Advanced Manufacturing of Febuxostat Intermediates via Non-Toxic Cyanation and Mild Oxidation
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for high-volume drugs like Febuxostat. Patent CN102229581B introduces a transformative preparation method for the critical intermediate 2-(3-formyl-4-hydroxy phenyl)-4-methyl-thiazole-5-carboxylic acid ethyl ester and its derivatives. This technology fundamentally shifts the synthetic paradigm by replacing hazardous cyanation agents with a benign hydroxylamine-based system and substituting corrosive formylation reagents with a controlled radical oxidation process. For global supply chains, this represents a pivotal advancement in cost reduction in pharmaceutical intermediates manufacturing, as it mitigates the environmental and safety liabilities associated with traditional heavy metal cyanides and strong acids. The methodology ensures a reliable supply of high-purity precursors essential for the final API synthesis.
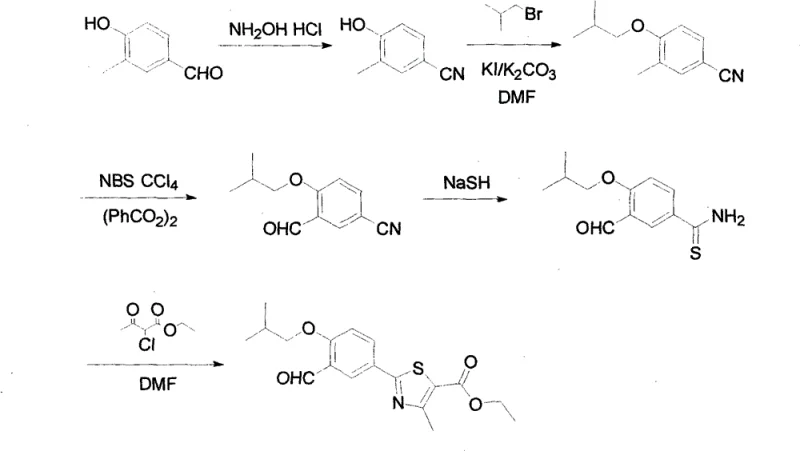
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Febuxostat intermediates has relied on chemically aggressive and environmentally hazardous reagents that pose significant challenges for modern GMP facilities. Prior art, such as the techniques described in US5614520, necessitates the use of potassium cyanide and cuprous cyanide, which are acutely toxic and require stringent waste management protocols to prevent environmental contamination. Furthermore, alternative routes like those in JP1994329647 employ trifluoroacetic acid and urotropine for formylation steps, creating severe corrosion issues for standard stainless steel reactors and increasing maintenance downtime. These conventional methods often involve harsh reaction conditions that can lead to unpredictable impurity profiles, complicating downstream purification and increasing the overall cost of goods sold due to lower yields and higher disposal fees.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these industrial bottlenecks by employing a multi-step sequence starting from readily available 3-methyl-4-hydroxy benzaldehyde. Instead of toxic cyanides, the process utilizes hydroxylamine hydrochloride and sodium formate to effect the conversion of aldehyde groups to nitriles under mild thermal conditions. Additionally, the introduction of the formyl group is achieved through a sophisticated benzylic oxidation using N-bromosuccinimide (NBS) and dibenzoyl peroxide, avoiding the need for corrosive acids entirely. This approach not only enhances operator safety but also simplifies the workup procedures, allowing for easier isolation of the intermediate through standard crystallization techniques, thereby streamlining the path to commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Benzylic Oxidation and Thiazole Cyclization
The core chemical innovation lies in the precise control of functional group transformations on the aromatic ring, specifically the conversion of the methyl group to an aldehyde and the subsequent construction of the thiazole heterocycle. The oxidation step utilizes a radical mechanism initiated by dibenzoyl peroxide, where N-bromosuccinimide selectively brominates the benzylic position of the 3-methyl-4-isobutoxy-benzonitrile. This benzylic bromide intermediate is then hydrolyzed and oxidized to the corresponding aldehyde, 2-isobutoxy-5-cyanobenzaldehyde, with high regioselectivity. Following this, the nitrile group undergoes thioamidation using sodium hydrosulfide, generating a thioamide species that serves as the nucleophile for the final ring closure. The cyclization with ethyl chloroacetoacetate proceeds via a classic Hantzsch-type condensation, forming the 4-methyl-thiazole-5-carboxylate scaffold with excellent structural fidelity.
Impurity control is inherently built into this mechanistic pathway through the use of specific stoichiometric ratios and mild reaction temperatures that suppress side reactions. For instance, the cyanation step using hydroxylamine avoids the formation of heavy metal complexes that are notoriously difficult to remove from the final API, thus ensuring a cleaner impurity profile. The etherification step, catalyzed by potassium iodide in DMF, proceeds efficiently to protect the phenolic hydroxyl group without affecting the nitrile functionality, demonstrating high chemoselectivity. By maintaining reaction temperatures between 50°C and 60°C during critical steps, the process minimizes thermal degradation and polymerization byproducts, resulting in a crude product that requires minimal purification effort to meet high-purity pharmaceutical intermediate specifications.
How to Synthesize 2-(3-Cyano-4-isobutoxy phenyl)-4-methyl-thiazole-5-carboxylic Acid Efficiently
The synthesis protocol described offers a standardized workflow for producing the target thiazole intermediate with consistent quality and yield. The process begins with the conversion of the starting aldehyde to a nitrile, followed by etherification to install the isobutoxy chain, which is crucial for the biological activity of the final drug. Subsequent oxidation of the methyl group regenerates the aldehyde functionality at the correct position for thiazole ring formation. The detailed operational parameters, including solvent choices like DMF and carbon tetrachloride, and specific molar ratios for reagents like isobutyl bromide, are optimized to maximize throughput. For a comprehensive breakdown of the exact experimental conditions and stoichiometry required for replication, please refer to the standardized synthesis steps provided below.
- Convert 3-methyl-4-hydroxy benzaldehyde to the corresponding nitrile using hydroxylamine hydrochloride and sodium formate.
- Perform etherification with isobutyl bromide followed by benzylic oxidation using NBS to generate the aldehyde functionality.
- Execute thioamidation and subsequent cyclization with ethyl chloroacetoacetate to form the thiazole core.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers substantial benefits that directly impact the bottom line and supply continuity for pharmaceutical manufacturers. By eliminating the dependency on controlled substances like sodium cyanide, companies can bypass complex regulatory hurdles and security protocols associated with the procurement and storage of剧毒 chemicals. The shift to milder reagents also translates to extended equipment lifespan, as the absence of trifluoroacetic acid prevents the rapid degradation of reactor linings and valves, leading to significant capital expenditure savings over time. Furthermore, the simplified purification processes reduce solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous catalysts with commodity chemicals like hydroxylamine hydrochloride and sodium formate drastically lowers raw material costs. Additionally, the avoidance of heavy metal catalysts eliminates the need for costly scavenging resins or complex filtration steps required to meet residual metal limits, further driving down processing expenses. The higher selectivity of the new route implies fewer batch failures and less material loss during purification, contributing to an overall more economical production model.
- Enhanced Supply Chain Reliability: Utilizing widely available starting materials such as 3-methyl-4-hydroxy benzaldehyde ensures a stable supply base that is less susceptible to market volatility compared to specialized cyanide sources. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor fluctuations in temperature or mixing rates. This reliability is critical for maintaining continuous production lines and meeting the rigorous delivery timelines demanded by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and conditions that are easily managed in large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The reduction in toxic waste streams simplifies effluent treatment, making it easier for facilities to maintain compliance with local environmental protection agencies. This eco-friendly profile enhances the corporate social responsibility standing of the manufacturer and facilitates smoother regulatory approvals for new drug filings.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility for potential partners. Understanding these details is essential for R&D teams evaluating technology transfer and procurement officers assessing long-term supply contracts.
Q: How does this method avoid toxic cyanide reagents?
A: The process replaces traditional potassium cyanide or cuprous cyanide with a safer system utilizing hydroxylamine hydrochloride and sodium formate for the conversion of aldehydes to nitriles.
Q: What are the advantages regarding equipment corrosion?
A: By eliminating the use of trifluoroacetic acid (TFA) and urotropine typically required for formylation, the process significantly reduces corrosion risks to reactor vessels and piping.
Q: Is this route suitable for large-scale production?
A: Yes, the reaction conditions are mild (typically 50-60°C) and utilize common solvents like DMF and ethanol, making the process highly scalable and operationally simple for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the one described in CN102229581B to deliver superior quality intermediates for the global gout therapy market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2-(3-cyano-4-isobutoxy phenyl)-4-methyl-thiazole-5-carboxylic acid meets the highest industry standards for impurity profiles and residual solvents.
We invite you to collaborate with us to optimize your supply chain for Febuxostat production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project timelines and quality objectives effectively.
