Advanced Synthesis of Febuxostat Intermediate: A Cost-Effective Bromination Strategy for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of efficient gout treatments has placed significant spotlight on Xanthine Oxidase Inhibitors, particularly Febuxostat and its critical precursors. Patent CN101863854A, filed in October 2010, introduces a robust and economically viable synthetic methodology for producing 2-(3-cyano-4-isobutoxy)phenyl-4-methyl-5-thiazolecarboxylic acid, a pivotal intermediate in the Febuxostat value chain. This technical disclosure represents a substantial evolution from prior art by strategically replacing expensive halogenating agents with cost-effective alternatives while maintaining exceptional product integrity. The core innovation lies in a six-step sequence originating from readily available 4-hydroxybenzonitrile, navigating through regioselective bromination, etherification, cyanation, and thioamide formation before final cyclization. For R&D directors and process chemists, this route offers a compelling alternative to nitro-reduction pathways or iodine-dependent syntheses, promising streamlined operations and reduced environmental footprint. The detailed experimental embodiments within the patent demonstrate consistent reproducibility across varying scales, indicating a mature process ready for technology transfer. By addressing the specific pain points of raw material volatility and purification complexity found in earlier methods, this patent establishes a new benchmark for manufacturing efficiency in the hyperuricemia therapeutic sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this thiazole-carboxylic acid scaffold has been plagued by reliance on hazardous reagents and inefficient purification protocols that hinder commercial viability. As illustrated in the prior art analysis, Route 1 depends on 3-nitro-4-hydroxybenzaldehyde, necessitating a dangerous diazotization step that poses significant safety risks during scale-up due to the potential for exothermic runaway reactions. Furthermore, Route 2, while shorter in step count, critically relies on column chromatography for purifying key intermediates like 4-isobutoxy-1,3-benzenedicarbonitrile, a technique that is notoriously difficult to translate from laboratory glassware to industrial reactors due to solvent consumption and throughput limitations. Perhaps most critically, the inventors' own previous work, designated as Route 5 in the background section, utilized iodine for the initial halogenation step. While effective, iodine is a high-value commodity with fluctuating market prices and higher volatility, which drastically inflates the Cost of Goods Sold (COGS) for large-scale production. These conventional pathways collectively suffer from either prohibitive safety profiles, unscalable purification requirements, or excessive raw material costs, creating a clear market gap for a more pragmatic manufacturing solution.
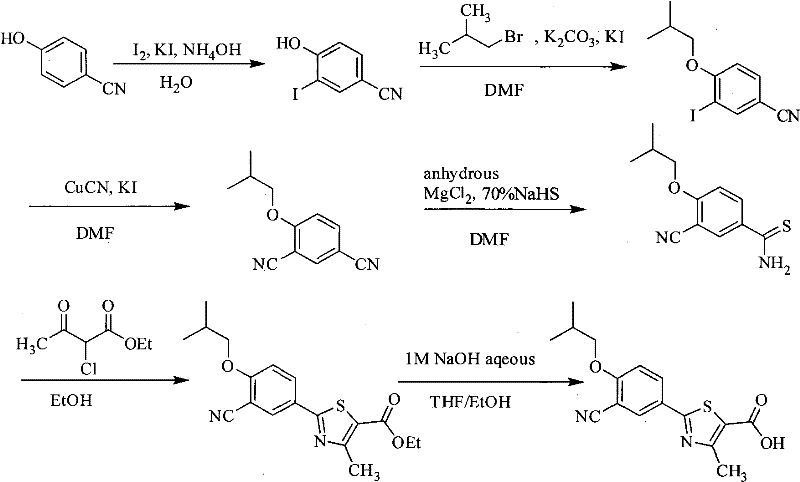
The Novel Approach
The patented methodology presented in CN101863854A decisively breaks these bottlenecks by implementing a bromine-based halogenation strategy coupled with crystallization-driven purification. Instead of utilizing expensive iodine, the process employs molecular bromine in the presence of a catalytic amount of iodine to achieve high-yield regioselective bromination of 4-hydroxybenzonitrile. This substitution alone delivers a profound impact on the economic model of the synthesis, leveraging the abundance and lower cost of bromine reagents. The subsequent steps are meticulously optimized to avoid chromatographic separation; for instance, the alkylation step yields a crude oil that can be directly telescoped into the cyanation reaction after simple aqueous workup. The introduction of anhydrous magnesium chloride during the thioformylation step is another distinct improvement, facilitating the selective conversion of one nitrile group to a thioamide without affecting the other, thereby ensuring high regiochemical fidelity. This holistic approach results in a linear, scalable process where intermediates are isolated as solids or stable oils via filtration and distillation, perfectly aligning with the requirements for GMP-compliant API intermediate manufacturing.
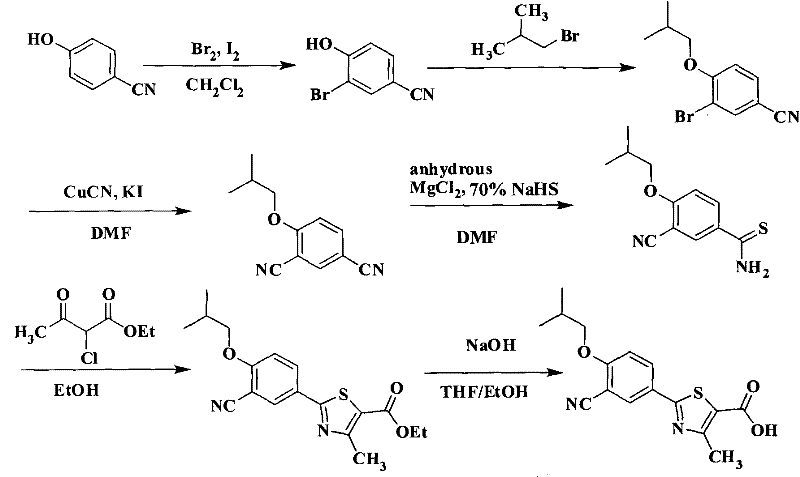
Mechanistic Insights into Regioselective Bromination and Thioformylation
The success of this synthetic route hinges on the precise control of regioselectivity during the initial electrophilic aromatic substitution and the subsequent nucleophilic displacement. In the first step, the hydroxyl group of 4-hydroxybenzonitrile acts as a strong ortho/para director; however, the para position is blocked by the nitrile group, directing the incoming bromine electrophile exclusively to the ortho position relative to the hydroxyl group (position 3). The use of dichloromethane as a solvent at low temperatures (-5 to 0°C) is critical here to suppress poly-bromination and ensure the formation of 3-bromo-4-hydroxybenzonitrile with minimal byproduct formation. Following alkylation, the Rosenmund-von Braun reaction utilizes cuprous cyanide to replace the bromine atom with a nitrile group. This transformation typically requires high temperatures (100-150°C) in polar aprotic solvents like DMF to overcome the activation energy barrier, and the addition of potassium iodide serves as a catalyst to facilitate the halogen exchange, enhancing the reaction rate and yield. The mechanistic elegance continues in the thioformylation step, where sodium hydrosulfide attacks one of the two nitrile groups. The presence of anhydrous magnesium chloride is postulated to coordinate with the nitrile nitrogen, increasing the electrophilicity of the carbon atom and directing the nucleophilic attack to the specific nitrile group ortho to the isobutoxy moiety, thus preventing the formation of bis-thioamides or other regioisomers.
Impurity control is inherently built into this mechanism through the physical properties of the intermediates. The bromination byproduct, if any poly-brominated species form, remains in the mother liquor during the aqueous quench with sodium bisulfite, while the desired mono-bromo product precipitates or extracts cleanly. During the cyanation step, the rigorous washing protocol involving ammonia and brine effectively removes copper residues and unreacted cuprous cyanide, which are common contaminants that can poison downstream catalysts or affect drug safety. The final cyclization with ethyl 2-chloroacetoacetate proceeds via a Hantzsch-like thiazole synthesis mechanism, where the sulfur of the thioamide attacks the alpha-carbon of the chloro-ketone. The high purity of the final product (HPLC ≥ 99.9%) suggests that the crystallization conditions specified—using ethanol or ethyl acetate—are highly effective at rejecting structurally similar impurities, such as the unhydrolyzed ethyl ester or open-chain precursors. This level of purity is achieved without the need for preparative HPLC or column chromatography, validating the robustness of the crystallization drivers embedded in the process design.
How to Synthesize 2-(3-cyano-4-isobutoxy)phenyl-4-methyl-5-thiazolecarboxylic acid Efficiently
The synthesis of this high-value pharmaceutical intermediate requires strict adherence to the reaction parameters outlined in the patent to ensure optimal yield and safety. The process begins with the careful addition of bromine to a cooled solution of 4-hydroxybenzonitrile, a step that demands precise temperature control to manage the exotherm and prevent over-halogenation. Subsequent alkylation with isobutyl bromide utilizes a phase transfer catalyst or potassium iodide to accelerate the SN2 reaction in polar solvents. The cyanation step is the most energy-intensive, requiring prolonged heating in DMF, followed by a meticulous workup to remove heavy metal residues. Finally, the thioformylation and cyclization steps must be monitored to ensure complete conversion before proceeding to the final hydrolysis, which liberates the free carboxylic acid. For a comprehensive understanding of the specific molar ratios, temperature profiles, and isolation techniques required to replicate this process successfully, please refer to the standardized synthesis guide below.
- Bromination of 4-hydroxybenzonitrile using bromine and iodine catalyst in dichloromethane to form 3-bromo-4-hydroxybenzonitrile.
- Alkylation with isobutyl bromide and base to yield 3-bromo-4-isobutoxybenzonitrile.
- Rosenmund-von Braun cyanation using cuprous cyanide to generate 4-isobutoxy-1,3-benzenedicarbonitrile.
- Selective thioformylation using sodium hydrosulfide and anhydrous magnesium chloride.
- Cyclization with ethyl 2-chloroacetoacetate followed by alkaline hydrolysis to obtain the final carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this bromine-based synthetic route offers tangible benefits that extend far beyond simple chemical yield improvements. The primary economic driver is the substitution of iodine with bromine in the initial functionalization step. Iodine is a strategic resource with significant price volatility and supply constraints, whereas bromine is produced in massive quantities globally, ensuring a stable and predictable supply chain. This raw material swap fundamentally alters the cost structure of the intermediate, reducing the exposure to commodity price spikes. Furthermore, the elimination of column chromatography—a unit operation that is notoriously solvent-intensive and labor-heavy—drastically reduces the operational expenditure (OPEX) associated with solvent recovery and waste disposal. The process relies on crystallization and filtration, which are standard, scalable unit operations in any multipurpose chemical plant, thereby lowering the barrier to entry for contract manufacturing organizations (CMOs) and reducing the lead time for technology transfer.
- Cost Reduction in Manufacturing: The economic advantages of this route are anchored in the replacement of high-cost iodine reagents with inexpensive bromine, alongside the removal of chromatographic purification steps. By avoiding column chromatography, the process significantly lowers solvent consumption and waste treatment costs, which are major components of manufacturing expenses in the fine chemical sector. The high yields reported in the patent embodiments, particularly in the alkylation and bromination steps, minimize material loss and maximize the throughput of the final API intermediate. Additionally, the use of common industrial solvents like dichloromethane, DMF, and ethanol simplifies solvent sourcing and recycling logistics, further contributing to a leaner cost profile compared to routes requiring exotic reagents or complex separations.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of commodity-grade starting materials such as 4-hydroxybenzonitrile and isobutyl bromide, which are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the synthetic steps, characterized by wide operating windows for temperature and reaction time as demonstrated in the patent examples, ensures consistent production output even with minor variations in raw material quality. This resilience is crucial for maintaining uninterrupted supply to downstream API manufacturers, especially in the context of increasing regulatory scrutiny on drug shortages. The ability to produce intermediates with >99.9% purity directly from crystallization reduces the need for reprocessing or rework, streamlining the production schedule and ensuring on-time delivery commitments are met reliably.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is designed for industrial feasibility, avoiding the use of explosive diazonium salts found in alternative pathways. The waste streams generated are primarily aqueous salts and organic solvents that can be managed through standard wastewater treatment and distillation protocols, aligning with modern green chemistry principles. The absence of heavy metal catalysts in the final steps (after the copper cyanide removal) simplifies the residual metal testing and clearance required for pharmaceutical registration. Scalability is further enhanced by the fact that all key intermediates are isolable solids or stable oils, allowing for flexible batch scheduling and inventory management. This makes the process adaptable to both pilot-scale campaigns and multi-ton commercial production runs without the need for specialized equipment modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this Febuxostat intermediate, derived directly from the patent specifications and industry best practices. Understanding these details is essential for evaluating the feasibility of integrating this material into your supply chain. The answers reflect the specific process advantages and quality standards established in the referenced intellectual property.
Q: Why is the bromination route preferred over the iodination route for this intermediate?
A: The bromination route utilizes bromine, which is significantly more cost-effective and industrially abundant compared to iodine used in previous methods (Route 5). Additionally, the bromination process described avoids the high volatility and handling hazards associated with large-scale iodination, leading to better process safety and lower raw material costs.
Q: Does this synthesis require column chromatography for purification?
A: No, a key advantage of this patented method is that it eliminates the need for column chromatography purification, which was a bottleneck in other reported routes (such as Route 2). The intermediates are purified via crystallization or extraction, making the process highly suitable for multi-kilogram and ton-scale manufacturing.
Q: What is the achieved purity of the final Febuxostat intermediate?
A: The patent reports that the final product, 2-(3-cyano-4-isobutoxy)phenyl-4-methyl-5-thiazolecarboxylic acid, achieves an HPLC purity of greater than or equal to 99.9%, meeting stringent requirements for pharmaceutical API production without requiring complex downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3-cyano-4-isobutoxy)phenyl-4-methyl-5-thiazolecarboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the bromination route described in CN101863854A are fully realized in our manufacturing facilities. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-(3-cyano-4-isobutoxy)phenyl-4-methyl-5-thiazolecarboxylic acid meets the >99.9% HPLC purity standard required for global regulatory filings. Our commitment to process safety and environmental stewardship ensures that your supply chain remains resilient and compliant with international standards.
We invite you to collaborate with us to optimize your sourcing strategy for this critical gout medication intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this efficient synthetic route can lower your overall project costs. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey from development to commercial launch with reliable, high-quality chemical solutions.
