Optimizing Paroxetine Intermediate Production: A Technical Analysis of Diborane Reduction Protocols
Optimizing Paroxetine Intermediate Production: A Technical Analysis of Diborane Reduction Protocols
Introduction: Redefining Efficiency in Antidepressant Intermediate Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with operational safety, particularly for high-volume antidepressant agents. Patent CN1119012A presents a pivotal advancement in the preparation of arylpiperidinemethanols, specifically targeting the critical intermediate for Paroxetine. This intellectual property outlines a novel reduction strategy that replaces traditional, hazardous hydride reagents with an in situ diborane generation system. By shifting the paradigm from lithium aluminum hydride to a boron-based reduction protocol, the process addresses fundamental bottlenecks in thermal management and reagent handling that have long plagued the commercial synthesis of complex heterocyclic amines. For R&D directors and process engineers, this represents a significant opportunity to refine impurity profiles while enhancing the overall safety quotient of the manufacturing suite. The technical nuances of this approach suggest a pathway that is not only chemically elegant but also industrially pragmatic, offering a reliable foundation for scaling complex pharmaceutical intermediates.
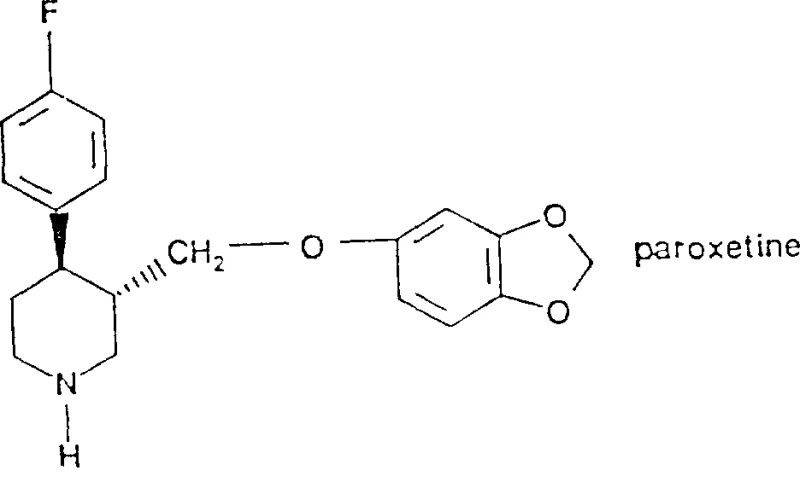
The structural complexity of Paroxetine demands precise stereochemical control and functional group tolerance during synthesis. The patent details the conversion of a piperidine-2,6-dione derivative into the corresponding hydroxymethyl piperidine, a transformation that is notoriously sensitive to reaction conditions. Conventional methods often struggle with the selectivity required to reduce specific carbonyl functionalities without affecting the nitrogen-containing ring or causing racemization. The methodology described in CN1119012A leverages the unique reactivity of diborane to achieve this selectivity under relatively mild conditions. This is particularly relevant for procurement and supply chain stakeholders who require consistent quality across batches. By adopting this refined synthetic route, manufacturers can mitigate the risks associated with exothermic runaways and reagent instability, thereby securing a more predictable supply chain for this high-value active pharmaceutical ingredient intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of lactams and esters to amines and alcohols in this chemical class has relied heavily on lithium aluminum hydride (LiAlH4). While chemically effective on a small laboratory scale, LiAlH4 presents severe challenges when translated to commercial manufacturing environments. The reagent is pyrophoric, requiring strictly anhydrous conditions and specialized handling equipment to prevent ignition upon exposure to atmospheric moisture. Furthermore, the reduction reaction with LiAlH4 is highly exothermic, generating significant heat that can be difficult to dissipate in large-scale reactors. This thermal burden creates process control vulnerabilities, where localized hot spots can lead to decomposition, increased impurity formation, or even safety incidents. For supply chain heads, the reliance on such hazardous materials complicates logistics, storage, and waste disposal, driving up the total cost of ownership. The necessity for rigorous quenching procedures to destroy excess hydride also adds time and complexity to the batch cycle, reducing overall plant throughput and increasing the potential for human error during the workup phase.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes diborane generated in situ from sodium borohydride and a Lewis acid source, such as boron trifluoride etherate or hydrogen chloride gas. This strategic shift fundamentally alters the safety and efficiency profile of the reaction. Sodium borohydride is significantly more stable and easier to handle than lithium aluminum hydride, reducing the hazard classification of the raw materials inventory. The in situ generation allows for the controlled release of the active reducing species, preventing the accumulation of high concentrations of reactive diborane and moderating the exotherm. This level of control is crucial for maintaining product quality, as it minimizes thermal degradation of the sensitive piperidine scaffold. From a commercial perspective, this method simplifies the engineering requirements for the reactor setup, allowing for the use of standard glass-lined or stainless steel equipment without the need for extreme containment measures. The result is a process that is inherently safer, more economical, and better suited for the rigorous demands of GMP manufacturing, offering a clear competitive advantage in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Diborane-Catalyzed Reduction
The core of this technological advancement lies in the mechanistic behavior of diborane towards the specific functional groups present in the piperidine-2,6-dione substrate. Diborane acts as a potent electrophilic reducing agent, coordinating with the oxygen atoms of the carbonyl and ester groups. Unlike nucleophilic hydride sources that attack the carbonyl carbon directly, diborane's mechanism involves the formation of a triacyloxyborane intermediate, which subsequently undergoes reduction to the alcohol. This pathway exhibits remarkable chemoselectivity, preferentially reducing the carboxyl and lactam functionalities while leaving other sensitive groups intact. For the R&D director, understanding this mechanism is key to optimizing reaction parameters. The patent specifies the use of inert solvents like tetrahydrofuran (THF) or dimethoxyethane (DME), which stabilize the borane complexes and facilitate the reaction kinetics. The precise control of temperature during the generation phase, maintained between 0-5°C, ensures that the diborane is formed at a rate that matches its consumption, preventing side reactions that could arise from excess reagent concentration.
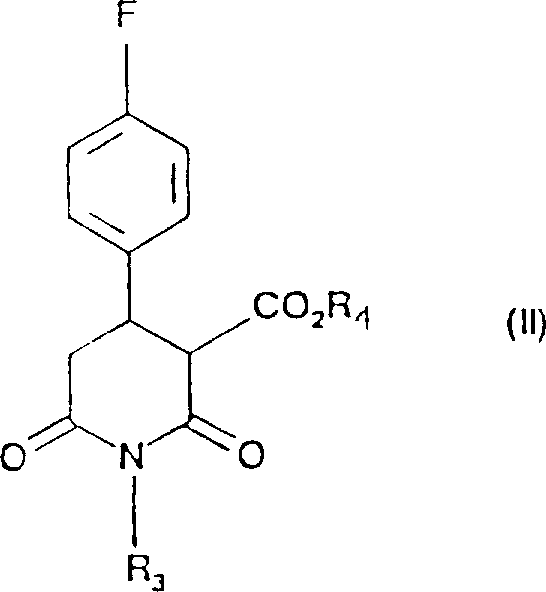
Furthermore, the impurity control mechanism is intrinsically linked to the温和 nature of the diborane reduction. In conventional hydride reductions, over-reduction or ring-opening of the piperidine moiety can occur if the thermal energy is not strictly managed. The novel protocol mitigates this by employing a gradual warming sequence, allowing the reaction to proceed from 0°C to ambient temperatures and finally to a mild elevated range of 20-40°C. This thermal ramp provides the activation energy necessary for the completion of the reduction without crossing the threshold where degradation pathways become dominant. The subsequent workup involving acidic quenching effectively hydrolyzes the boron-ester intermediates, releasing the free alcohol and forming water-soluble borates that are easily removed during the aqueous extraction. This clean separation profile is vital for achieving the high-purity specifications required for pharmaceutical intermediates, ensuring that the final product meets stringent regulatory standards for residual metals and organic impurities.
How to Synthesize (±)-trans-4-(4'-fluorophenyl)-3-hydroxymethyl-N-methylpiperidine Efficiently
Implementing this synthesis requires strict adherence to the sequential addition and temperature control protocols outlined in the patent data. The process begins with the preparation of a slurry of sodium borohydride in an anhydrous solvent, cooled to cryogenic conditions to manage the initial reactivity. The substrate, typically a trans-3-ethoxycarbonyl-4-(4'-fluorophenyl)-N-methylpiperidine-2,6-dione, is then introduced, followed by the slow addition of the boron source to generate the active reducing agent. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up.
- Preparation of the reaction mixture by dissolving sodium borohydride in an inert solvent like THF or DME and cooling to 0-5°C.
- In situ generation of diborane by adding boron trifluoride etherate or hydrogen chloride gas to the borohydride solution in the presence of the substrate.
- Controlled warming of the reaction to 20-40°C followed by acidic quenching and solvent exchange to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this diborane-based protocol offers substantial strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material supply chain. Sodium borohydride is a commodity chemical with a stable global supply, whereas high-purity lithium aluminum hydride can be subject to availability fluctuations and higher price volatility. By eliminating the dependency on specialized, hazardous hydrides, the manufacturing process becomes more resilient to market shocks. Additionally, the reduced safety hazards translate directly into lower insurance premiums and reduced costs associated with hazardous waste disposal. The ability to perform the reaction in common solvents like THF or DME, which are easily recovered and recycled, further enhances the economic efficiency of the process. This alignment with green chemistry principles not only reduces the environmental footprint but also ensures compliance with increasingly stringent environmental regulations, safeguarding the facility against potential operational shutdowns.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous lithium aluminum hydride results in significant direct material cost savings. Moreover, the milder reaction conditions reduce the energy load required for cooling and thermal management, lowering utility costs per batch. The simplified workup procedure, which avoids complex quenching steps and facilitates easier product isolation, reduces labor hours and increases equipment turnover rates. These cumulative efficiencies drive down the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market without compromising margin integrity.
- Enhanced Supply Chain Reliability: The robustness of the sodium borohydride-based system ensures consistent batch-to-batch performance, minimizing the risk of production delays caused by failed batches or safety incidents. The use of widely available solvents and reagents reduces the risk of supply disruptions, ensuring a continuous flow of materials to the production line. This reliability is critical for meeting the just-in-time delivery expectations of downstream API manufacturers, strengthening the partnership between the intermediate supplier and the final drug product maker. The process stability also allows for more accurate forecasting and inventory planning, optimizing working capital utilization.
- Scalability and Environmental Compliance: The inherent safety of the in situ diborane generation makes this process highly scalable from pilot plant to multi-ton commercial production. The reduced generation of hazardous waste and the potential for solvent recovery align with sustainable manufacturing goals, enhancing the corporate social responsibility profile of the production site. This environmental compliance is increasingly a prerequisite for qualifying as a supplier to major multinational pharmaceutical companies, who prioritize sustainability in their vendor selection criteria. The process design facilitates easy integration into existing facilities, minimizing the need for capital expenditure on new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented reduction technology. These insights are derived directly from the experimental data and process descriptions within CN1119012A, providing clarity on the operational feasibility and advantages of the method. Understanding these details is essential for technical teams evaluating the technology for adoption.
Q: Why is diborane preferred over lithium aluminum hydride for this reduction?
A: Diborane offers superior chemoselectivity and process safety. Unlike lithium aluminum hydride, which is pyrophoric and generates intense exotherms difficult to control on large scales, in situ generated diborane allows for milder temperature profiles and eliminates the need for hazardous anhydrous handling protocols, significantly reducing operational risk.
Q: What are the critical impurity controls in this synthesis?
A: The primary impurity concern is the over-reduction or incomplete reduction of the lactam carbonyls. The protocol specifies precise temperature ramping (0-5°C generation, 20-40°C reaction) and stoichiometric control of the boron source to ensure selective reduction of the ester/carbonyl functionality to the alcohol while maintaining the integrity of the piperidine ring structure.
Q: Is this process suitable for multi-ton commercial manufacturing?
A: Yes, the replacement of batch-sensitive reagents like LiAlH4 with in situ diborane generation greatly enhances scalability. The use of common solvents like THF or DME and standard acidic workup procedures aligns well with existing GMP infrastructure, facilitating a smoother technology transfer from pilot plant to commercial production without requiring specialized containment equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paroxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the pharmaceutical supply chain. Our team of expert chemists has extensively evaluated the diborane reduction protocol described in CN1119012A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Paroxetine intermediate meets the highest international standards. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your drug development timelines.
We invite you to collaborate with us to optimize your supply chain for this critical antidepressant intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your project's success. Together, we can ensure the consistent availability of high-quality pharmaceutical intermediates for the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
