Advanced Base-Free Vacuum Process for High-Purity Aryl Carboxamide Manufacturing and Scale-Up
Advanced Base-Free Vacuum Process for High-Purity Aryl Carboxamide Manufacturing and Scale-Up
The chemical industry constantly seeks more efficient pathways to produce complex molecules, and patent CN102015649B introduces a transformative method for preparing aryl carboxamides of formula (I). This innovation addresses critical bottlenecks in traditional acylation reactions by eliminating the need for auxiliary bases and utilizing a controlled vacuum environment. The process enables the reaction of an acid chloride of formula (II) with an arylamine of formula (III) in a suitable non-aqueous solvent, achieving high yields without the formation of problematic salt by-products. This technical breakthrough is particularly relevant for the production of high-value agrochemical active ingredients and intermediates, where purity and process robustness are paramount. By shifting the reaction equilibrium through pressure control rather than chemical scavengers, manufacturers can achieve a cleaner reaction profile that simplifies isolation and reduces environmental impact. The versatility of this method allows for the synthesis of a wide range of substituted aryl carboxamides, including those with sensitive functional groups that might degrade under harsher conventional conditions. 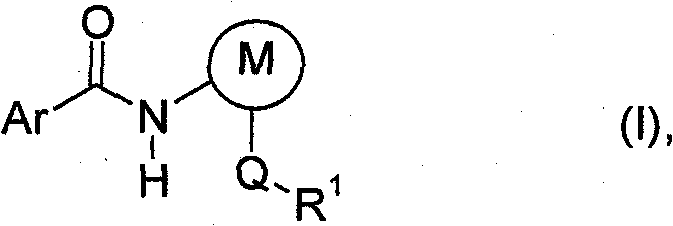
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing carboxamides from acid chlorides and amines often rely heavily on the use of auxiliary bases to neutralize the hydrogen chloride by-product generated during the reaction. While chemically effective on a small scale, these methods present significant challenges when translated to industrial manufacturing environments. The formation of amine hydrochloride salts necessitates additional downstream processing steps, such as aqueous workups or filtration, which increase solvent consumption and waste generation. Furthermore, literature references such as JP-A 2001/172276 indicate that attempting to run these reactions without bases in highly concentrated solutions often leads to severe mixing problems and the formation of lumps. These physical issues drastically reduce the yield of valuable products and can cause blockages in large-scale reactors, posing safety risks and operational delays. The reliance on large volumes of solvent to mitigate these mixing issues further exacerbates the cost and environmental footprint, making conventional base-mediated or highly dilute processes economically unviable for modern cost-sensitive supply chains.
The Novel Approach
The novel approach described in the patent data overcomes these historical limitations by employing a sophisticated vacuum-assisted technique that operates effectively without auxiliary bases. By initially charging the acid chloride and establishing a specific pressure range of 0 to 700 mbar, the process controls the boiling point of the solvent to maintain optimal reaction kinetics. The arylamine is then metered in approximately stoichiometric amounts, preventing local excesses that could lead to side reactions or precipitation issues. This method allows for much higher concentrations compared to prior art, significantly reducing the volume of solvent required per kilogram of product. The absence of base means no inorganic salts are formed, resulting in a reaction mixture that is far easier to process and purify. This streamlined workflow not only enhances the overall yield, with examples showing up to 96% efficiency, but also improves the safety profile by removing the exothermic risks associated with base neutralization. The result is a robust, scalable protocol that aligns perfectly with the needs of a reliable agrochemical intermediate supplier seeking to optimize production costs.
Mechanistic Insights into Base-Free Vacuum Acylation
The core mechanism of this process relies on the precise manipulation of physical parameters to drive the chemical equilibrium towards the desired amide bond formation. In the absence of a base to scavenge protons, the reaction generates hydrogen chloride, which must be managed to prevent the protonation of the starting amine, a phenomenon that would deactivate the nucleophile and halt the reaction. The application of reduced pressure facilitates the continuous removal of volatile components and helps maintain the reaction mixture in a state of gentle boiling, which enhances mass transfer between the acid chloride and the arylamine. 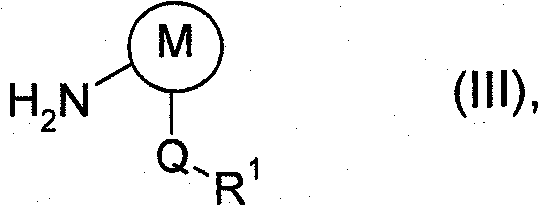
Impurity control is inherently built into this mechanism through the strict control of stoichiometry and addition rates. By metering the arylamine over a period of 0.5 to 20 hours, the concentration of the free amine is kept low relative to the acid chloride, minimizing the risk of di-acylation or other side reactions that often plague batch processes. The use of aromatic hydrocarbons like toluene as solvents provides a stable medium that supports the solubility of both reactants and the product, preventing premature crystallization that could lead to the lump formation observed in prior art. The process temperature, typically maintained between 20°C and 120°C, is carefully selected to ensure sufficient energy for the reaction while avoiding thermal degradation of sensitive substituents such as halogens or fluoroalkyl groups. This delicate balance ensures that the final product meets stringent purity specifications required for pharmaceutical and agrochemical applications, reducing the need for extensive recrystallization steps.
How to Synthesize Aryl Carboxamides Efficiently
Implementing this synthesis route requires careful attention to the sequence of operations and the maintenance of the vacuum environment throughout the reaction cycle. The process begins with the dissolution of the acid chloride in a dry, non-aqueous solvent, followed by the establishment of the target vacuum pressure before any heating is applied. Once the system is stabilized, the arylamine solution is introduced gradually, allowing the reaction to proceed smoothly without sudden exotherms or precipitation events. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding temperature ramps and cooling profiles. Adhering to these protocols ensures that the benefits of the base-free mechanism are fully realized, delivering consistent quality and high throughput for commercial production batches.
- Charge the acid chloride into a suitable non-aqueous solvent such as toluene within the reaction vessel.
- Establish a reduced pressure environment ranging from 0 to 700 mbar to facilitate solvent boiling and reaction control.
- Meter in the arylamine in approximately stoichiometric amounts over a period of 0.5 to 20 hours while maintaining temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages that go beyond simple chemical efficiency. The elimination of auxiliary bases and the reduction in solvent usage directly address the growing pressure to reduce manufacturing costs and environmental liabilities in the fine chemical sector. By simplifying the workup procedure, manufacturers can significantly reduce the time required to turn around batches, thereby enhancing supply chain reliability and responsiveness to market demand fluctuations. The robustness of the process against mixing issues means that scale-up from pilot to commercial production is less risky, ensuring a continuous supply of critical intermediates without the interruptions often caused by reactor fouling or yield failures. This stability is crucial for maintaining long-term contracts with downstream formulators who depend on consistent quality and delivery schedules for their own production lines.
- Cost Reduction in Manufacturing: The removal of auxiliary bases eliminates the cost of purchasing these reagents and the subsequent costs associated with disposing of the salt waste generated. Furthermore, the ability to run the reaction at higher concentrations reduces the volume of solvent needed, which lowers both raw material costs and the energy required for solvent recovery and distillation. This qualitative improvement in process efficiency leads to substantial cost savings over the lifecycle of the product, making the supply chain more resilient against raw material price volatility. The simplified isolation process also reduces labor and equipment time, contributing to a lower overall cost of goods sold without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: The avoidance of lump formation and mixing problems ensures that reactors can operate continuously without the need for frequent cleaning or maintenance interventions. This operational stability minimizes unplanned downtime and ensures that production schedules are met consistently, which is vital for just-in-time manufacturing environments. The use of commercially available starting materials and common solvents like toluene further reduces supply risk, as these commodities are widely sourced and less prone to shortages compared to specialized reagents. This reliability makes the manufacturer a more attractive partner for long-term supply agreements, providing peace of mind to procurement teams managing complex global supply networks.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing standard unit operations such as vacuum distillation and metered addition that are easily replicated in large-scale facilities. The reduction in waste generation aligns with increasingly strict environmental regulations, reducing the burden on waste treatment facilities and lowering compliance costs. The ability to handle sensitive functional groups without degradation means that a wider range of valuable intermediates can be produced using this single platform technology, maximizing asset utilization. This environmental and operational efficiency positions the manufacturing site as a sustainable partner in the global supply chain, meeting the ESG goals of modern multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-free vacuum process for aryl carboxamide synthesis. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The insights provided here clarify the operational boundaries and benefits, facilitating informed decision-making for R&D and procurement teams alike.
Q: Why is the base-free condition critical for this synthesis?
A: Eliminating auxiliary bases prevents the formation of salt by-products, which simplifies downstream purification and significantly reduces waste treatment costs in industrial manufacturing.
Q: How does the vacuum pressure affect the reaction yield?
A: Operating under reduced pressure (0-700 mbar) allows the reaction mixture to boil at lower temperatures, preventing thermal degradation and ensuring high conversion rates without lump formation.
Q: What solvents are compatible with this process?
A: Aromatic hydrocarbons like toluene and xylenes are particularly preferred due to their ability to dissolve reactants effectively while maintaining stability under the specified vacuum and temperature conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Carboxamide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced processes like the one described in CN102015649B to deliver superior value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can translate laboratory innovations into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aryl carboxamide meets the exacting standards required for agrochemical and pharmaceutical applications. Our commitment to technical excellence allows us to offer customized solutions that optimize both performance and cost for our clients.
We invite you to discuss how our capabilities can support your supply chain optimization goals. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific product requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing protocols can enhance your project's success. Partner with us to secure a reliable supply of high-quality intermediates that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
