the activation barrier while remaining mild enough to prevent thermal decomposition of sensitive functional groups such as halogens or trifluoromethyl groups. This careful balance of pressure and temperature is crucial for maintaining the integrity of the complex molecular architectures found in modern agrochemicals.
Impurity control is another critical aspect where this mechanism excels, particularly in the context of avoiding hydrolysis of the acid chloride. The patent specifies the use of substantially anhydrous organic solvents with water content strictly controlled to about 0.5-5g water/mol acyl chlorides. Excess water would lead to the hydrolysis of the acid chloride back to the carboxylic acid, consuming the valuable starting material and generating difficult-to-remove impurities. The base-free nature of the reaction also means there are no inorganic salts formed, which simplifies the purification profile significantly. The product can often be isolated directly by crystallization from the reaction mixture upon cooling, or after a simple wash with aqueous alkali if necessary. This minimizes the number of unit operations required, reducing the potential for mechanical losses and contamination. For R&D directors focused on purity and impurity profiles, this mechanism offers a clean route that minimizes the generation of closely related impurities, ensuring the final API intermediate meets rigorous regulatory standards.
How to Synthesize Aryl Carboxamides Efficiently
The synthesis of these valuable intermediates requires precise adherence to the patented protocol to ensure optimal yield and purity. The process begins with the dissolution of the acid chloride in a dry aromatic solvent, followed by the establishment of the vacuum conditions. The aryl amine is then metered in slowly over a period of 0.5 to 20 hours, allowing the reaction to proceed in a controlled manner. Detailed standardized synthesis steps see the guide below.
- Dissolve the acid chloride of formula (II) in a suitable non-aqueous aromatic solvent such as toluene or xylene.
- Establish a reduced pressure environment between 0 to 700 mbar and heat the solution to a reaction temperature of 20-120°C.
- Meter in the aryl amine of formula (III) in approximately stoichiometric amounts over 0.5 to 20 hours, then isolate the valuable product via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-free acylation technology offers transformative benefits that extend well beyond the laboratory bench. The elimination of auxiliary bases represents a direct reduction in raw material costs, as there is no longer a need to purchase, store, and handle large quantities of inorganic or organic bases. Furthermore, the absence of salt byproducts significantly reduces the volume of waste streams generated during the workup phase, leading to lower disposal costs and a smaller environmental footprint. This streamlined process also enhances the overall throughput of the manufacturing facility, as the simplified isolation steps allow for faster batch turnover times. By reducing the complexity of the synthesis, manufacturers can achieve greater consistency in production, which is vital for maintaining long-term supply contracts with major agrochemical companies. The robustness of this method ensures that supply continuity is maintained even during periods of high demand, providing a competitive edge in the global market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of auxiliary bases and the associated neutralization steps. In traditional methods, the cost of bases and the subsequent disposal of salt waste can account for a significant portion of the total manufacturing expense. By eliminating these components, the process achieves substantial cost savings through reduced material consumption and waste treatment fees. Additionally, the ability to operate at higher concentrations reduces the volume of solvent required per kilogram of product, lowering both solvent purchase costs and energy costs associated with solvent recovery and distillation. The high yields reported in the examples, often exceeding 90%, further contribute to cost efficiency by maximizing the utilization of expensive starting materials. These cumulative effects result in a significantly reduced cost of goods sold, making the final intermediates more price-competitive.
- Enhanced Supply Chain Reliability: Supply chain reliability is heavily dependent on the robustness and scalability of the manufacturing process. The method described in the patent avoids the use of sensitive reagents or extreme conditions that could lead to batch failures or delays. The reactants, acid chlorides and aryl amines, are generally commercially available or can be synthesized via established routes, ensuring a stable supply of starting materials. The simplified workup procedure, which often involves direct crystallization, reduces the dependency on complex purification equipment and specialized labor. This simplicity translates to a more resilient supply chain that is less susceptible to disruptions caused by equipment maintenance or operator error. For buyers, this means a more predictable delivery schedule and a lower risk of supply shortages, which is critical for just-in-time manufacturing models in the agrochemical industry.
- Scalability and Environmental Compliance: Scaling up chemical processes often introduces new challenges related to heat transfer and mixing, but this method is specifically designed to address these issues. The controlled metering of the amine and the use of reduced pressure ensure that the exotherm is managed effectively, preventing runaway reactions even in large reactors. The reduction in solvent usage and waste generation aligns perfectly with increasingly stringent environmental regulations, facilitating easier permitting and compliance. The process generates minimal hazardous waste, primarily consisting of spent solvent which can be recycled, and avoids the generation of saline wastewater. This environmental compatibility not only reduces regulatory burden but also enhances the corporate social responsibility profile of the manufacturer. As the industry moves towards greener chemistry, this scalable and eco-friendly process positions the supplier as a leader in sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the operational parameters and benefits for potential partners and stakeholders.
Q: Why is the absence of an auxiliary base significant in this acylation process?
A: Eliminating auxiliary bases removes the formation of salt byproducts (such as amine hydrochlorides), which simplifies the workup procedure, reduces waste generation, and significantly lowers the cost of raw materials and disposal.
Q: How does the reduced pressure condition improve the reaction yield?
A: Operating under reduced pressure (0-700 mbar) facilitates the continuous removal of hydrogen chloride gas generated during the reaction, driving the equilibrium forward and preventing the degradation of sensitive reactants.
Q: What specific agrochemical active ingredients can be synthesized using this method?
A: This versatile platform technology is applicable to the synthesis of key fungicides and insecticides including Boscalid, Bixafen, Benodanil, and various fluorinated biphenyl pyrazole carboxamides used in modern crop protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Carboxamides Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN102015649A for the production of high-value agrochemical intermediates. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of this base-free acylation process, guaranteeing consistent quality and yield. By partnering with us, you gain access to a supply chain that is both robust and responsive to your specific needs.
We invite you to discuss how our capabilities can support your project goals and help you achieve your cost and quality targets. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can add value to your supply chain. Let us be your trusted partner in bringing these advanced agrochemical solutions to the global market.
