Advanced Synthesis of Paroxetine Key Intermediates: Transforming Waste into Value for Global Pharma Supply Chains
Advanced Synthesis of Paroxetine Key Intermediates: Transforming Waste into Value for Global Pharma Supply Chains
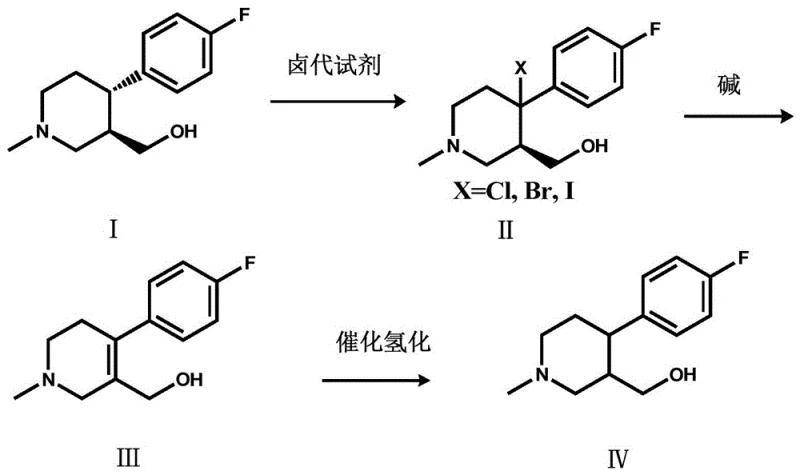
The pharmaceutical industry constantly seeks methods to optimize synthetic routes, particularly for high-volume antidepressants like Paroxetine hydrochloride. Patent CN109020872B introduces a transformative approach to synthesizing a critical key intermediate, specifically focusing on the valorization of waste streams. Traditionally, the production of chiral drugs generates significant amounts of the undesired enantiomer, which is often discarded or sold at a loss. This patent details a robust three-step chemical sequence that converts the unwanted (+)-trans-4-(4-fluorophenyl)-3-hydroxymethyl-1-methylpiperidine into the racemic (+/-)-trans-4-(4-fluorophenyl)-3-hydroxymethyl-1-methylpiperidine. By effectively recycling the non-active isomer back into a usable racemic pool, this technology offers a compelling solution for enhancing raw material utilization and reducing the environmental footprint of API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the conventional synthesis of Paroxetine hydrochloride, the process typically relies on chiral resolution or asymmetric synthesis to obtain the pharmacologically active (-)-trans enantiomer. A major bottleneck in these traditional workflows is the generation of the (+)-trans enantiomer as a stoichiometric byproduct. Historically, this (+)-isomer has been treated as chemical waste because it lacks the specific spatial configuration required for the drug's therapeutic effect. Disposing of this material not only represents a direct financial loss of expensive starting materials but also incurs additional costs related to waste treatment and environmental compliance. Furthermore, relying solely on fresh raw materials for the active isomer places unnecessary strain on the supply chain, creating volatility in pricing and availability for manufacturers who cannot leverage their entire output stream efficiently.
The Novel Approach
The methodology disclosed in CN109020872B breaks this cycle of waste through a clever sequence of functional group manipulations. Instead of discarding the (+)-isomer, the process subjects it to benzylic halogenation, followed by an elimination reaction to form an alkene intermediate, and finally, a catalytic hydrogenation. This sequence effectively erases the stereochemical information at the chiral center during the formation of the planar alkene intermediate, allowing the subsequent hydrogenation to produce a racemic mixture. This racemic product can then be fed back into the resolution process or utilized in synthetic routes that accept the racemate. This approach transforms a liability into an asset, fundamentally altering the economics of the production line by maximizing the yield of usable intermediate from every batch of starting material processed.
Mechanistic Insights into Radical Halogenation and Catalytic Hydrogenation
The core of this synthetic strategy lies in the precise control of radical chemistry and catalytic reduction. The first step involves a radical benzylic halogenation where a halogen atom (Cl, Br, or I) is introduced at the benzylic position of the piperidine ring. Using initiators like dibenzoyl peroxide (BPO) or azobisisobutyronitrile (AIBN), the reaction proceeds via a radical chain mechanism. The choice of halogenating agent, such as N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), is critical for controlling selectivity and preventing over-halogenation. The solvent system, often comprising dichloromethane or tetrahydrofuran, plays a vital role in stabilizing the radical intermediates and ensuring homogeneous reaction conditions. This step converts the stable alcohol precursor into a reactive halide, setting the stage for the subsequent elimination.
Following halogenation, the process employs a base-mediated elimination to generate a double bond, effectively creating a planar sp2-hybridized carbon center which destroys the original chirality. Strong organic bases like N,N-diisopropylethylamine (DIPEA) or DBU are preferred to drive this E2 elimination efficiently without affecting other sensitive functional groups on the molecule. The final step is a catalytic hydrogenation using noble metal catalysts such as palladium on carbon (Pd/C) or platinum on carbon (Pt/C). Under hydrogen pressures ranging from 0.5 MPa to 3.5 MPa, the alkene is reduced back to a saturated piperidine ring. Since the hydrogen can attack the planar double bond from either face with roughly equal probability, the result is a racemic mixture. This mechanistic pathway ensures high purity and minimizes the formation of complex impurities, making it highly suitable for GMP manufacturing environments.
How to Synthesize (+/-)-trans-4-(4-fluorophenyl)-3-hydroxymethyl-1-methylpiperidine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to ensure safety and reproducibility. The process begins with the dissolution of the (+)-isomer in a suitable solvent, followed by the controlled addition of the halogenating agent and initiator. Temperature control is paramount during the exothermic halogenation phase to prevent runaway reactions. Once the halide is formed and isolated, it is subjected to elimination conditions using a stoichiometric amount of base at elevated temperatures. The resulting alkene is then purified and transferred to a hydrogenation reactor.
- Perform benzylic halogenation on (+)-trans-4-(4-fluorophenyl)-3-hydroxymethyl-1-methylpiperidine using NBS or NCS with a radical initiator like BPO in solvents such as dichloromethane or THF.
- Conduct an elimination reaction on the resulting halogenated compound using a strong base like DIPEA or DBU in solvents like 1,2-dichloroethane to form the alkene intermediate.
- Execute catalytic hydrogenation on the alkene intermediate using Pd/C or Pt/C catalyst under hydrogen pressure (0.5-3.5 MPa) in ethanol or methanol to obtain the final racemic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic advantages beyond simple chemistry. The primary benefit is the drastic improvement in raw material efficiency. By converting what was previously waste into a valuable feedstock, manufacturers can significantly reduce the volume of fresh starting materials required per kilogram of final API produced. This reduction in material intensity directly correlates to lower variable costs and reduced exposure to fluctuations in the global market prices of fluorinated building blocks and piperidine derivatives. Furthermore, minimizing waste generation simplifies the environmental compliance burden, potentially lowering the costs associated with hazardous waste disposal and regulatory reporting.
- Cost Reduction in Manufacturing: The ability to recycle the unwanted (+)-enantiomer back into the production cycle effectively increases the overall yield of the synthesis without requiring new capital investment in reactors. This internal recycling loop means that for every ton of active ingredient produced, the consumption of upstream precursors is substantially lowered. Additionally, the reagents used in this process, such as NBS and common organic bases, are commodity chemicals available from multiple global suppliers, ensuring competitive pricing and avoiding reliance on single-source proprietary catalysts that often carry high price premiums.
- Enhanced Supply Chain Reliability: Diversifying the source of intermediates is a key risk mitigation strategy. By enabling the use of the racemic mixture derived from waste streams, manufacturers are less dependent on the strict stereochemical purity of incoming raw materials. This flexibility allows supply chain teams to source cheaper racemic starting materials if available, or to buffer against shortages of specific chiral pools. The robustness of the reaction conditions, which tolerate a range of solvents and temperatures, further ensures that production can continue even if specific grades of solvents are temporarily unavailable, thereby enhancing overall supply continuity.
- Scalability and Environmental Compliance: The process utilizes standard unit operations familiar to any contract development and manufacturing organization (CDMO). The hydrogenation step, while requiring pressure equipment, is a mature technology widely available in modern facilities. From an environmental perspective, the "waste-to-value" proposition aligns perfectly with green chemistry principles. Reducing the mass of waste solvent and chemical byproducts lowers the facility's E-factor (mass of waste per mass of product), which is increasingly becoming a key performance indicator for sustainable manufacturing and a requirement for partnerships with top-tier pharmaceutical innovators.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patent is essential for evaluating its fit within your existing manufacturing portfolio. The following questions address common concerns regarding the practical application of this waste-conversion technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for technical due diligence.
Q: How does this patent address the issue of waste in Paroxetine synthesis?
A: The patent describes a method to chemically convert the unwanted (+)-trans isomer, typically a waste byproduct in chiral resolution processes, back into the racemic (+/-)-mixture. This allows the material to be re-entered into the production cycle, significantly reducing raw material waste and improving overall atom economy.
Q: What are the critical reaction conditions for the halogenation step?
A: The halogenation step preferably uses radical initiators like BPO or AIBN with halogenating agents such as NBS or NCS. The reaction is typically conducted in solvents like dichloromethane or THF at temperatures between 20°C and 60°C, ensuring high conversion rates while minimizing side reactions.
Q: Is this process scalable for commercial manufacturing?
A: Yes, the process utilizes standard industrial unit operations including radical substitution, elimination, and catalytic hydrogenation. The use of common solvents like ethanol and robust catalysts like Pd/C suggests high feasibility for scale-up from pilot plants to multi-ton commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paroxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the competitive landscape of antidepressant manufacturing. Our technical team has extensively analyzed the pathway described in CN109020872B and possesses the expertise to implement this waste-recycling strategy at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for downstream API synthesis.
We invite you to collaborate with us to optimize your supply chain for Paroxetine intermediates. By leveraging our technical capabilities, you can achieve substantial cost savings and improve your sustainability metrics. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term production goals.
