Revolutionizing Oxime Ether Production: A Green Heteropolyacid Catalytic Approach for Commercial Scale
Revolutionizing Oxime Ether Production: A Green Heteropolyacid Catalytic Approach for Commercial Scale
The landscape of fine chemical synthesis is undergoing a transformative shift towards sustainability and efficiency, driven by stringent regulatory requirements and the economic necessity for greener processes. Patent CN114394913B, published in early 2023, presents a groundbreaking methodology for the synthesis of oxime ether derivatives, a critical structural motif found in numerous pharmaceutical agents and agrochemical intermediates. This innovation departs radically from traditional reliance on toxic halides and expensive transition metal catalysts, introducing a robust system utilizing heteropolyacids as green solid acid catalysts. By enabling the direct O-alkylation of oximes with readily available alcohols under mild conditions, this technology addresses long-standing challenges in atom economy and waste generation. For industry leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic depth and commercial viability of this patent is essential for securing a competitive edge in the global market.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxime ether linkage has been fraught with significant chemical and operational inefficiencies that burden both R&D budgets and supply chain logistics. Traditional synthetic routes predominantly rely on the condensation of carbonyl compounds with O-alkyl hydroxylamine hydrochlorides or the O-alkylation of oximes using activated alkylating agents such as halides, acetates, or phosphates. These conventional pathways invariably necessitate the use of corrosive chlorinated solvents and stoichiometric amounts of strong bases or oxidants, leading to the generation of substantial quantities of hazardous waste. Furthermore, many established protocols depend on transition metal catalysts, such as iridium or scandium complexes, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). The removal of these metal residues requires complex and costly purification steps, often involving specialized scavengers or repeated chromatography, which drastically reduces overall process throughput and increases the cost reduction in fine chemical manufacturing efforts.
The Novel Approach
In stark contrast, the methodology disclosed in CN114394913B leverages the unique acidic properties of heteropolyacids to facilitate a direct and clean etherification reaction between oximes and alcohols. This novel approach eliminates the need for pre-activated alkylating agents, allowing for the direct use of simple alcohols, including tertiary alcohols which are typically unreactive under standard conditions. The reaction proceeds efficiently in green solvents like dimethyl carbonate (DMC), avoiding the environmental liabilities associated with dichloromethane or 1,2-dichloroethane. Crucially, the only byproduct of this transformation is water, resulting in exceptional atom economy and simplifying the isolation of the target high-purity oxime ether derivatives. This paradigm shift not only enhances the safety profile of the manufacturing process but also aligns perfectly with modern green chemistry principles, offering a scalable solution for the commercial scale-up of complex organic intermediates.
Mechanistic Insights into Heteropolyacid-Catalyzed O-Alkylation
The efficacy of this synthetic route lies in the sophisticated interaction between the heteropolyacid catalyst and the reactant molecules within the reaction medium. Heteropolyacids, such as phosphotungstic acid, function as strong Brønsted acids that can effectively protonate the hydroxyl group of the alcohol substrate, thereby converting a poor leaving group into a reactive species capable of nucleophilic attack. Simultaneously, the catalyst stabilizes the developing positive charge on the carbon center, facilitating the formation of a carbocation intermediate, particularly when tertiary or benzylic alcohols are employed. The oxime nitrogen then acts as a nucleophile, attacking this activated electrophilic center to form the O-N bond. This mechanism bypasses the high energy barriers associated with direct substitution on unactivated alcohols, allowing the reaction to proceed smoothly at temperatures ranging from 25°C to 100°C. The presence of additives like anhydrous magnesium sulfate further drives the equilibrium forward by sequestering the water produced, ensuring high conversion rates without the need for harsh dehydrating agents.
From a quality control perspective, the metal-free nature of this catalytic system provides a distinct advantage in managing the impurity profile of the final product. Traditional transition metal catalysis often leaves trace amounts of toxic metals that are difficult to remove and can catalyze degradation pathways during storage. By utilizing organic-soluble heteropolyacids that can be easily neutralized and removed during the aqueous workup, the risk of metal contamination is virtually eliminated. This results in a cleaner crude reaction mixture, reducing the burden on downstream purification units and ensuring that the stringent purity specifications required for pharmaceutical applications are met with greater consistency. The robustness of the catalyst across a wide range of electronic environments on the aromatic rings of the substrates further underscores the versatility of this mechanism, making it suitable for diverse molecular architectures.
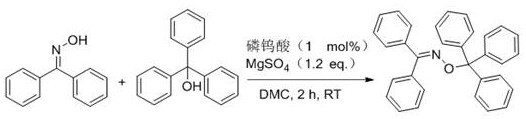
How to Synthesize Benzophenone O-Trityl Oxime Efficiently
The practical implementation of this technology is demonstrated through the efficient synthesis of benzophenone O-trityl oxime, a representative high-value intermediate. The process involves dissolving benzophenone oxime and triphenylmethanol in dimethyl carbonate, followed by the addition of a catalytic amount of phosphotungstic acid and anhydrous magnesium sulfate. The mixture is stirred at room temperature for a short duration, after which the reaction is quenched with a mild base like triethylamine. This straightforward protocol highlights the operational simplicity of the method, requiring no specialized equipment or inert atmosphere conditions. For detailed procedural specifics regarding reagent ratios, temperature profiles, and purification techniques, please refer to the standardized synthesis guide below.
- Dissolve the starting substrate alcohols and oxime compounds in a green solvent such as dimethyl carbonate (DMC).
- Add a heteropolyacid catalyst (e.g., phosphotungstic acid) and an additive like anhydrous magnesium sulfate to the reaction mixture.
- Stir the mixture at temperatures between 25°C and 100°C for 0.5 to 12 hours under air atmosphere, then neutralize and purify the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this heteropolyacid-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The shift away from precious metal catalysts and hazardous halogenated reagents fundamentally alters the cost structure of production, removing dependencies on volatile commodity markets for rare earth metals. Additionally, the use of commercially abundant alcohols as alkylating agents simplifies raw material sourcing, enhancing supply chain reliability and reducing the risk of disruptions caused by the scarcity of specialized reagents. The simplified workup procedure, characterized by simple filtration and neutralization rather than complex metal scavenging, significantly accelerates the production cycle, allowing for faster turnaround times and improved responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated purification infrastructure leads to substantial cost savings in the overall production budget. By avoiding the use of stoichiometric activators and toxic solvents, the process reduces raw material expenditure and waste disposal costs, creating a more economically sustainable manufacturing model that improves profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing widely available alcohols and stable solid acid catalysts mitigates the risks associated with sourcing specialized or regulated chemical precursors. This stability ensures a consistent supply of high-purity oxime ether derivatives, reducing lead time for high-purity active pharmaceutical ingredients and allowing manufacturers to maintain robust inventory levels even during periods of global supply chain stress.
- Scalability and Environmental Compliance: The green nature of the solvent system and the absence of heavy metals simplify the regulatory approval process for new drug filings, as residual solvent and metal limits are easier to meet. The high atom economy and minimal waste generation facilitate easier scale-up from laboratory to commercial production, ensuring that environmental compliance is maintained effortlessly as production volumes increase to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthetic methodology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the operational parameters and scope of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary advantages of using heteropolyacids over transition metals in oxime ether synthesis?
A: Heteropolyacids offer a metal-free catalytic system that eliminates the risk of heavy metal residues in the final product, simplifies post-treatment procedures, and reduces environmental pollution compared to traditional transition metal catalysts.
Q: Can this synthetic method accommodate diverse substrate structures?
A: Yes, the method demonstrates broad substrate applicability, successfully synthesizing oxime ethers from aromatic, aliphatic, and heteroatom-containing oximes and alcohols with high yields, often exceeding 90%.
Q: Is the solvent system used in this process environmentally friendly?
A: Absolutely. The process utilizes green solvents like dimethyl carbonate (DMC), which has low toxicity and high biodegradability, replacing hazardous chlorinated solvents commonly used in conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxime Ether Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain leadership in the competitive fine chemicals sector. Our team of expert chemists has thoroughly analyzed the potential of the heteropolyacid-catalyzed oxime ether synthesis described in CN114394913B and is fully prepared to leverage this knowledge for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of oxime ether derivatives meets the highest international standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to explore how this green catalytic method can optimize your specific supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production needs, demonstrating the economic viability of switching to this metal-free process. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your chemical manufacturing operations.
