Advanced Palladium-Catalyzed Asymmetric Hydrogenation for Scalable Chiral Indoline Production
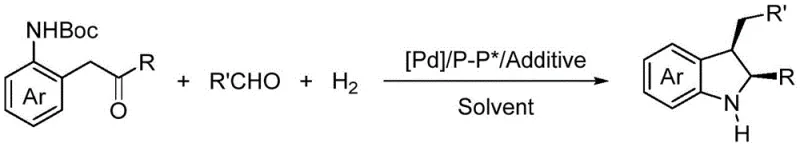
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access chiral scaffolds that serve as the backbone for bioactive molecules. Patent CN112028727A introduces a transformative methodology for the synthesis of chiral indolines through palladium-catalyzed asymmetric hydrogenation of 2,3-disubstituted indoles generated in situ. This technology addresses critical bottlenecks in the production of high-purity pharmaceutical intermediates by combining indole formation and hydrogenation into a seamless tandem process. The strategic advantage lies in the avoidance of isolating unstable indole intermediates, which traditionally complicates supply chains and reduces overall yield. By leveraging a homogeneous palladium system with chiral bisphosphine ligands, this method achieves exceptional enantioselectivity, with enantiomeric excess values reaching up to 97%. For R&D Directors and Procurement Managers, this represents a significant opportunity to streamline the manufacturing of complex heterocyclic compounds. The robustness of the catalytic system ensures that the production of these valuable building blocks can be scaled reliably, meeting the stringent purity specifications required for modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for chiral indolines often rely on the pre-formation and isolation of indole derivatives before subjecting them to hydrogenation conditions. This multi-step approach introduces significant inefficiencies, including the need for additional purification stages and the handling of potentially unstable intermediates that may degrade during storage or transfer. Furthermore, conventional catalytic systems frequently require harsh reaction conditions, such as elevated temperatures or excessive hydrogen pressures, which can compromise safety and increase energy consumption. The reliance on stoichiometric chiral auxiliaries in older methods also generates substantial chemical waste, conflicting with modern green chemistry principles and environmental compliance standards. These limitations collectively drive up the cost of goods sold and extend lead times, creating friction in the supply chain for high-purity pharmaceutical intermediates. Additionally, the variability in stereochemical outcomes with older catalysts often necessitates costly recrystallization steps to achieve the required optical purity, further eroding profit margins and operational efficiency.
The Novel Approach
The methodology disclosed in CN112028727A revolutionizes this landscape by employing a one-pot tandem reaction strategy that generates the indole intermediate in situ followed immediately by asymmetric hydrogenation. This approach drastically simplifies the operational workflow, eliminating the isolation step and thereby enhancing atom economy and step efficiency. The use of a palladium catalyst coordinated with specialized chiral bisphosphine ligands allows the reaction to proceed under mild conditions, typically between 30°C and 60°C, with moderate hydrogen pressures. This gentler operational window not only improves safety profiles but also preserves the integrity of sensitive functional groups on the substrate. The process demonstrates broad substrate tolerance, accommodating various electronic and steric environments on the aromatic rings, which is crucial for the diverse needs of medicinal chemistry programs. By integrating formation and reduction into a single vessel, the novel approach minimizes solvent usage and waste generation, aligning perfectly with sustainability goals while delivering superior yields and stereocontrol.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the precise orchestration of the catalytic cycle involving the palladium-chiral ligand complex. The reaction initiates with the acid-catalyzed condensation of an aminoketone and an aldehyde to form the 2,3-disubstituted indole intermediate within the reaction mixture. This transient species is immediately captured by the active palladium hydride species generated under hydrogen atmosphere. The chiral environment provided by ligands such as (R)-SegPhos or (R)-H8-BINAP dictates the facial selectivity of the hydrogen addition to the C=C double bond of the indole ring. The steric bulk and electronic properties of these ligands create a highly defined chiral pocket that favors the formation of one enantiomer over the other, resulting in the observed high enantiomeric excess. The presence of an acid additive, such as ethanesulfonic acid, plays a critical role in activating the substrate and facilitating the protonation steps necessary for the catalytic turnover. This mechanistic elegance ensures that the reaction proceeds with high fidelity, minimizing the formation of unwanted byproducts or racemic material.
Controlling the impurity profile is paramount for any process intended for pharmaceutical applications, and this mechanism offers inherent advantages in that regard. The high stereoselectivity of the catalyst system means that the primary impurity concern shifts from difficult-to-separate enantiomers to simpler diastereomers or unreacted starting materials, which are generally easier to remove via standard chromatographic or crystallization techniques. The mild reaction conditions also prevent thermal degradation pathways that often lead to complex, polymeric impurities in harsher processes. Furthermore, the use of commercially available and well-defined catalyst precursors ensures batch-to-batch consistency, a critical factor for regulatory compliance. The ability to tune the ligand structure allows chemists to optimize the process for specific substrates, ensuring that even challenging molecules can be produced with the stringent purity specifications required for clinical trials and commercial launch. This level of control over the chemical outcome translates directly into reduced risk and faster time-to-market for new drug candidates.
How to Synthesize Chiral Indolines Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction parameters to maximize efficiency and yield. The process begins with the pre-formation of the active catalyst species by mixing a palladium metal precursor, such as palladium trifluoroacetate, with the chosen chiral bisphosphine ligand in a solvent like acetone. This mixture is stirred at room temperature to ensure complete complexation before the solvent is removed under vacuum. The resulting catalyst solid is then transferred to the reaction vessel containing the substrate mixture of aminoketone and aldehyde, along with the necessary acid additive and reaction solvent. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the palladium catalyst by mixing a palladium metal precursor with a chiral bisphosphine ligand in acetone, stirring at room temperature, and removing the solvent under vacuum.
- Combine the catalyst with the substrate mixture containing an aminoketone and an aldehyde in a suitable solvent like trifluoroethanol or dichloromethane, adding an acid additive.
- Conduct the hydrogenation reaction under hydrogen pressure (100-1000 psi) at controlled temperatures (30-60°C), followed by workup and purification to isolate the chiral indoline.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly impact the bottom line and supply chain resilience. The elimination of intermediate isolation steps translates to significant cost reduction in pharmaceutical intermediates manufacturing by reducing labor, solvent, and time requirements. The use of commercially available catalysts and mild reaction conditions lowers the barrier to entry for production, allowing for flexible manufacturing strategies that can adapt to fluctuating market demands. For Supply Chain Heads, the robustness of the process ensures reliable delivery schedules, as the risk of batch failure due to unstable intermediates is virtually eliminated. The high atom economy and reduced waste generation also simplify environmental compliance, reducing the costs associated with waste disposal and regulatory reporting. These factors combine to create a more agile and cost-effective supply chain capable of supporting the rapid development cycles of the modern pharmaceutical industry.
- Cost Reduction in Manufacturing: The streamlined one-pot process significantly lowers operational expenses by removing the need for intermediate workup and purification stages. This reduction in unit operations decreases solvent consumption and energy usage, leading to substantial cost savings over the lifecycle of the product. Additionally, the high yields and selectivity minimize the loss of valuable starting materials, further optimizing the cost structure. The ability to use standard equipment without specialized high-pressure or high-temperature requirements also reduces capital expenditure and maintenance costs. These efficiencies allow for more competitive pricing strategies while maintaining healthy profit margins in a cost-sensitive market.
- Enhanced Supply Chain Reliability: The stability of the in situ generation method ensures consistent production output, mitigating the risks associated with supply disruptions. By avoiding the storage and transport of unstable indole intermediates, the process reduces logistical complexities and potential quality degradation during transit. The use of readily available raw materials and catalysts means that sourcing is straightforward and less susceptible to geopolitical or market volatility. This reliability is crucial for maintaining continuous manufacturing operations and meeting the just-in-time delivery expectations of global pharmaceutical clients. The predictable nature of the reaction kinetics also allows for accurate production planning and inventory management.
- Scalability and Environmental Compliance: The mild reaction conditions and simple workup procedures make this process highly amenable to commercial scale-up of complex pharmaceutical intermediates. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, minimizing the ecological footprint of the manufacturing process. The ability to scale from gram to ton quantities without significant re-optimization provides a clear path from R&D to commercial production. This scalability ensures that the technology can support both small-batch clinical supply and large-scale commercial manufacturing, offering flexibility as the drug candidate progresses through development stages. The green chemistry attributes of the process also enhance the corporate sustainability profile, which is increasingly important to stakeholders and customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These answers are derived directly from the patent data and practical experience with similar catalytic systems to provide clarity on feasibility and performance. Understanding these details is essential for making informed decisions about adopting this technology for your specific production needs. The information covers catalyst selection, reaction conditions, and scalability considerations to ensure a comprehensive understanding of the process capabilities.
Q: What are the key advantages of the in situ generation method for indoles?
A: The in situ generation method eliminates the need to isolate unstable indole intermediates, significantly improving atom economy and step efficiency while reducing overall processing time and waste generation.
Q: Which chiral ligands provide the highest enantioselectivity in this process?
A: Chiral bisphosphine ligands such as (R)-SegPhos, (R)-MeO-Biphep, and (R)-H8-BINAP have demonstrated superior performance, achieving enantiomeric excess values up to 97% under optimized conditions.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available catalysts and mild reaction conditions, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the manufacturing plant. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. Our facility is equipped to handle the specific requirements of palladium-catalyzed reactions, including safe hydrogenation protocols and efficient catalyst recovery systems. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and regulatory compliance, allowing you to focus on your core drug discovery activities with confidence.
We invite you to discuss how this innovative synthesis technology can optimize your current manufacturing processes and reduce overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. Let us help you navigate the complexities of chiral synthesis and secure a reliable supply of high-quality intermediates for your pipeline. Together, we can accelerate the development of life-saving medicines through superior chemical manufacturing excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
